Yusak Mangara Tua Siahaan 1 , Tasya Meidy Pradhana 2 , Yang Yang Endro Arjuna 3 , Vinson Hartoyo 4 , Alexander Erick Purnomo 5 , Rosaria Oktafiani Darmawan 6
Author affiliations:
Background: Occipital neuralgia (ON) is a rare condition characterized by stabbing pain along the greater and lesser occipital nerves. Standard treatments often provide limited relief. Botulinum toxin type A (BoNT-A), previously used for headache, is being explored as a potential treatment for ON. This study aims to assess its safety and effectiveness.
Methodology: A comprehensive literature search was carried out in PubMed, EuropePMC, ScienceDirect, and Google Scholar using keywords such as “Occipital Neuralgia,” “Greater Occipital Nerve,” “Botulinum Toxin A,” “Onabotulinum Toxin A,” “Botulinum Toxin Type-A,” “BoNT-A,” “Botox,” and “Pain,” covering publications up to April 2025. The primary outcome was the Visual Analog Scale (VAS) score, while secondary outcomes included reported adverse events.
Results: This systematic review encompasses six studies with 69 participants. Despite variations in follow-up durations (2, 4, and 12 weeks), all studies reported significant reductions in VAS. Furthermore, no adverse effects related to BoNT-A administration were observed.
Conclusion: In summary, this review indicates that BoNT-A effectively alleviates pain in occipital neuralgia, with benefits observable as early as two weeks post-injection and lasting up to 12 weeks. However, due to variations in dosage, injection techniques, and study methodologies, further well-controlled trials are necessary to confirm its efficacy.
Keywords: Occipital Neuralgia, Botulinum Toxin Type-A, Pain
Citation: Siahaan YMT, Pradhana TM, Arjuna YYE, Hartoyo V, Purnomo AE, Darmawan RO. Efficacy and Safety of Botulinum Toxin Injection for Occipital Neuralgia: A Systematic Review. Anaesth. pain intensive care 2026;30(3):357-364. DOI: 10.35975/apic.v30i3.3175
Recevised: May 29, 2025; Revised: August 29, 2025; Accepted: December 15, 2026
According to the International Headache Society (IHS), occipital neuralgia (ON) is an uncommon yet severe craniofacial pain disorder marked by unilateral or bilateral episodes of sharp, shooting, or stabbing pain in the posterior scalp, aligning with the distribution of the greater occipital nerve (GON) and/or lesser occipital nerve (LON).1 Although occipital neuralgia (ON) is rare, with an incidence of approximately 3.2 per 100,000 and a slightly higher prevalence in females, it can be a debilitating condition that significantly impacts patients' quality of life due to severe pain and functional limitations.2
ON can arise from various causes, including anatomical, traumatic, iatrogenic, and vascular factors. Anatomically, the occipital nerves are vulnerable to compression at several points along their path, such as the C2 nerve root, the obliquus capitis inferior and semispinalis capitis muscles, and the point where they pass through the trapezius muscle or near the occipital artery. Muscle hypertrophy, tension, or spasms in the surrounding muscles can further contribute to nerve compression and prolong ON symptoms.3,4
Numerous conservative and pharmacological treatments have been used in managing ON, including nonsteroidal anti-inflammatory drugs (NSAIDs), anticonvulsants, tricyclic antidepressants, and muscle relaxants.5 However, a significant number of ON patients remain refractory to these conservative therapies, necessitating the exploration of interventional options such as occipital nerve blocks, pulsed radiofrequency (PRF), cryoneurolysis, and botulinum toxin A (BoNT-A) injections.4
BoNT-A, a potent neurotoxin derived from Clostridium botulinum, has emerged as a promising therapeutic agent for ON due to its ability to inhibit acetylcholine release at neuromuscular junctions, leading to muscle relaxation.6 Studies suggest that BoNT-A relieves pain by blocking the release of neuropeptides like CGRP and substance P, thereby reducing neurogenic inflammation.7,8 At the injection site, it prevents their release from sensory nerve endings in the skin and muscles, while centrally, it suppresses glutamate release in the spinal cord, preventing pain sensitization by limiting the activation of second-order neurons and glial cells.9–11
BoNT-A is effective in treating various headaches, including tension-type headaches, cervicogenic headaches, and migraines.12–15 It is also widely used for neuropathic pain conditions such as trigeminal neuralgia, postherpetic neuralgia, diabetic neuropathy, and other chronic pain syndromes.16–18 Given its established role in these conditions, BoNT-A has gained increasing attention as a potential treatment for occipital neuralgia, particularly in patients who remain refractory to conventional therapies.19–24 However, evidence regarding its efficacy and safety in ON remains limited. This study aims to systematically review and analyze the available literature on BoNT-A for ON, evaluating its effectiveness in pain reduction, improvement in quality of life, duration of pain relief, and associated adverse effects to provide a comprehensive understanding of its therapeutic potential in ON management.
The systematic review was performed accordance to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. The review protocol was listed in the PROSPERO International Prospective Register of Systematic Reviews, assigned registration number CRD420251019940.
Data were obtained by conducting searches in the following databases: PubMed, EuropePMC, ScienceDirect, and Google Scholar. The search involved using keywords such as “Occipital Neuralgia,” “Greater Occipital Nerve,” “Botulinum Toxin A,” “Onabotulinum Toxin A,” “Botulinum Toxin Type-A,” “BoNT-A,” “Botox,” and “Pain,” which were combined using Boolean operators like “AND” and “OR.” A comprehensive list of the search terms is provided in Table 1. All searches and data processing were completed by April, 2025. 1
This systematic review included all study types that u assessed the effectiveness of Botulinum Toxin for treating Occipital Neuralgia. To be eligible, studiesneeded to report baseline/pre-treatment Visual Analogue Scale (VAS) scores, post-treatment VAS scores, and secondary outcomes, including any adverse events. Excluded studies were those published in non-English languages, conducted on animals or cadavers, consisting of abstracts or conference papers, or not available in full text. The risk of bias in each study was evaluated using the Risk of Bias in Randomized Trials (RoB 2) tool, the Risk of Bias in Non-randomized Studies (ROBINS-I) tool, and the JBI tool for case series and case reports.
The database search and data extraction were conducted by three authors (TM, YYEA, AEP). Any disagreements were resolved through discussions with a Neurology Expert (YS) until a consensus was reached. The data were organized systematically using Google Sheets. Each study was extracted into several key components, including Study ID, Country, Sample Size, Median Age, Potential Causes of ON, ON Duration, Follow-up, BTX-A Dosage, Injection Location and Technique, Side of ON, Pre- and Post-treatment VAS Scores, and Adverse Effects.
3.1. Literature Search
A total of 1,536 articles were identified from databases including PubMed, Europe PMC, ScienceDirect, and Google Scholar. After removing 649 duplicate articles, 887 remained. These were screened by abstract, resulting in the exclusion of 873 articles. Fourteen articles were then evaluated for eligibility, and after reviewing the full texts, six were excluded due to duplication, and one was excluded for not focusing on Occipital Neuralgia. As a esult, six studies were deemed eligible for further review. Figure 1. illustrates the flow of studies through the PRISMA diagram.
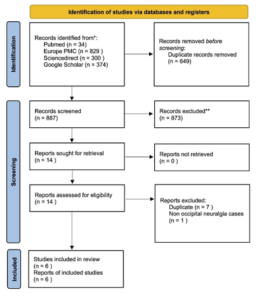
Figure 1: Study flow through the PRISMA
3.2. Study Characteristics
This systematic review included six studies: two pilot studies (pre-RCT), two prospective cohort studies, one case series, and one case report, with a total of 69 patients. Four of the studies were conducted in the USA, one in Brazil, and one in Korea. The majority of participants were female, and most had Occipital Neuralgia lasting for over one year, with follow-up periods ranging from 4 to 12 weeks. The characteristics of the included studies are presented in Table 2. The risk of bias using the RoB 2 Tool, ROBINS-I Tool, JBI for Case Report, and Case Series can be seen in Figure 2, Figure 3, Table 3, and Table 4, respectively.

Figure 2: Risk of bias of the included studies using RoB.2 tool

Figure 3: Risk of bias of the included studies using RoB.1 tool
3.3. Pre and Post Treatment Score
Table 5. presents the results of pre- and post-treatment assessments. All studies demonstrated an improvement in VAS scores comparing pre- and post-treatment following BTX-A administration. In the 12-week follow- up, the VAS scores decreased from 7.66 ± 0.70 to 0.87 ± 0.64, from 5.75 ± 2.37 to 3.41 ± 1.40, and from 9.31 ± 0.89 to 3.68 ± 2.31 in the studies by Kim et al. (2021), Taylor et al. (2008), and Marcolla et al. (2021), respectively. In the 4-week follow-up, the scores improved from 4.1 ± 2.6 to 3.0 ± 2.5, from 8 ± 1.8 to 2 ± 2.7, and from 5 to 0 in the studies by Ea et al. (2016), Kapural et al. (2007), and Ea et al. (2016), respectively. No side effects were reported following BTX-A adminstration.
This systematic review evaluated the efficacy of BoNT-A for ON treatment through a pooled analysis of six studies involving 69 patients. Pain reduction was assessed using VAS scores, comparing post-treatment outcomes to baseline. This study indicated that BoNT-A consistently reduced pain intensity, with significant improvements starting at two weeks post-injection. Notably, three studies followed patients for 12 weeks, highlighting BoNT-A's clinical potential in ON treatment. The most pronounced effect was reported in the study by Kim et al. (2021)(20), where VAS scores decreased from 7.66 ± 0.70 to 0.87 ± 0.64 over a 12-week period. Similar trends were reported by Marcolla et al. (2021) and Taylor et al. (2008), reinforcing the therapeutic potential of BoNT-A.19,21
Despite promising findings, variability in study design and treatment protocols poses challenges in establishing standardized clinical recommendations. The studies included in this review ranged from pilot studies and case series to prospective cohorts, all with relatively small sample sizes. The absence of comparative studies further limits the ability to assess BoNT-A’s effectiveness relative to other treatments, such as nerve blocks or pharmacologic therapy. Significant heterogeneity was observed in study protocols, including variations in BoNT-A dosing (20–120 IU), injection techniques, and follow-up durations (four weeks in three studies and 12 weeks in three studies).
As of this manuscript's writing, there is no established consensus on the optimal BoNT-A dosage or injection technique for ON therapy, as reflected in the reviewed studies. Four out of six studies administered injections directly into the greater occipital nerve (GON), likely based on BoNT-A’s ability to inhibit neurotransmitter release and reduce nociceptive sensitization. However, in two studies by Ea et al., BoNT-A was administered bilaterally at 16, 24, or 31 injection sites, with 5 units per site, including the occipitalis, temporalis, trapezius, and cervical paraspinal muscles. These variations may explain the inconsistent therapeutic outcomes reported, including the generally short duration of analgesia and the relapsing nature of ON. Anatomically, the GON may be compressed at multiple vulnerable sites, including: 1. between the semispinalis capitis and inferior oblique muscles; 2. at the entry or exit points of the semispinalis capitis; 3. at the entrance or exit points of the trapezius muscle; 4. at the fascial insertion of the trapezius near the occipital ridge; and 5. by the occipital artery near the nerve’s distal segment at the skull base.25 These anatomical landmarks may have informed the injection site selection in previous studies.
However, the absence of injections into some key compressive muscles could be attributed to their deep anatomical location and proximity to vital neurovascular structures, posing technical and safety challenges. Given these considerations, we propose incorporating ultrasound-guided (USG) injections targeting adjacent muscles suspected of contributing to nerve compression. This approach may enhance precision, reduce procedural risks, and potentially extend the duration of pain relief in ON management. We suggest future research prioritize well-designed RCTs with larger sample sizes, standardized dosing protocols, consistent outcome measures, and extended follow-up periods to establish BoNT-A as a reliable treatment for ON. Additionally, comparative studies with alternative therapies are essential to better evaluate BoNT-A’s clinical efficacy and optimize treatment strategies.
In conclusion, this systematic review demonstrates that BoNT-A offers promising therapeutic benefits for ON, consistently reducing pain intensity beginning two weeks post-injection and, in some cases, lasting up to 12 weeks. Its analgesic effects are mediated through both peripheral neuromuscular blockade and central modulation of pain-related neurotransmitters and ion channels. However, due to variations in dosage, injection sites, and techniques across existing studies, as well as small sample sizes and short follow-up durations, further well-designed randomized controlled trials with standardized protocols are needed to confirm BoNT-A’s efficacy, determine optimal treatment parameters, and establish its place in routine clinical practice for ON management.
Author affiliations:
- Yusak Mangara Tua Siahaan. Jl. Jenderal Sudirman No.15, Tangerang, 15810, Banten, Indonesia; Email:dryusaks2002@gmail.com; ORCİD: {0000-0002-5916-1453}
- Tasya Meidy Pradhana. Jl. Jenderal Sudirman No.15, Tangerang, 15810, Banten, Indonesia; Email:tasyameidy12@gmail.com;
- Yang Yang Endro Arjuna (Address: Jl. Jenderal Sudirman No.15, Tangerang, 15810, Banten, Indonesia; Email:endroarjuna07@gmail.com;
- Vinson Hartoyo (Address: Taman Ubud Indah, Jl. Ubud Indah VIII No. 69, Lippo Karawaci, Tangerang, Banten 15811, Indonesia. Email: hartoyo@gmail.com)
- Alexander Erick Purnomo. Jenderal Sudirman No.15, Tangerang, 15810, Banten, Indonesia; Email: alexandererick771@gmail.com
- Rosaria Oktafiani Darmawan. Jenderal Sudirman No.15, Tangerang, 15810, Banten, Indonesia; Email: rosariadarmawan@gmail.com
ABSTRACT
Background: Occipital neuralgia (ON) is a rare condition characterized by stabbing pain along the greater and lesser occipital nerves. Standard treatments often provide limited relief. Botulinum toxin type A (BoNT-A), previously used for headache, is being explored as a potential treatment for ON. This study aims to assess its safety and effectiveness.
Methodology: A comprehensive literature search was carried out in PubMed, EuropePMC, ScienceDirect, and Google Scholar using keywords such as “Occipital Neuralgia,” “Greater Occipital Nerve,” “Botulinum Toxin A,” “Onabotulinum Toxin A,” “Botulinum Toxin Type-A,” “BoNT-A,” “Botox,” and “Pain,” covering publications up to April 2025. The primary outcome was the Visual Analog Scale (VAS) score, while secondary outcomes included reported adverse events.
Results: This systematic review encompasses six studies with 69 participants. Despite variations in follow-up durations (2, 4, and 12 weeks), all studies reported significant reductions in VAS. Furthermore, no adverse effects related to BoNT-A administration were observed.
Conclusion: In summary, this review indicates that BoNT-A effectively alleviates pain in occipital neuralgia, with benefits observable as early as two weeks post-injection and lasting up to 12 weeks. However, due to variations in dosage, injection techniques, and study methodologies, further well-controlled trials are necessary to confirm its efficacy.
Keywords: Occipital Neuralgia, Botulinum Toxin Type-A, Pain
Citation: Siahaan YMT, Pradhana TM, Arjuna YYE, Hartoyo V, Purnomo AE, Darmawan RO. Efficacy and Safety of Botulinum Toxin Injection for Occipital Neuralgia: A Systematic Review. Anaesth. pain intensive care 2026;30(3):357-364. DOI: 10.35975/apic.v30i3.3175
Recevised: May 29, 2025; Revised: August 29, 2025; Accepted: December 15, 2026
1. INTRODUCTION
According to the International Headache Society (IHS), occipital neuralgia (ON) is an uncommon yet severe craniofacial pain disorder marked by unilateral or bilateral episodes of sharp, shooting, or stabbing pain in the posterior scalp, aligning with the distribution of the greater occipital nerve (GON) and/or lesser occipital nerve (LON).1 Although occipital neuralgia (ON) is rare, with an incidence of approximately 3.2 per 100,000 and a slightly higher prevalence in females, it can be a debilitating condition that significantly impacts patients' quality of life due to severe pain and functional limitations.2
ON can arise from various causes, including anatomical, traumatic, iatrogenic, and vascular factors. Anatomically, the occipital nerves are vulnerable to compression at several points along their path, such as the C2 nerve root, the obliquus capitis inferior and semispinalis capitis muscles, and the point where they pass through the trapezius muscle or near the occipital artery. Muscle hypertrophy, tension, or spasms in the surrounding muscles can further contribute to nerve compression and prolong ON symptoms.3,4
Numerous conservative and pharmacological treatments have been used in managing ON, including nonsteroidal anti-inflammatory drugs (NSAIDs), anticonvulsants, tricyclic antidepressants, and muscle relaxants.5 However, a significant number of ON patients remain refractory to these conservative therapies, necessitating the exploration of interventional options such as occipital nerve blocks, pulsed radiofrequency (PRF), cryoneurolysis, and botulinum toxin A (BoNT-A) injections.4
BoNT-A, a potent neurotoxin derived from Clostridium botulinum, has emerged as a promising therapeutic agent for ON due to its ability to inhibit acetylcholine release at neuromuscular junctions, leading to muscle relaxation.6 Studies suggest that BoNT-A relieves pain by blocking the release of neuropeptides like CGRP and substance P, thereby reducing neurogenic inflammation.7,8 At the injection site, it prevents their release from sensory nerve endings in the skin and muscles, while centrally, it suppresses glutamate release in the spinal cord, preventing pain sensitization by limiting the activation of second-order neurons and glial cells.9–11
BoNT-A is effective in treating various headaches, including tension-type headaches, cervicogenic headaches, and migraines.12–15 It is also widely used for neuropathic pain conditions such as trigeminal neuralgia, postherpetic neuralgia, diabetic neuropathy, and other chronic pain syndromes.16–18 Given its established role in these conditions, BoNT-A has gained increasing attention as a potential treatment for occipital neuralgia, particularly in patients who remain refractory to conventional therapies.19–24 However, evidence regarding its efficacy and safety in ON remains limited. This study aims to systematically review and analyze the available literature on BoNT-A for ON, evaluating its effectiveness in pain reduction, improvement in quality of life, duration of pain relief, and associated adverse effects to provide a comprehensive understanding of its therapeutic potential in ON management.
2. Research Methodology
The systematic review was performed accordance to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. The review protocol was listed in the PROSPERO International Prospective Register of Systematic Reviews, assigned registration number CRD420251019940.
Data were obtained by conducting searches in the following databases: PubMed, EuropePMC, ScienceDirect, and Google Scholar. The search involved using keywords such as “Occipital Neuralgia,” “Greater Occipital Nerve,” “Botulinum Toxin A,” “Onabotulinum Toxin A,” “Botulinum Toxin Type-A,” “BoNT-A,” “Botox,” and “Pain,” which were combined using Boolean operators like “AND” and “OR.” A comprehensive list of the search terms is provided in Table 1. All searches and data processing were completed by April, 2025. 1
| Table 1: Keyword Search Terms | |
| Source | Search Terms |
| Pubmed | (((Occipital neuralgia) OR (Greater occipital nerve)) AND (((((Botulinum Toxin A) OR (Onabotulinum Toxin A)) OR (Botulinum Toxin Type-A)) OR (BoNT-A)) OR (Botox))) AND (pain) |
| EuropePMC | (((Occipital neuralgia) OR (Greater occipital nerve)) AND (((((Botulinum Toxin A) OR (Onabotulinum Toxin A)) OR (Botulinum Toxin Type-A)) OR (BoNT-A)) OR (Botox))) AND (pain) |
| Sciencedirect | (((Occipital neuralgia) OR (Greater occipital nerve)) AND (((((Botulinum Toxin A) OR (Onabotulinum Toxin A)) OR (Botulinum Toxin Type-A)) OR (BoNT-A)) OR (Botox))) AND (pain) |
| Google Scholar | (((Occipital neuralgia) OR (Greater occipital nerve)) AND (((((Botulinum Toxin A) OR (Onabotulinum Toxin A)) OR (Botulinum Toxin Type-A)) OR (BoNT-A)) OR (Botox))) AND (pain) |
This systematic review included all study types that u assessed the effectiveness of Botulinum Toxin for treating Occipital Neuralgia. To be eligible, studiesneeded to report baseline/pre-treatment Visual Analogue Scale (VAS) scores, post-treatment VAS scores, and secondary outcomes, including any adverse events. Excluded studies were those published in non-English languages, conducted on animals or cadavers, consisting of abstracts or conference papers, or not available in full text. The risk of bias in each study was evaluated using the Risk of Bias in Randomized Trials (RoB 2) tool, the Risk of Bias in Non-randomized Studies (ROBINS-I) tool, and the JBI tool for case series and case reports.
The database search and data extraction were conducted by three authors (TM, YYEA, AEP). Any disagreements were resolved through discussions with a Neurology Expert (YS) until a consensus was reached. The data were organized systematically using Google Sheets. Each study was extracted into several key components, including Study ID, Country, Sample Size, Median Age, Potential Causes of ON, ON Duration, Follow-up, BTX-A Dosage, Injection Location and Technique, Side of ON, Pre- and Post-treatment VAS Scores, and Adverse Effects.
3. RESULTS
3.1. Literature Search
A total of 1,536 articles were identified from databases including PubMed, Europe PMC, ScienceDirect, and Google Scholar. After removing 649 duplicate articles, 887 remained. These were screened by abstract, resulting in the exclusion of 873 articles. Fourteen articles were then evaluated for eligibility, and after reviewing the full texts, six were excluded due to duplication, and one was excluded for not focusing on Occipital Neuralgia. As a esult, six studies were deemed eligible for further review. Figure 1. illustrates the flow of studies through the PRISMA diagram.

Figure 1: Study flow through the PRISMA
3.2. Study Characteristics
This systematic review included six studies: two pilot studies (pre-RCT), two prospective cohort studies, one case series, and one case report, with a total of 69 patients. Four of the studies were conducted in the USA, one in Brazil, and one in Korea. The majority of participants were female, and most had Occipital Neuralgia lasting for over one year, with follow-up periods ranging from 4 to 12 weeks. The characteristics of the included studies are presented in Table 2. The risk of bias using the RoB 2 Tool, ROBINS-I Tool, JBI for Case Report, and Case Series can be seen in Figure 2, Figure 3, Table 3, and Table 4, respectively.
| Table 2. Study Characteristics | |||||||
| Study ID (year) | Study Design | Country | Sample Size (F) | Median Age | Possible ON Causes | ON Duration | Follow up |
| Kim, et al., (2021) | Pilot Study (Pre-RCT) | Korea | 8(5) | 46.3±7.8 | NA | NA | 12 weeks |
| Taylor, et al., (2008) | Pilot Study (Pre-RCT) | USA | 6(4) | 40.38 ± 10.13 | NA | >1 year | 12 weeks |
| Marcolla, et al., (2021) | Prospective Cohort | Brazil | 29(28) | 50.41±11.9 | NA | 11.04±0.02 years | 12 weeks |
| Ea, et al., (2016) | Prospective Cohort | USA | 19(15) | 55.4 | Cervical spondylosis, Cervical Degenerative disk disease, Whiplash injury, Head/neck trauma, C1 fracture, Unknown | 7.2 ± 9.0 years. | 4 weeks |
| Kapural, et al., (2007) | Case Series | USA | 6(5) | 57.2 | Cervical spinal stenosis, whiplash injury, cervical failed back surgery syndrome, unknown | 3.5±1.4 years | 4 weeks |
| Ea, et al., (2016) | Case Report | USA | 1 | 50 | Gunshot Injury | 14 months | 4 weeks |

Figure 2: Risk of bias of the included studies using RoB.2 tool

Figure 3: Risk of bias of the included studies using RoB.1 tool
| Table 3. Risk of bias of included studies using jbi case report | |
| JBI Checklist question | Ea, et al., 2016 |
| Were patient’s demographic characteristics clearly described? | Yes |
| Was the patient’s history clearly described and presented as a timeline? | Yes |
| Was the current clinical condition of the patient on presentation clearly described? | Yes |
| Were diagnostic tests or assessment methods and the results clearly described? | Yes |
| Was the intervention(s) or treatment procedure(s) clearly described? | Yes |
| Was the post-intervention clinical condition clearly described? | Yes |
| Were adverse events (harms) or unanticipated events identified and described? | Unclear |
| Does the case report provide takeaway lessons? | Yes |
| Table 4. Risk of Bias of Included studies using JBI Case Series | |
| JBI Checklist Questions | Kapural, et al., (2007) |
| Were there clear criteria for inclusion in the case series? | Yes |
| Was the condition measured in a standard, reliable way for all participants included in the case series? | Yes |
| Were valid methods used for identification of the condition for all participants included in the case series? | Yes |
| Did the case series have consecutive inclusion of participants? | Yes |
| Did the case series have complete inclusion of participants? | Unclear |
| Was there clear reporting of the demographics of the participants in the study? | Yes |
| Was there clear reporting of clinical information of the participants? | Yes |
| Were the outcomes or follow up results of cases clearly reported? | Yes |
| Was there clear reporting of the presenting site(s)/clinic(s) demographic information? | Yes |
| Was statistical analysis appropriate? | Yes |
3.3. Pre and Post Treatment Score
Table 5. presents the results of pre- and post-treatment assessments. All studies demonstrated an improvement in VAS scores comparing pre- and post-treatment following BTX-A administration. In the 12-week follow- up, the VAS scores decreased from 7.66 ± 0.70 to 0.87 ± 0.64, from 5.75 ± 2.37 to 3.41 ± 1.40, and from 9.31 ± 0.89 to 3.68 ± 2.31 in the studies by Kim et al. (2021), Taylor et al. (2008), and Marcolla et al. (2021), respectively. In the 4-week follow-up, the scores improved from 4.1 ± 2.6 to 3.0 ± 2.5, from 8 ± 1.8 to 2 ± 2.7, and from 5 to 0 in the studies by Ea et al. (2016), Kapural et al. (2007), and Ea et al. (2016), respectively. No side effects were reported following BTX-A adminstration.
| Table 5. Results of the Study | |||||||||
| Study | BTX-A Dosage | Injection Location & Technique | Side | VAS Score | Adverse Effect | ||||
| Unilateral | Bilateral | Baseline | 2 wks | 4 wks | 12 Weeks | ||||
| Kim, et al., (2021) | 20 units per pain site (5 units at each of 4 injection points) | One-third of the virtual line between the inion and mastoid process, plus three points forming an inverted triangle ~3 cm from this site. | NA | NA | 7.66 ± 0.70 | 3.38± 2.50 | 1.13±0.99 | 0.87±0.64 | NR |
| Taylor, et al., (2008) | 50 units reconstituted in 3cc of NS | Inferolateral to the occipital protuberance along the superior nuchal line. | 1 | 5 | 5.75±2.37 | 3.651 ± 2.896 | NA | 3.418±1.40 | NR |
| Marcolla, et al., (2021) | Flasks of 100 units were diluted using 2ml of NS, maximum Dose of 30 units per region and 60 units per patient | Approximately one-third of the distance along an imaginary line from the external occipital protuberance to the central mastoid region, where Tinel's + | 6 | 23 | 9.31±0.89 | NA | NA | 3.68±2.31 | NR |
| Ea, et al., (2016) | 5 units per level | Administered in the occipitalis, temporalis, trapezius, cervical paraspinal, corrugator, procerus, and frontalis muscles. | NA | NA | 4.1 ± 2.6 | NA | 3.0 ± 2.5 | NA | NR |
| Kapural, et al., (2007) | 50 units | Using a stimulating needle with a 1.0 mV initial current, adjusted to ≤0.5 mV at the superior nuchal line. | NA | NA | 8±1.8 | NA | 2±2.7 | NA | NR |
| Ea, et al., (2016) | 120 units divided among 24 injection points | Across 24 sites in the trapezius, occipitalis, temporalis, and cervical paraspinal muscles. | NA | NA | 5 | NA | 0 | NA | NR |
4. DISCUSSION
This systematic review evaluated the efficacy of BoNT-A for ON treatment through a pooled analysis of six studies involving 69 patients. Pain reduction was assessed using VAS scores, comparing post-treatment outcomes to baseline. This study indicated that BoNT-A consistently reduced pain intensity, with significant improvements starting at two weeks post-injection. Notably, three studies followed patients for 12 weeks, highlighting BoNT-A's clinical potential in ON treatment. The most pronounced effect was reported in the study by Kim et al. (2021)(20), where VAS scores decreased from 7.66 ± 0.70 to 0.87 ± 0.64 over a 12-week period. Similar trends were reported by Marcolla et al. (2021) and Taylor et al. (2008), reinforcing the therapeutic potential of BoNT-A.19,21
Despite promising findings, variability in study design and treatment protocols poses challenges in establishing standardized clinical recommendations. The studies included in this review ranged from pilot studies and case series to prospective cohorts, all with relatively small sample sizes. The absence of comparative studies further limits the ability to assess BoNT-A’s effectiveness relative to other treatments, such as nerve blocks or pharmacologic therapy. Significant heterogeneity was observed in study protocols, including variations in BoNT-A dosing (20–120 IU), injection techniques, and follow-up durations (four weeks in three studies and 12 weeks in three studies).
As of this manuscript's writing, there is no established consensus on the optimal BoNT-A dosage or injection technique for ON therapy, as reflected in the reviewed studies. Four out of six studies administered injections directly into the greater occipital nerve (GON), likely based on BoNT-A’s ability to inhibit neurotransmitter release and reduce nociceptive sensitization. However, in two studies by Ea et al., BoNT-A was administered bilaterally at 16, 24, or 31 injection sites, with 5 units per site, including the occipitalis, temporalis, trapezius, and cervical paraspinal muscles. These variations may explain the inconsistent therapeutic outcomes reported, including the generally short duration of analgesia and the relapsing nature of ON. Anatomically, the GON may be compressed at multiple vulnerable sites, including: 1. between the semispinalis capitis and inferior oblique muscles; 2. at the entry or exit points of the semispinalis capitis; 3. at the entrance or exit points of the trapezius muscle; 4. at the fascial insertion of the trapezius near the occipital ridge; and 5. by the occipital artery near the nerve’s distal segment at the skull base.25 These anatomical landmarks may have informed the injection site selection in previous studies.
However, the absence of injections into some key compressive muscles could be attributed to their deep anatomical location and proximity to vital neurovascular structures, posing technical and safety challenges. Given these considerations, we propose incorporating ultrasound-guided (USG) injections targeting adjacent muscles suspected of contributing to nerve compression. This approach may enhance precision, reduce procedural risks, and potentially extend the duration of pain relief in ON management. We suggest future research prioritize well-designed RCTs with larger sample sizes, standardized dosing protocols, consistent outcome measures, and extended follow-up periods to establish BoNT-A as a reliable treatment for ON. Additionally, comparative studies with alternative therapies are essential to better evaluate BoNT-A’s clinical efficacy and optimize treatment strategies.
5. CONCULSION
In conclusion, this systematic review demonstrates that BoNT-A offers promising therapeutic benefits for ON, consistently reducing pain intensity beginning two weeks post-injection and, in some cases, lasting up to 12 weeks. Its analgesic effects are mediated through both peripheral neuromuscular blockade and central modulation of pain-related neurotransmitters and ion channels. However, due to variations in dosage, injection sites, and techniques across existing studies, as well as small sample sizes and short follow-up durations, further well-designed randomized controlled trials with standardized protocols are needed to confirm BoNT-A’s efficacy, determine optimal treatment parameters, and establish its place in routine clinical practice for ON management.
6. REFERENCES
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38(1):1-211. [PubMed] DOI: 1177/0333102417738202
- Koopman JSHA, Dieleman JP, Huygen FJ, de Mos M, Martin CGM, Sturkenboom MCJM. Incidence of facial pain in the general population. Pain. 2009;147(1):122-127. [PubMed] DOI: 1016/j.pain.2009.08.023
- Choi I, Jeon SR. Neuralgias of the head: occipital neuralgia. J Korean Med Sci. 2016;31(4):479. [PubMed] DOI: 3346/jkms.2016.31.4.479
- Pan W, Peng J, Elmofty D. Occipital neuralgia. Curr Pain Headache Rep. 2021;25(9):61. [PubMed] DOI: 1007/s11916-021-00972-1
- Vanelderen P, Lataster A, Levy R, Mekhail N, van Kleef M, Van Zundert J. Occipital neuralgia. Pain Pract. 2010;10(2):137-144. [PubMed] DOI: 1111/j.1533-2500.2009.00355.x
- Sandrini G, De Icco R, Tassorelli C, Smania N, Tamburin S. Botulinum neurotoxin type A for the treatment of pain: not just in migraine and trigeminal neuralgia. J Headache Pain. 2017;18(1):38.[PubMed] DOI: 1186/s10194-017-0744-z
- Oh HM, Chung M. Botulinum toxin for neuropathic pain: a review of the literature. Toxins. 2015;7(8):3127-3154. [PubMed] DOI: 3390/toxins7083127
- Bagues A, Hu J, Alshanqiti I, Chung MK. Neurobiological mechanisms of botulinum neurotoxin-induced analgesia for neuropathic pain. Pharmacol Ther. 2024;259:108668. [PubMed] DOI: 1016/j.pharmthera.2024.108668
- Dolly O. Synaptic transmission: inhibition of neurotransmitter release by botulinum toxins. Headache. 2003;43(s1):16-24. [PubMed] DOI: 1046/j.1526-4610.43.7s.4.x
- Durham PL, Cady R. Insights into the mechanism of onabotulinumtoxinA in chronic migraine. Headache. 2011;51(10):1573-1577. [PubMed] DOI: 1111/j.1526-4610.2011.02022.x
- Graven-Nielsen T, Arendt-Nielsen L. Peripheral and central sensitization in musculoskeletal pain disorders: an experimental approach. Curr Rheumatol Rep. 2002;4(4):313-321. [PubMed] DOI: 1007/s11926-002-0040-y
- Bruloy E, Sinna R, Grolleau JL, Bout-Roumazeilles A, Berard E, Chaput B. Botulinum toxin versus placebo: a meta-analysis of prophylactic treatment for migraine. Plast Reconstr Surg. 2019;143(1):239-250. [PubMed] DOI: 1097/PRS.0000000000005111
- Dhanasekara CS, Payberah D, Chyu JY, Shen CL, Kahathuduwa CN. The effectiveness of botulinum toxin for chronic tension-type headache prophylaxis: a systematic review and meta-analysis. Cephalalgia. 2023;43(3). [PubMed] DOI: 1177/03331024221150231
- Roland SB, Pripp AH, Msomphora MR, Kvarstein G. The efficacy of botulinum toxin A treatment for tension-type or cervicogenic headache: a systematic review and meta-analysis of randomized, placebo-controlled trials. Scand J Pain. 2021;21(4):635-652. [PubMed] DOI:1515/sjpain-2021-0038
- Hazewinkel MHJ, Bink T, Hundepool CA, Duraku LS, Zuidam JM. Nonsurgical treatment of neuralgia and cervicogenic headache: a systematic review and meta-analysis. Plast Reconstr Surg Glob Open. 2022;10(7):e4412. [PubMed] DOI: 1097/GOX.0000000000004412
- Datta Gupta A, Edwards S, Smith J, Snow J, Visvanathan R, Tucker G, et al. A systematic review and meta-analysis of efficacy of botulinum toxin A for neuropathic pain. Toxins. 2022;14(1):36. [PubMed] DOI: 3390/toxins14010036
- Ostrowski H, Roszak J, Komisarek O. Botulinum toxin type A as an alternative way to treat trigeminal neuralgia: a systematic review. Neurol Neurochir Pol. 2019. [PubMed] DOI: 5603/PJNNS.a2019.0030
- Meng F, Peng K, Yang JP, Ji FH, Xia F, Meng XW. Botulinum toxin-A for the treatment of neuralgia: a systematic review and meta-analysis. J Pain Res. 2018;11:2343-2351. [PubMed] DOI: 2147/JPR.S168650
- Marcolla IMG, Camargo CHF, Coutinho L, Ferreira MG, Tiburtino Meira A, Piovesan EJ, et al. Treatment of occipital neuralgia using onabotulinum toxin A. Acta Neurol Scand. 2022;145(2):193-199. [PubMed] DOI: 1111/ane.13533
- Kim H, Jang B, Kim ST. Botulinum toxin type-A (Botulax®) treatment in patients with intractable chronic occipital neuralgia: a pilot study. Toxins. 2021;13(5):332. [PubMed] DOI: 3390/toxins13050332
- Taylor M, Silva S, Cottrell C. Botulinum toxin type-A (BOTOX®) in the treatment of occipital neuralgia: a pilot study. Headache. 2008;48(10):1476-1481. [PubMed] DOI: 1111/j.1526-4610.2008.01089.x
- Kapural L, Stillman M, Kapural M, McIntyre P, Guirgius M, Mekhail N. Botulinum toxin occipital nerve block for the treatment of severe occipital neuralgia: a case series. Pain Pract. 2007;7(4):337-340. [PubMed] DOI: 1111/j.1533-2500.2007.00150.x
- Ea A, Gray T. OnabotulinumtoxinA nerve blocks in the treatment of occipital neuralgia. Pain Stud Treat. 2016;4(3):29-34. DOI: 4236/pst.2016.43005
- Ea A, Gray T. OnabotulinumtoxinA in the treatment of occipital neuralgia following gunshot injury. Pain Stud Treat. 2016;4(4):43-47. DOI: 4236/pst.2016.44007
- de Oca Mora T, Zúñiga-Cordova JS, Castillo-Rangel C, Marín G, Zarate-Calderon C, Borges PGLB, et al. Anatomic localization and compression points of occipital nerves: therapeutic insights using K-means and cadaveric atlas. Oper Neurosurg. 2025;30(1):61-69.[PubMed] DOI: 1227/ons.0000000000001598