Ahmed Sobhy Basuni, MD
Department of Anesthesiology and Surgical Critical Care, Tanta University Hospital, Tanta (Egypt)
Correspondence: Dr. Ahmed Sobhy Basuni, MD, Department of Anesthesiology and Surgical Critical Care, Tanta University Hospital, Tanta (Egypt)
ABSTRACT
Aim: To investigate the effect of intracuff alkalinized lidocaine on sedative/analgesic requirements for mechanically ventilated patients and its consequence on patient-ventilator interaction.
Methodology: 64 patients who were expected to need ventilatory support for a period of more than 48 hours were randomly divided into two groups. In Group S, the endotracheal tube (ETT) cuffs were inflated with normal saline. In Group L, the ETT cuffs were inflated with equal volumes of lidocaine 2% and sodium bicarbonate 8.4%. The investigator and the surgical intensive care unit staff were blinded to the cuff-filled solutions. Sedation was maintained with propofol and fentanyl infusions in both groups. Total requirement for propofol and fentanyl, frequency and severity of cough and number of ineffective triggering were recorded.
Results: There was a significant reduction (about 30%) in the requirement for propofol and fentanyl in patients who received intracuff alkalinized lidocaine (P < 0.001). The frequency and severity of cough were significantly lower in Group L compared to Group S, and the frequency of ineffective triggering was significantly lower in Group L for both comparisons (P < 0.001).
Conclusion: Intracuff alkalinized lidocaine increases ETT tolerance and hence, decreases the sedatives/analgesic requirements for mechanically ventilated patients, thus improving patient-ventilator synchronization.
Key words: Intracuff lidocaine; Alkalinization; ETT cuff; ETT; Sedatives; Analgesics
Citation: Basuni AS. Intracuff alkalinized lidocaine reduces sedative/analgesic requirement for mechanically ventilated patients. Anaesth Pain & Intensive Care 2013;17(3):228-232
INTRODUCTION
Endotracheal tube (ETT) is a source of discomfort and pain in mechanically ventilated, intensive care unit (ICU) patients, in which ETT has to be retained for a long time. Tracheal tube discomfort is primarily caused by cuff irritation that increases airway secretions and, hence, exacerbates cough and produces more discomfort. To keep the patient in a comfortable state a significant amount of sedatives and analgesics are usually administered, particularly in the first few days, even though their cumulative effect may increase ICU stay, morbidity and mortality. In addition, these drugs may decrease inspiratory muscle efforts and increase patient-ventilator asynchrony, particularly ineffective triggering.1 Therefore, any strategy to decrease sedative/analgesic requirements is appreciated.
The use of lidocaine hydrochloride with or without addition of sodium bicarbonate (i.e., alkalinization) for filling the cuff of ETT instead of air has been studied during general anesthesia.2-5 Intracuff alkalinized lidocaine has been demonstrated to continuously diffuse across the cuff wall, anesthetize the underlying tracheal mucosa and, thereby, reduce the ETT-induced emergence phenomena from general anesthesia, particularly, during surgery of long duration.3-5 The application of this technique in ICU patients has not been described previously, despite that it would seem logical in an attempt to improve ETT tolerance and thereby, to reduce the requirements for sedative/analgesic drugs. Therefore, the aim of the present study was to investigate the effect of intracuff alkalinized lidocaine on sedative/analgesic requirements for ICU patients undergoing mechanical ventilation and the consequence of this technique on patient-ventilator interaction.
METHODOLOGY
Institutional review board approval and written informed consent from patients’ next of kins were obtained. The study was carried out in the Surgical Intensive Care Unit (SICU) of Tanta University Hospital during the period from October 2012 to June 2013. Patients who expected to require ventilatory support for a period of more than 48 hours were consecutively enrolled in the study. The exclusion criteria were age < 18 years, BMI > 35 kg/m2, post cardiac arrest, ventilation through tracheostomy, hemodynamic instability, need for PEEP ≥ 8 cmH2O, excessive amount of respiratory secretions, brain injury, and history of chronic obstructive pulmonary disease, cardiovascular, hepatic or renal disease. Patients were excluded from the study after enrollment, if ventilation was deemed ineffective without muscle relaxation.
Patients were randomly assigned to either Group S (control group), where the ETT cuffs were inflated with normal saline, or Group L (intervention group) where the ETT cuffs were inflated with a mixture of lidocaine 2% (Xylocaine®, AstraZeneca, Paris, France) and sodium bicarbonate (NaHCO3) 8.4% (Sodium bicarbonate 8.4%, Otsuka pharmaceutical Co., Egypt) at a ratio of 1:1 ml. Randomization was carried out according to a computer-generated random numbers table, and syringes containing 10 ml of either saline or alkalinized lidocaine were prepared by the hospital pharmacy. The author and the SICU staff were blinded to the nature of syringes filled with solutions.
Oral endotracheal intubation was performed after administration of an IV bolus of propofol (Diprivan®, Fresenius Kabi, Austria) 0.5–1.5mg/kg and cisatracurium besylate (Nimbex®, GlaxoSmithKline, Egypt) 0.5mg/kg using an ETT tube (3TM, Zhejiang S. Medical Device Co., Ltd, China) 7-7.5 mm inner diameter for women and 7.5-8 mm inner diameter for men. The ETT cuffs (high volume-low pressure type) were inflated with either saline or alkalinized lidocaine according to group assignment at the minimal occlusive volume (i.e., no leakage was detected under controlled ventilation). They were ventilated with an eVent™ ventilator (eVent Medical Ltd, Galway, Ireland) using pressure assisted control ventilation. Ventilatory sittings were adjusted to obtain a tidal volume around 6-8 ml/kg delivered with inspiratory flow rate ≥ 60 L/min. Pressure triggering sensitivity was set between -5 and -1 cmH2O, respiratory rate < 30/min, and PEEP to maintain PaO2 > 90mmHg with an FiO2 < 0.6.
Sedation was maintained with baseline infusions of propofol 0.2 – 1 mg/kg/h to achieve a score of 3 – 4 on the sedation agitation scale (SAS)(6) and fentanyl (Fentanyl, Hameln Pharmaceutical GmbH, Germany) 25-100 µg/h to achieve a score of 0 on the behavioral pain scale (BPS).7 The levels of sedation and analgesia were monitored hourly. If at any time they were assessed to be outside the target levels, nurses achieved the target levels either by administering additional boluses of either propofol 10 mg or fentanyl 25 µg or by altering the infusion rates. Reversible causes of anxiety and agitation including needing explanations/reassurance, excessive light or sounds, and airway obstruction were continuously excluded before altering the infusion rates.
During the first 24 hours of mechanical ventilation, total requirements for propofol and fentanyl were recorded. The episodes of coughing not related to endotracheal suctioning were counted and evaluated according to a three points scale;
1 = mild; single episode of cough/hr that requires no interference;
2 = moderate; two episodes of cough/hr requiring no interference;
3 = severe; more than 2 episodes of cough/hr or cough that require increasing the depth of sedation to maintain effective ventilation)
The ventilator airway pressure-time and flow-time waveforms were captured, downloaded and enlarged for counting the numbers of ineffective triggering; defined as a simultaneous decrease in airway pressure and an increase in airflow without assisted cycle.8
The primary endpoint for the statistical analysis was the total requirements for propofol and fentanyl in the first 24 hours of mechanical ventilation. A previous study by Mallick et al9 required 25 patients per group to determine that inflation of the ETT cuff with alkalinized lidocaine would reduce the sedative/analgesic requirements by 35% with 95% power (α = 0.05). We aimed to include 32 patients per group after predicting a 25% dropout rate. Data were analyzed using SPSS version 20, and presented as mean ± SD, or number (%) as appropriate. After testing data distribution, unpaired student’s t-test was used for comparison of parametric data between the groups. Nonparametric data (patients’ sex, primary diagnosis, survival, and severity of cough) were assessed using Kruskal-Wallis test. A P value < 0.05 was considered statistically significant.
RESULTS
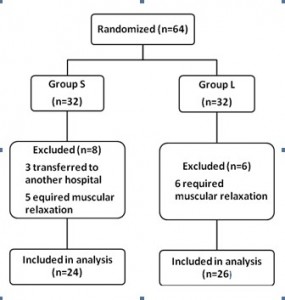
A total number of 64 patients of both sexes were recruited in the study, and 14 out of these were excluded from subsequent analysis: 3 patients in Group S were transferred to another hospital and 11 patients (5 patients in Group S and 6 patients in Group L) required neuromuscular block during the study period because of deterioration of oxygenation. A total of 24 patients remained in Group S and 26 patients in Group L (Figure 1). Patients’ data were comparable in both groups; P > 0.05 (Table 1). They had no significant organ dysfunction except respiratory failure. No cuff ruptures or problems were encountered during endotracheal intubation and there was no difference in the volume of solutions injected into the cuff between groups (7.9 ± 1.9 for the saline group and 6.8 ± 2.4 ml for the alkalinized lidocaine group; P= 0.281; P > 0.05).
Figure 1: Patient trail profile
Table 2 shows that there was a significant reduction (overall reduction about 20–25%) in the requirements for propofol and fentanyl in patients who received intracuff alkalinized lidocaine (P < 0.001). The frequency and severity of cough were significantly lower in Group L compared to Group S and the frequency of ineffective triggering was significantly lower in Group L (P < 0.001 for both comparisons).
Table 1: Patient data
|
Variables |
Group S (n = 24) |
Group L (n = 26) |
p value |
| Age, yr |
48.75 ± 10.41 |
49.15 ± 10.54 |
0.342 |
| Weight, kg |
83.65 ± 8.94 |
84.84 ± 10.16 |
0.642 |
| Sex, M/F |
14 / 10 |
13 / 13 |
0.559 |
| APACHE II score |
15 ± 4 |
18 ± 2 |
0.217 |
| SOFA |
6 ± 3 |
8 ± 2 |
0.189 |
| Primary diagnosis, n (%) | |||
| pneumonia |
7 (29%) |
9 (35%) |
0.683 |
| peritonitis |
8 (33%) |
9 (35%) |
0.925 |
| pancreatitis |
5 (21%) |
4 (15%) |
0.620 |
| Massive blood transfusion |
4 (17%) |
4 (15%) |
0.903 |
| HR, /min |
99.1 ± 13.3 |
100.7 ± 16.6 |
0.273 |
| MAP, mmHg |
52.7 ± 9.40 |
56.1 ± 8.78 |
0.531 |
| CVP, cmH2O |
10 ± 2 |
11 ± 1 |
0.246 |
| RR, /min |
23 ± 3 |
21 ± 2 |
0.311 |
| pH |
7.26 ± 0.06 |
7.29 ± 0.13 |
0.714 |
| SaO2 |
84.6 ± 0.87 |
81.3 ± 1.22 |
0.832 |
| PaO2, mmHg |
59.3 ± 7.60 |
53 ± 11.22 |
0.532 |
| PaCO2, mmHg |
29.3 ± 3.26 |
28.2 ± 5.61 |
0.616 |
| PaO2/FiO2 |
183.2 ± 44 |
193.2 ± 51 |
0.934 |
| VT, ml |
501 ± 46 |
460 ± 55 |
0.681 |
| Paw, cmH2O |
23 ± 4 |
26 ± 5 |
0.457 |
| Pmean, cmH2O |
9 ± 3 |
10 ± 2 |
0.463 |
| Pplat, cmH2O |
26 ± 5 |
28 ± 6 |
0.852 |
| PEEP, cmH2O |
6 ± 2 |
6 ± 1 |
0.553 |
| Duration of ventilation, day |
5 ± 2 |
4 ± 3 |
0.523 |
| ICU stay, day |
10 ± 3 |
9 ± 2 |
0.435 |
| Survival, n (%) |
18 (75%) |
16 (62%) |
0.313 |
APACHE II score: Acute Physiology and Chronic Health Evaluation II score. SOFA: Sequential Organ Failure Assessment. HR (Heart rate), MAP (Mean arterial pressure), CVP (Central venous pressure), RR (Respiratory rate), pH, SaO2 (Oxygen saturation), PaO2 (Arterial oxygen tension), and PaCO2 (Arterial carbon dioxide tension) were recorded immediately before mechanical ventilation. SaO2 and PaO2 were obtained at FiO2 (Inspired oxygen fraction) = 40%. VT: Tidal volume. Paw: Peak airway pressure. Pmean: Mean airway pressure. Pplat: Plateau pressure. PEEP: Positive end expiratory pressure.
Table 2: Propofol and fentanyl requirements, frequency and severity of cough, frequency of ineffective triggering during the 24 hours of the study in both groups:
|
Parameters |
Group S (n = 24) |
Group L (n = 26) |
p–value |
| Propofol, mg |
1743 ± 438 |
1392 ± 512 |
0.001 |
| Fentanyl, µg |
784 ± 226 |
593 ± 341 |
0.001 |
| Cough, n |
22 ± 5 |
12 ± 7 |
0.001 |
| Mild cough |
23% |
76% |
0.034 |
| Moderate cough |
49% |
24% |
|
| Severe cough |
28% |
0 |
|
| Ineffective triggering, n |
4320 ± 672 |
2880 ± 553 |
0.001 |
Patients in Group L appeared to be less troubled by artificial ventilation and pulmonary suctioning compared with those of Group S. There was a trend of reduced hypertension and tachycardia in the alkalinized lidocaine group but it was not significant.
DISCUSSION
The present study demonstrated a significant reduction in the total requirements for propofol and fentanyl and established a lower frequency and severity of cough in the intervention group than the control group. In other words, intracuff alkalinized lidocaine increased ETT tolerance. Furthermore, intracuff alkalinized lidocaine reduced the frequency of ineffective triggering.
Consistent with the current results, Hirota et al. reported that inflation of the tracheostomy tube cuff with 5 ml lidocaine solution 4% significantly reduced tube discomfort in patients undergoing tracheostomy following oral cancer resection as evaluated by a visual analogue scale.10 In other studies, instillation of lidocaine onto tracheal mucosa reduced post-extubation cough on emergence from general anesthesia,11 and requirements of sedative/analgesic in artificially ventilated patients.9 This technique was limited by incomplete effectiveness in controlling of cough reflex as the ETT cuff shields underlying mucosa from exposure to instilled lidocaine, possible bronchospasm due to mucosal stimulation from lidocaine instillation, and need for special ETT (LITA tube; laryngotracheal instillation of local anesthetic).11 Intracuff lidocaine has been shown to overcome the problems experienced with the LITA tube.12-15 However, a recent study failed to demonstrate the effectiveness of intracuff non-alkalinized 4% lidocaine in reducing coughing during emergence from general anesthesia in smokers.16
The ETT cuff is manufactured from a thin semipermeable hydrophobic polyvinyl chloride membrane that allows slow diffusion of lidocaine and blocking of nociceptive stimuli (i.e., irritant and stretch stimuli) caused by the ETT and its cuff.14 Alkalization of lidocaine allowed for quicker and more efficient diffusion, and hence use of small doses of lidocaine (40 mg).3-5 It has been shown in vitro that variations in volume and concentration of NaHCO3 injected into the cuff had no effect on the diffusion of lidocaine.3,5 However, the lidocaine : NaHCO3 (8.4%) ratio (1:1) used in the present study has been shown to result in earlier reduction of anesthesia and spontaneous ventilation at the end of surgery, less cough and restlessness before suctioning and extubation, and marked reduction in post-extubation sore throat during the first postoperative day.5
Lidocaine is known to be absorbed rapidly from the tracheobronchial mucosa. However, for systemic lidocaine to be effective in reducing ETT discomfort a very higher maximal plasma concentration of lidocaine is required (IV lidocaine 2 mg/kg gives plasma lidocaine levels > 3 µg/ml)17 than that attained in case of lidocaine diffusion with 8.4% NaHCO3 (< 0.08 µg/ml),3,4,18 suggesting that improved ETT tolerance after intracuff alkalinized lidocaine is a local rather than a systemic effect.
Ineffective triggering occurs when patient’s effort fails to drop airway pressure below ventilator trigger sensitivity. Though ineffective triggering results mainly from improper ventilatory settings (e.g. inappropriate trigger sensitivity) or abnormal pulmonary mechanics,1,19 sedatives and analgesics have been shown to depress the inspiratory derive and decrease the inspiratory muscle effort, and thus increase ineffective triggering.20,21 In the present study, all patients had acute lung injury/acute respiratory distress syndrome and hence, presumably comparable pulmonary mechanics; also, they were set on low trigger sensitivities (from -5 to -1 cmH2O). Therefore, it is not surprising that the significant difference in frequency of ineffective triggering may be attributed to the difference in sedative/analgesic requirements.1,20 However, the reduction of ineffective triggering may be partly attributed to increased ETT tolerance and patient’s comfort associated with intracuff alkalinized lidocaine.
Monitoring of sedation and analgesia in ICU is inexact. However, the SAS is one of the most valid and reliable tools for assessment of quality and depth of sedation in adult ICU patients.6 Similarly, the BPS is a valid and reliable tool for assessment of pain.7
Saline may not be a true control. Singh et al. reported that use of saline or 2% lidocaine without alkalization as a liquid media for filling the ETT cuff reduced post-extubation reactions.22 However, it was reasonable to use saline as a control as otherwise it would have been difficult to carry out the study in a blinded fashion.
The total dose of lidocaine 68 ± 24 mg over 24 hours in this study (the mean volume used per ETT was 6.8 ± 2.4 ml i.e., 3.4 ± 1.2 ml of 2% lidocaine) is markedly smaller than the lidocaine doses used in the previous studies,12-15 and it is unlikely that toxicity would occur given the dose and duration of this study. On the other hand, lidocaine and NaHCO3 mixture could be irritative if a cuff ruptures. However, in vitro and in vivo studies showed no cuff rupture or obstruction.5,12-15 This is in agreement with the current results. Conversely, some cases of cuff rupture have been reported when lidocaine was used as lubricant or for local anesthesia.23
CONCLUSION
In conclusion, intracuff alkalinized lidocaine improves ETT tolerance leading to reduction of sedatives and analgesic requirements for mechanically ventilated patients and improvement of patient-ventilator synchronization. This technique offers advantages for patients with cardiovascular disease, intracranial or intraocular hypertension, or hyperactive pulmonary disease. Local anesthetics with longer duration of action (e.g. levobupivacaine) may be beneficial but their safety and efficacy should be further investigated.
Acknowledgment: I sincerely thank the staff of SICU of Tanta University Hospital for their help in this study.
Funding: This work was funded by the authors.
IRB: IRB of Faculty of Medicine, Tanta University.
REFERENCES
- Wit MD, Pedram S, Best AM, Epstein SK. Observational study of patient-ventilator asynchrony and relationship to sedation level. Journal of critical care 2009;24(1):74-80. [PubMed] [Free full text] doi: 10.1016/j.jcrc.2008.08.011. Epub 2009 Jan 17.
- Tanaka Y, Nakayama T, Nishimori M, Sato Y, Furuya H. Lidocaine for preventing postoperative sore throat. Cochrane Database Syst Rev. 2009;(3):CD004081.
- Estebe JP, Dollo G, Le Corre P, Le Naoures A, Chevanne F, Le Verge R, et al. Alkalinization of intracuff lidocaine improves endotracheal tube-induced emergence phenomena. Anesth Analg 2002;94(1):227-30. [PubMed]
- Estebe JP, Delahaye S, Le Corre P, Dollo G, Le Naoures A, Chevanne F, et al. Alkalinization of intracuff lidocaine and use of gel lubrication protect against tracheal tube- induced emergence phenomena. Br J Anaesth 2004;92(3):361-6. [PubMed] [Free full text]
- Estebe JP, Gentili M, Le Corre P, Dollo G, Chevanne F, Ecoffey C. Alkalinization of intracuff lidocaine: efficacy and safety. Anesth Analg 2005;101(5):1536-41. [PubMed]
- Barr J, Fraser GL, Puntillo K, Ely EW, Gelinas C, Dasta JF, et al. Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med 2013;41(1):263-306. [PubMed] doi: 10.1097/CCM.0b013e3182783b72
- Payen JF, Bru O, Bosson JL, Lagrasta A, Novel E, Deschaux I, et al. Assessing pain in critically ill sedated patients by using a behavioral pain scale. Crit Care Med 2001;29(12):2258-63. [PubMed]
- Thille AW, Rodriguez P, Cabello B, Lellouche F, Brochard L. Patient-ventilator asynchrony during mechanical ventilation: Prevalence and risk factors. Intensive Care Med 2006;32:1515-22. [PubMed]
- Mallick A, Smith SN, Bodenham AR. Local anesthesia to the airway reduces sedation requirements in patients undergoing artificial ventilation. Br J Anaesth 1996;77(6):731-4. [PubMed] [Free full text]
- Hirota w, kobayashi w, igarashi k, yagihashi y, kimura h, strupish j, et al. lidocaine added to a tracheostomy tube cuff reduces tube discomfort. Can J Anesth 2000;47(5):412-4. [PubMed]
- Gonzalez RM, Bjerke RJ, Drobycki T, Stapelfeldt WH, Green JM, Janowitz MJ, et al. Prevention of endotracheal tube induced coughing during emergence from general anesthesia. Anesth Analg1994;79(4):792-5. [PubMed]
- Sconso JM, Moscicki JC, Difazio CA. In vitro diffusion of lidocaine across endotracheal tube cuffs. Reg Anesth 1990;15(1):37-40. [PubMed]
- Navarro RM, Baughman VL. Lidocaine in the endotracheal tube cuff reduces postoperative sore throat. J Clin Anesth 1997;9(5):394-7. [PubMed]
- Fagan C, Frizelle HP, Laffey J, Hannon V, Carey M. The effects of intracuff lidocaine on endotracheal-tube-induced emergence phenomena after general anesthesia. Anesth Analg 2000;91(1):201-5. [PubMed]
- Altintas F, Bozkurt P, Kaya G, Akkan Gl. Lidocaine 10% in the endotracheal tube cuff: blood concentrations, hemodynamic and clinical effects. Eur J Anaesthesiol 2000;17(7):436-42. [PubMed]
- Wetzel LE, Ancona AL, Cooper AS, Kortman AJ, Loniewski GB, Lebeck LL. The effectiveness of 4% intracuff lidocaine in reducing coughing during emergence from general anesthesia in smokers undergoing procedures lasting less than 1.5 hours. AANA J. 2008;76(2):105-8. [PubMed]
- Yukioka H, Yoshimoto N, Nishimura K, Fujimori M. Intravenous lidocaine as a suppressant of coughing during tracheal intubation. Anesth Analg 1985;64(12):1189-92. [PubMed]
- Dollo G, Estebe JP, Le Corre P, Chevanne F, Ecoffey C, Le Verge R. Endotracheal tube cuffs filled with lidocaine as a drug delivery system: in vitro and in vivo investigations. Eur J Pharm Sci 2001;13(3):319-23. [PubMed]
- Chao DC, Scheinhorn DJ, Stearn-Hassenpflug M. Patient-ventilator trigger asynchrony in prolonged mechanical ventilation. Chest 1997;112(2):1592-9. [PubMed] [Free full text]
- Grasso S, Fanelli V, Cafarelli A, Dalfino L, Ingenito G, Ancona G, et al. Patient ventilator interaction during PSV at different levels of sedation in ALI patients. Intensive Care Med 2004:30:A13.
- Yang L, Huang YT, MacIntyre NR. Patient-ventilator synchrony during pressure-targeted versus flow-targeted small tidal volume assisted ventilation. J Crit Care 2007;22(3):252-7. [PubMed]
- Singh M, Malhotra N, Malhotra S. Tracheal morbidity following tracheal intubation: comparison of air, saline and lignocaine used for inflating cuff. J Anaesthesiol Clin Pharmacol. 2007;23(2):163-7.
- Walmsley AJ, Burville LM, Davis TP. Cuff failure in polyvinyl chloride tracheal tubes sprayed with lignocaine. Anaesthesia 1988;4(5):399-401. [PubMed]