Rameesha Musharaf 1 , Sairah Sadaf 2 , Amjad Siddique 3 , Tahreem Saeed 4
Authors affiliations:
Background & objective: Post-operative pain management is crucial for recovery and daily function. The transversus abdominis plane (TAP) block is a common regional anesthesia technique that effectively relieves abdominal wall pain. This study evaluates the effect of combining dexmedetomidine and bupivacaine in TAP blocks for pain control in abdominal surgery patients and the need for rescue analgesia. We assessed the effect of combining dexmedetomidine into bupivacaine in the TAP block on postoperative analgesia.
Methodology: A randomized controlled trial was conducted at Sheikh Zayed Hospital, Rahim Yar Khan, including 80 patients undergoing abdominal surgeries: exploratory laparotomy (20), ileostomy reversal (25), laparoscopic cholecystectomy (12), open cholecystectomy (10), and paraumbilical hernia mesh repair (13). Patients were randomized into Group A (20 mL of 0.25% bupivacaine with 0.25 mcg/kg dexmedetomidine) and Group B (20 mL of 0.25% bupivacaine alone). Ultrasound-guided TAP blocks were performed, and postoperative pain, hemodynamics, sedation, nausea, and rescue analgesia requirements were monitored.
Results: Age and gender were comparable between groups, while BMI was significantly lower in Group A (P = 0.001). Group A showed better pain control on VAS at 6 hours (P = 0.002) and 12 hours (P = 0.025). Sedation levels and rescue analgesia use were similar in both groups; however, patient satisfaction was higher in Group A (70.1% vs. 37.4%, P = 0.003), with a lower incidence of hypertension (10.0% vs. 25.0%, P = 0.011). Group A exhibited elevated SpO2 values prior to TAP block injection (P = 0.048), although no significant differences were seen at subsequent time intervals. The incorporation of dexmedetomidine did not influence postoperative nausea or sedation.
Conclusion: This study illustrates that the combination of dexmedetomidine and bupivacaine in TAP blocks yields improved analgesia and increased patient satisfaction relative to bupivacaine alone in individuals undergoing abdominal surgery.
Keywords: Abdominal Surgery; Anesthesia; Bupivacaine; Dexmedetomidine; Pain Management; TAP Block
Citation: Musharaf R, Sadaf S, Siddique A, Saeed T. Comparison of pain relief by bupivacaine with or without dexmedetomidine in transversus abdominis plane block for postoperative analgesia in patients undergoing abdominal surgeries. Anaesth. pain intensive care 2026;30(3):350-356. DOI: 10.35975/apic.v30i3.3174
Received: December 16, 2025; Revised: January 05, 2026; Accepted: January 06, 2026
Postoperative pain, defined as "an unpleasant sensory and emotional experience associated with actual or potential tissue damage", remains a significant clinical challenge (1 Its perception is highly subjective and influenced by a complex interplay of medical and socioeconomic factors.6
In the context of abdominal surgery, inadequate postoperative analgesia may result in complications including delayed mobility, thromboembolic events, and pulmonary problems, which can extend hospitalization and contribute to atelectasis due to limited chest expansion following abdominal incisions.2,3 Chronic postsurgical pain is a considerable concern, with studies indicating that 54% of patients report pain after hernia repair, and 30% still experience pain two years post-operation.4 The primary goal of postoperative pain management is to mitigate these adverse effects and facilitate recovery. While opioids have traditionally been the cornerstone of treatment, their associated morbidity and mortality have driven a paradigm shift toward multimodal analgesic strategies.5
The American Society of Anesthesiology advocates for this approach, which employs multiple agents targeting different pain pathways to enhance efficacy and reduce side effects.7 This strategy aims not only to alleviate pain but also to improve overall patient outcomes by minimizing stress and managing adverse effects like nausea, vomiting, and sedation.8 Despite these advancements, approximately 80% of surgical patients still experience acute postoperative pain, with up to 75% reporting moderate to severe intensity,9 highlighting a critical need for more effective solutions. The transversus abdominis plane (TAP) block has emerged as a key peripheral nerve block technique for managing somatic pain following abdominal surgery, effectively reducing postoperative opioid consumption and discomfort.10,11 This technique, in conjunction with adjuncts such as dexamethasone, midazolam, and dexmedetomidine, has demonstrated potential in prolonging analgesia while reducing side effects. Various adjuncts, including dexamethasone (for its anti-inflammatory and antiemetic effects), midazolam (for its sedative and anxiolytic properties), and notably, dexmedetomidine (an α2-adrenoceptor agonist for its analgesic and opioid-sparing effects), have been investigated for this purpose;12,13 focusing on evaluating dexmedetomidine in this context. This research assesses the synergistic effects of dexmedetomidine and bupivacaine when combined in patients undergoing abdominal surgery at Sheikh Zayed Hospital, Rahim Yar Khan.
This study evaluates the effects of bupivacaine combined with dexmedetomidine versus bupivacaine alone for postoperative analgesia, adhering to Flick's (2015) research methodology for data collection and analysis. A randomized controlled trial (RCT) was conducted in accordance with the CONSORT standards.14 utilizing a positivist approach to ensure objective data collection and analysis. The study was performed at Sheikh Zayed Medical College and Hospital, selected for accessibility and familiarity, from August 2024 to November 2025, to facilitate comprehensive data collection on heart rate, blood pressure, and side effects.
The primary data collection concentrated on clinical data, including heart rate, mean arterial pressure, Visual Analogue Scale (VAS), and sedation levels following surgery.15 Non-probabilistic consecutive sampling was employed, with individuals chosen according to certain inclusion and exclusion criteria. Participants were randomly allocated into two groups using block randomization (lottery system) with variable block sizes (4 and 6) to ensure balanced group allocation throughout the study. The required sample size of 80 participants was determined a priori based on a power analysis using VAS score estimates from previous literature. Upon obtaining ethical approval, volunteers from the Surgery department undergoing elective surgery received either a TAP block with bupivacaine mixed with dexmedetomidine or bupivacaine alone.
The TAP block was performed under ultrasound guidance using a lateral approach. The dexmedetomidine in combination with lower dose 0.25 mcg/kg was based on safety. This dose has been commonly used and reported in multiple studies investigating dexmedetomidine as a perineural adjuvant for peripheral nerve blocks. It has been shown safe without a substantial increase in the incidence of hemodynamic instability (e.g., bradycardia, hypotension) or excessive sedation, which are concerns associated with higher systemic dose.
To ensure analgesia coverage for the upper abdominal dermatomes, which are not reliably blocked by the lateral approach alone, all patients also received rescue analgesia upon pain complaint. This combined approach aimed to provide comprehensive somatic analgesia for the entire abdominal incision. Post-operative follow-up assessed pain levels, sedation, and side effects.
2.1. Data Analysis
SPSS version 23 was utilized for data analysis, applying T-tests to assess pain reduction (VAS), heart rate, and secondary outcomes. Chi-square tests were employed for categorical data, and stratification was utilized to account for confounders.
The study comprised 80 patients undergoing abdominal surgeries, categorized as follows: 20 exploratory laparotomies, 25 ileostomy reversals, 12 laparoscopic cholecystectomies, 10 open cholecystectomies, and 13 mesh repairs of paraumbilical hernias. A single consultant anesthetist with extensive prior experience in ultrasound-guided regional anesthesia administered all TAP blocks for both study groups to ensure uniform technique and competency. The demographic attributes of the study groups were analogous, with no notable disparities in age or gender distribution. The analysis encompassed age, gender, and BMI (Table 1).
Table 1 illustrates that the mean age was 34.0 years (95% CI: 31.9–36.1) in Group A (Bupivacaine + Dexmedetomidine) and 31.0 years in Group B (Bupivacaine Alone), with no statistically significant difference (P = 0.104), suggesting that age did not affect study outcomes. Figure 1B indicates that Group A comprised 57.5% males and 42.5% females, whereas Group B contained 67.5% males and 32.5% females; the difference was not statistically significant (P = 0.210), demonstrating a balanced gender distribution. Figure 1C illustrates a mean BMI of 28.4 kg/m² for the cohort, signifying an overweight population with a nearly normal variation spanning from 20.5 to 36.3 kg/m². Figure 1D demonstrates that Group A exhibited a mean BMI of 26.7 ± 3.8, while Group B presented a significantly elevated mean BMI of 30.1 ± 4.8 (P = 0.001). Our observation time interval was up to 12 hours. We received the complaint of pain in patients underwent cholecystectomy after 24 hours and used rescue analgesia. This study was not aim to compare pain between procedures but to compare the analgesic efficacy of two drug regimens within the context of a mixed surgical groups, which reflects common clinical practice.
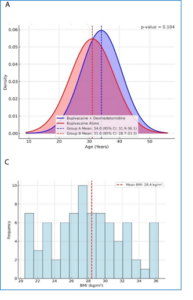
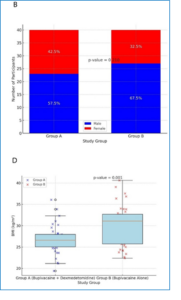
Figure 1. Vital signs and outcomes comparisons (A) Heart rate changes over time (B) Comparison of MAP at different intervals (C) Comparison of hypertension (D) Comparison of SpO2 at different intervals (E) Comparison of VAS at different intervals (F) RSS Scores Comparison for Group A vs. Group B (G) Comparison of Nausea Between Group A and B (H) Comparison of Sedation vs. No Sedation (I) Rescue Analgesia Comparison Between
[Group A = Bupivacaine + Dexmedetomidine; and Group B = Bupivacaine Alone]
Heart rate patterns throughout the period exhibited no significant differences between groups at any interval (Table 2), with all p-values over 0.05. A slight difference during the third hour (P = 0.116) was not statistically significant, suggesting that dexmedetomidine exerted no substantial impact on heart rate. Mean arterial pressure (MAP) was consistent throughout the groups during the study period (Table 2), with no significant differences noted (all P -values > 0.05); the minor fluctuation at six hours (P = 0.075) was likewise not significant, indicating comparable hemodynamic stability. Figure 2C illustrates that hypertension occurred much less often in Group A (10.0%) compared to Group B (25.0%) (P = 0.011), although hypotension was limited in both groups, indicating enhanced hemodynamic regulation in Group A. Oxygen saturation levels in Group A were elevated prior to TAP (Table 2; P = 0.048), but no significant differences were noted subsequently, indicating similar SpO₂ values over time.
Pain scores assessed by VAS (Table 3) were markedly reduced in Group A after 1 hour (P = 0.037), 3 hours (P < 0.001), 6 hours (P = 0.003), and 12 hours (P = 0.026), indicating enhanced analgesia with dexmedetomidine. The sedation ratings evaluated using the RSS (Table 3) were comparable across groups for the majority of time periods (all p-values > 0.05), with a nearly significant difference observed at 1 hour (P = 0.052).
Postoperative nausea was marginally reduced in Group A (10.1%) relative to Group B (17.4%) (Table 4), however the difference lacked statistical significance (p = 0.329). The incidence of sedation was slightly elevated in Group B (57.4%) compared to Group A (47.6%) (Table 4), although this difference was not statistically significant (P = 0.371). Both groups attained 100% effective pain reduction without the need for rescue medications (Table 4), demonstrating exceptional analgesic results in both cohorts.
The study of patient satisfaction revealed that Group A (Bupivacaine + Dexmedetomidine) had a markedly larger percentage of extremely satisfied patients (70.1%) in contrast to Group B (37.4%) (P = 0.003), signifying enhanced overall satisfaction with the anesthetic protocol in Group A.
The incidence of postoperative nausea by gender indicated a greater prevalence in male patients of Group B (23.3%) compared to Group A (10.0%), albeit this disparity was not statistically significant (P = 0.198). Female patients had a greater occurrence in Group B (16.7%) relative to Group A (8.3%), however this difference lacked statistical significance (P = 0.548). Analysis of BMI compared to those in Group A (10.0%), but this difference was not statistically significant (P = 0.186). Data revealed that patients with a BMI ≤25 in Group B experienced more nausea (27.3%) compared to Group A (41.2%) (P = 0.018), whereas no significant difference was noted in patients with BMI >25 (P > 0.999).
Postoperative pain is a prominent surgical consequence that hinders physical function and elevates morbidity rates. Inadequate pain management results in problems including patient suffering, prolonged immobilization, thromboembolic occurrences, and respiratory complications. Earlier studies showed that the transversus abdominis plane block is an essential element of multimodal analgesia for abdominal procedures.9,18 This study sought to assess the effects of Bupivacaine combined with Dexmedetomidine (Group A) to Bupivacaine alone (Group B) on postoperative outcomes, encompassing sedation, analgesia, nausea, and patient satisfaction.
Heart rate and mean arterial pressure (MAP) were consistent during the observation period, with no significant changes between the groups. In previous studies, both anesthesia protocols yielded steady hemodynamic results, signifying cardiovascular safety.19 Comparable findings have been documented in prior research.20
The principal finding indicated a diminished occurrence of postoperative nausea in Group A (Bupivacaine + Dexmedetomidine) relative to Group B (Bupivacaine Alone), especially among male patients. No significant differences were noted in females; however, the reduced nausea rates in males with Dexmedetomidine indicate its antiemetic ability, as corroborated by Zhong et al. (2015).21 Group A had superior pain control at various time intervals, corroborating Dexmedetomidine's efficacy in augmenting postoperative analgesia, consistent with the findings of Imani et al. (2021).22
The sedation levels were comparable across the two groups, exhibiting no significant changes. Group A exhibited somewhat elevated sedation levels; however, this did not influence recovery. Group A exhibited superior patient satisfaction, especially with comfort and pain alleviation, indicating that Dexmedetomidine improved the overall patient experience despite comparable sedative levels.
This research possesses multiple limitations. The sample size was limited, and a larger, more heterogeneous cohort may produce different outcomes. The research concentrated solely on the early postoperative phase, neglecting to evaluate the long-term effects of Dexmedetomidine on pain and drowsiness. Even after accounting for gender and age, additional unrecognized factors, such as comorbidities, may affect outcomes. Furthermore, although BMI was recorded, its influence on nausea, sedation, and pain management was not thoroughly investigated.
The combination of dexmedetomidine into bupivacaine enhanced patient satisfaction, decreased postoperative nausea (particularly in males), and offered superior pain control, without markedly impacting sedation or hemodynamic stability. Additional research involving larger sample sizes and extended follow-up is suggested to validate these findings and investigate the long-term effects of dexmedetomidine in postoperative anesthesia.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
The study utilized the hospital resources only.
10. Acknowledgements
We express our sincere gratitude to the healthcare workers and the anesthesia department for their crucial support and help with the data collecting.
11. Authors’ contribution
RM conceived and designed the study, conducted research, provided research materials, collected and organized data, also analyzed and interpreted data; drafted the initial and final draft SS, AS reviewed the article and TS provided logistical support. SS also acts as a supervisor. All authors have critically reviewed and approved the final draft.
Authors affiliations:
- Rameesha Musharaf, Department of Anesthesia, Critical Care & Pain Management, Sheikh Zayed Medical College/ Hospital, Rahim Yar Khan, Pakistan; Email: rameeshamusharaf96@gmail.com
- Sairah Sadaf, Department of Anesthesia, Critical Care & Pain Management, Sheikh Zayed Medical College/ Hospital, Rahim Yar Khan, Pakistan; Email: sairahbabar@live.com
- Amjad Siddique, Department of Anesthesia, Critical Care & Pain Management, Sheikh Zayed Medical College/ Hospital, Rahim Yar Khan, Pakistan; Email: dramjadsiddique74@gmail.com
- Tahreem Saeed, Aziz Fatimah Medical & Dental College, Faisalabad, Pakistan; Email: tahreemsaeed.799@gmail.com
ABSTRACT
Background & objective: Post-operative pain management is crucial for recovery and daily function. The transversus abdominis plane (TAP) block is a common regional anesthesia technique that effectively relieves abdominal wall pain. This study evaluates the effect of combining dexmedetomidine and bupivacaine in TAP blocks for pain control in abdominal surgery patients and the need for rescue analgesia. We assessed the effect of combining dexmedetomidine into bupivacaine in the TAP block on postoperative analgesia.
Methodology: A randomized controlled trial was conducted at Sheikh Zayed Hospital, Rahim Yar Khan, including 80 patients undergoing abdominal surgeries: exploratory laparotomy (20), ileostomy reversal (25), laparoscopic cholecystectomy (12), open cholecystectomy (10), and paraumbilical hernia mesh repair (13). Patients were randomized into Group A (20 mL of 0.25% bupivacaine with 0.25 mcg/kg dexmedetomidine) and Group B (20 mL of 0.25% bupivacaine alone). Ultrasound-guided TAP blocks were performed, and postoperative pain, hemodynamics, sedation, nausea, and rescue analgesia requirements were monitored.
Results: Age and gender were comparable between groups, while BMI was significantly lower in Group A (P = 0.001). Group A showed better pain control on VAS at 6 hours (P = 0.002) and 12 hours (P = 0.025). Sedation levels and rescue analgesia use were similar in both groups; however, patient satisfaction was higher in Group A (70.1% vs. 37.4%, P = 0.003), with a lower incidence of hypertension (10.0% vs. 25.0%, P = 0.011). Group A exhibited elevated SpO2 values prior to TAP block injection (P = 0.048), although no significant differences were seen at subsequent time intervals. The incorporation of dexmedetomidine did not influence postoperative nausea or sedation.
Conclusion: This study illustrates that the combination of dexmedetomidine and bupivacaine in TAP blocks yields improved analgesia and increased patient satisfaction relative to bupivacaine alone in individuals undergoing abdominal surgery.
Keywords: Abdominal Surgery; Anesthesia; Bupivacaine; Dexmedetomidine; Pain Management; TAP Block
Citation: Musharaf R, Sadaf S, Siddique A, Saeed T. Comparison of pain relief by bupivacaine with or without dexmedetomidine in transversus abdominis plane block for postoperative analgesia in patients undergoing abdominal surgeries. Anaesth. pain intensive care 2026;30(3):350-356. DOI: 10.35975/apic.v30i3.3174
Received: December 16, 2025; Revised: January 05, 2026; Accepted: January 06, 2026
1. INTRODUCTION
Postoperative pain, defined as "an unpleasant sensory and emotional experience associated with actual or potential tissue damage", remains a significant clinical challenge (1 Its perception is highly subjective and influenced by a complex interplay of medical and socioeconomic factors.6
In the context of abdominal surgery, inadequate postoperative analgesia may result in complications including delayed mobility, thromboembolic events, and pulmonary problems, which can extend hospitalization and contribute to atelectasis due to limited chest expansion following abdominal incisions.2,3 Chronic postsurgical pain is a considerable concern, with studies indicating that 54% of patients report pain after hernia repair, and 30% still experience pain two years post-operation.4 The primary goal of postoperative pain management is to mitigate these adverse effects and facilitate recovery. While opioids have traditionally been the cornerstone of treatment, their associated morbidity and mortality have driven a paradigm shift toward multimodal analgesic strategies.5
The American Society of Anesthesiology advocates for this approach, which employs multiple agents targeting different pain pathways to enhance efficacy and reduce side effects.7 This strategy aims not only to alleviate pain but also to improve overall patient outcomes by minimizing stress and managing adverse effects like nausea, vomiting, and sedation.8 Despite these advancements, approximately 80% of surgical patients still experience acute postoperative pain, with up to 75% reporting moderate to severe intensity,9 highlighting a critical need for more effective solutions. The transversus abdominis plane (TAP) block has emerged as a key peripheral nerve block technique for managing somatic pain following abdominal surgery, effectively reducing postoperative opioid consumption and discomfort.10,11 This technique, in conjunction with adjuncts such as dexamethasone, midazolam, and dexmedetomidine, has demonstrated potential in prolonging analgesia while reducing side effects. Various adjuncts, including dexamethasone (for its anti-inflammatory and antiemetic effects), midazolam (for its sedative and anxiolytic properties), and notably, dexmedetomidine (an α2-adrenoceptor agonist for its analgesic and opioid-sparing effects), have been investigated for this purpose;12,13 focusing on evaluating dexmedetomidine in this context. This research assesses the synergistic effects of dexmedetomidine and bupivacaine when combined in patients undergoing abdominal surgery at Sheikh Zayed Hospital, Rahim Yar Khan.
2. METHODOLOGY
This study evaluates the effects of bupivacaine combined with dexmedetomidine versus bupivacaine alone for postoperative analgesia, adhering to Flick's (2015) research methodology for data collection and analysis. A randomized controlled trial (RCT) was conducted in accordance with the CONSORT standards.14 utilizing a positivist approach to ensure objective data collection and analysis. The study was performed at Sheikh Zayed Medical College and Hospital, selected for accessibility and familiarity, from August 2024 to November 2025, to facilitate comprehensive data collection on heart rate, blood pressure, and side effects.
The primary data collection concentrated on clinical data, including heart rate, mean arterial pressure, Visual Analogue Scale (VAS), and sedation levels following surgery.15 Non-probabilistic consecutive sampling was employed, with individuals chosen according to certain inclusion and exclusion criteria. Participants were randomly allocated into two groups using block randomization (lottery system) with variable block sizes (4 and 6) to ensure balanced group allocation throughout the study. The required sample size of 80 participants was determined a priori based on a power analysis using VAS score estimates from previous literature. Upon obtaining ethical approval, volunteers from the Surgery department undergoing elective surgery received either a TAP block with bupivacaine mixed with dexmedetomidine or bupivacaine alone.
The TAP block was performed under ultrasound guidance using a lateral approach. The dexmedetomidine in combination with lower dose 0.25 mcg/kg was based on safety. This dose has been commonly used and reported in multiple studies investigating dexmedetomidine as a perineural adjuvant for peripheral nerve blocks. It has been shown safe without a substantial increase in the incidence of hemodynamic instability (e.g., bradycardia, hypotension) or excessive sedation, which are concerns associated with higher systemic dose.
To ensure analgesia coverage for the upper abdominal dermatomes, which are not reliably blocked by the lateral approach alone, all patients also received rescue analgesia upon pain complaint. This combined approach aimed to provide comprehensive somatic analgesia for the entire abdominal incision. Post-operative follow-up assessed pain levels, sedation, and side effects.
2.1. Data Analysis
SPSS version 23 was utilized for data analysis, applying T-tests to assess pain reduction (VAS), heart rate, and secondary outcomes. Chi-square tests were employed for categorical data, and stratification was utilized to account for confounders.
3. RESULTS
The study comprised 80 patients undergoing abdominal surgeries, categorized as follows: 20 exploratory laparotomies, 25 ileostomy reversals, 12 laparoscopic cholecystectomies, 10 open cholecystectomies, and 13 mesh repairs of paraumbilical hernias. A single consultant anesthetist with extensive prior experience in ultrasound-guided regional anesthesia administered all TAP blocks for both study groups to ensure uniform technique and competency. The demographic attributes of the study groups were analogous, with no notable disparities in age or gender distribution. The analysis encompassed age, gender, and BMI (Table 1).
Table 1 illustrates that the mean age was 34.0 years (95% CI: 31.9–36.1) in Group A (Bupivacaine + Dexmedetomidine) and 31.0 years in Group B (Bupivacaine Alone), with no statistically significant difference (P = 0.104), suggesting that age did not affect study outcomes. Figure 1B indicates that Group A comprised 57.5% males and 42.5% females, whereas Group B contained 67.5% males and 32.5% females; the difference was not statistically significant (P = 0.210), demonstrating a balanced gender distribution. Figure 1C illustrates a mean BMI of 28.4 kg/m² for the cohort, signifying an overweight population with a nearly normal variation spanning from 20.5 to 36.3 kg/m². Figure 1D demonstrates that Group A exhibited a mean BMI of 26.7 ± 3.8, while Group B presented a significantly elevated mean BMI of 30.1 ± 4.8 (P = 0.001). Our observation time interval was up to 12 hours. We received the complaint of pain in patients underwent cholecystectomy after 24 hours and used rescue analgesia. This study was not aim to compare pain between procedures but to compare the analgesic efficacy of two drug regimens within the context of a mixed surgical groups, which reflects common clinical practice.


Figure 1. Vital signs and outcomes comparisons (A) Heart rate changes over time (B) Comparison of MAP at different intervals (C) Comparison of hypertension (D) Comparison of SpO2 at different intervals (E) Comparison of VAS at different intervals (F) RSS Scores Comparison for Group A vs. Group B (G) Comparison of Nausea Between Group A and B (H) Comparison of Sedation vs. No Sedation (I) Rescue Analgesia Comparison Between
[Group A = Bupivacaine + Dexmedetomidine; and Group B = Bupivacaine Alone]
| Table 1: Comparative demographic characteristics of the participants | |||||
| Variable | Metric | Group A | Group B | Total | P-value |
| Gender | Male | 23 (57.5) | 27 (67.5) | 50 (62.5) | 0.21 |
| Female | 17 (42.5) | 13 (32.5) | 30 (37.5) | ||
| Age (years) | 34.0 ± 6.7 | 31.0 ± 7.3 | — | 0.104 | |
| BMI (kg/m²) | Cohort | — | — | 28.4 ± 4.7 | |
| Range | — | — | 20.5–36.3 | ||
| BMI (kg/m²) | Group | 26.7 ± 3.8 | 30.1 ± 4.8 | — | 0.001 |
| BMI: Body Mass Index; Data presented as mean ± SD or n (%); P < 0,05 considered as significant | |||||
Heart rate patterns throughout the period exhibited no significant differences between groups at any interval (Table 2), with all p-values over 0.05. A slight difference during the third hour (P = 0.116) was not statistically significant, suggesting that dexmedetomidine exerted no substantial impact on heart rate. Mean arterial pressure (MAP) was consistent throughout the groups during the study period (Table 2), with no significant differences noted (all P -values > 0.05); the minor fluctuation at six hours (P = 0.075) was likewise not significant, indicating comparable hemodynamic stability. Figure 2C illustrates that hypertension occurred much less often in Group A (10.0%) compared to Group B (25.0%) (P = 0.011), although hypotension was limited in both groups, indicating enhanced hemodynamic regulation in Group A. Oxygen saturation levels in Group A were elevated prior to TAP (Table 2; P = 0.048), but no significant differences were noted subsequently, indicating similar SpO₂ values over time.
| Table 2: Hemodynamic and oxygenation parameters across time intervals | |||||||||
| Time Interval | Heart Rate | P-value | MAP | P-value | SpO₂ | P-value | |||
| Group A | Group B | Group A | Group B | Group A | Group B | ||||
| Before TAP | 77.0 ± 4.9 | 77.5 ± 4.7 | 0.658 | 77.68 ± 7.94 | 76.54 ± 7.76 | 0.523 | 93.15 ± 1.70 | 92.37 ± 1.96 | 0.048 |
| 15 minutes | 80.8 ± 5.7 | 82.0 ± 5.1 | 0.343 | 77.70 ± 6.79 | 76.52 ± 7.36 | 0.46 | 91.93 ± 1.53 | 91.32 ± 1.75 | 0.071 |
| 30 minutes | 84.5 ± 6.1 | 83.8 ± 6.6 | 0.611 | 83.42 ± 8.79 | 83.97 ± 9.04 | 0.783 | 90.75 ± 1.63 | 90.53 ± 1.62 | 0.552 |
| 1 hour | 85.3 ± 8.4 | 84.5 ± 10.0 | 0.698 | 85.47 ± 8.36 | 84.97 ± 9.89 | 0.808 | 90.20 ± 1.53 | 89.95 ± 1.65 | 0.476 |
| 3rd hour | 82.6 ± 14.8 | 87.7 ± 14.6 | 0.116 | 94.52 ± 9.65 | 92.89 ± 9.14 | 0.441 | 89.55 ± 1.61 | 89.88 ± 1.64 | 0.358 |
| 6th hour | 84.3 ± 14.8 | 84.3 ± 14.7 | 0.988 | 93.92 ± 9.43 | 97.92 ± 10.37 | 0.075 | 90.20 ± 1.53 | 89.35 ± 1.65 | 0.062 |
| 9th hour | 85.1 ± 5.8 | 84.4 ± 6.4 | 0.599 | 84.12 ± 7.40 | 84.44 ± 6.76 | 0.838 | 91.13 ± 1.51 | 90.93 ± 1.62 | 0.554 |
| 12th hour | 77.9 ± 7.1 | 76.9 ± 6.9 | 0.536 | 82.59 ± 7.77 | 82.07 ± 7.53 | 0.759 | 92.03 ± 1.54 | 91.83 ± 1.62 | 0.554 |
| TAP: Transversus Abdominis Plane; Data presented as mean ± SD or n (%); P <0.05 considered as significant | |||||||||
Pain scores assessed by VAS (Table 3) were markedly reduced in Group A after 1 hour (P = 0.037), 3 hours (P < 0.001), 6 hours (P = 0.003), and 12 hours (P = 0.026), indicating enhanced analgesia with dexmedetomidine. The sedation ratings evaluated using the RSS (Table 3) were comparable across groups for the majority of time periods (all p-values > 0.05), with a nearly significant difference observed at 1 hour (P = 0.052).
| Table 3: Pain and sedation scores across different time intervals | ||||||
| Time Interval | VAS Group A | VAS Group B | P-value | RSS Group A | RSS Group B | P-value |
| Before TAP | 0 | 0 | NA | 6.56 ± 1.14 | 6.44 ± 1.14 | 0.628 |
| 15 min | 1.04 ± 0.86 | 1.03 ± 0.82 | 0.897 | 6.44 ± 1.18 | 6.56 ± 1.02 | 0.614 |
| 30 min | 1.06 ± 0.74 | 1.14 ± 0.82 | 0.573 | 1.96 ± 0.79 | 2.14 ± 0.85 | 0.349 |
| 1 hour | 1.66 ± 0.65 | 2.01 ± 0.88 | 0.037 | 1.76 ± 0.79 | 2.11 ± 0.78 | 0.052 |
| 3rd hour | 1.41 ± 0.53 | 2.01 ± 0.81 | < 0.001 | 1.94 ± 0.86 | 2.01 ± 0.82 | 0.695 |
| 6th hour | 1.61 ± 0.77 | 2.16 ± 0.83 | 0.003 | 1.84 ± 0.79 | 1.99 ± 0.84 | 0.418 |
| 9th hour | 0.73 ± 0.77 | 1.39 ± 1.07 | 0.003 | 2.29 ± 0.81 | 2.04 ± 0.74 | 0.158 |
| 12th hour | 1.14 ± 0.91 | 1.66 ± 1.11 | 0.026 | 1.86 ± 0.84 | 1.89 ± 0.80 | 0.892 |
| Data presented as mean ± SD or n (%); P <0.05 considered as significant | ||||||
Postoperative nausea was marginally reduced in Group A (10.1%) relative to Group B (17.4%) (Table 4), however the difference lacked statistical significance (p = 0.329). The incidence of sedation was slightly elevated in Group B (57.4%) compared to Group A (47.6%) (Table 4), although this difference was not statistically significant (P = 0.371). Both groups attained 100% effective pain reduction without the need for rescue medications (Table 4), demonstrating exceptional analgesic results in both cohorts.
| Table 4: Comparative categorical clinical outcomes | ||||
| Outcome | Group A | Group B | Total | P-value |
| No hypertension | 36 (90.0) | 29 (72.5) | 65 (81.3) | 0.011 |
| Hypertension | 4 (10.0) | 10 (25.0) | 14 (17.5) | |
| Hypotension | 0 (0.0) | 1 (2.5) | 1 (1.3) | |
| Postoperative nausea | 4 (10.1) | 7 (17.4) | 11 (13.8) | 0.329 |
| Sedation required | 19 (47.6) | 23 (57.4) | 42 (52.5) | 0.371 |
| Rescue analgesia required | 0 (0) | 0 (0) | 0 (0) | NA |
| Data presented as n (%); P < 0,05 considered as significant | ||||
The study of patient satisfaction revealed that Group A (Bupivacaine + Dexmedetomidine) had a markedly larger percentage of extremely satisfied patients (70.1%) in contrast to Group B (37.4%) (P = 0.003), signifying enhanced overall satisfaction with the anesthetic protocol in Group A.
The incidence of postoperative nausea by gender indicated a greater prevalence in male patients of Group B (23.3%) compared to Group A (10.0%), albeit this disparity was not statistically significant (P = 0.198). Female patients had a greater occurrence in Group B (16.7%) relative to Group A (8.3%), however this difference lacked statistical significance (P = 0.548). Analysis of BMI compared to those in Group A (10.0%), but this difference was not statistically significant (P = 0.186). Data revealed that patients with a BMI ≤25 in Group B experienced more nausea (27.3%) compared to Group A (41.2%) (P = 0.018), whereas no significant difference was noted in patients with BMI >25 (P > 0.999).
4. DISCUSSION
Postoperative pain is a prominent surgical consequence that hinders physical function and elevates morbidity rates. Inadequate pain management results in problems including patient suffering, prolonged immobilization, thromboembolic occurrences, and respiratory complications. Earlier studies showed that the transversus abdominis plane block is an essential element of multimodal analgesia for abdominal procedures.9,18 This study sought to assess the effects of Bupivacaine combined with Dexmedetomidine (Group A) to Bupivacaine alone (Group B) on postoperative outcomes, encompassing sedation, analgesia, nausea, and patient satisfaction.
Heart rate and mean arterial pressure (MAP) were consistent during the observation period, with no significant changes between the groups. In previous studies, both anesthesia protocols yielded steady hemodynamic results, signifying cardiovascular safety.19 Comparable findings have been documented in prior research.20
The principal finding indicated a diminished occurrence of postoperative nausea in Group A (Bupivacaine + Dexmedetomidine) relative to Group B (Bupivacaine Alone), especially among male patients. No significant differences were noted in females; however, the reduced nausea rates in males with Dexmedetomidine indicate its antiemetic ability, as corroborated by Zhong et al. (2015).21 Group A had superior pain control at various time intervals, corroborating Dexmedetomidine's efficacy in augmenting postoperative analgesia, consistent with the findings of Imani et al. (2021).22
The sedation levels were comparable across the two groups, exhibiting no significant changes. Group A exhibited somewhat elevated sedation levels; however, this did not influence recovery. Group A exhibited superior patient satisfaction, especially with comfort and pain alleviation, indicating that Dexmedetomidine improved the overall patient experience despite comparable sedative levels.
5. LIMITATIONS
This research possesses multiple limitations. The sample size was limited, and a larger, more heterogeneous cohort may produce different outcomes. The research concentrated solely on the early postoperative phase, neglecting to evaluate the long-term effects of Dexmedetomidine on pain and drowsiness. Even after accounting for gender and age, additional unrecognized factors, such as comorbidities, may affect outcomes. Furthermore, although BMI was recorded, its influence on nausea, sedation, and pain management was not thoroughly investigated.
6. CONCLUSION
The combination of dexmedetomidine into bupivacaine enhanced patient satisfaction, decreased postoperative nausea (particularly in males), and offered superior pain control, without markedly impacting sedation or hemodynamic stability. Additional research involving larger sample sizes and extended follow-up is suggested to validate these findings and investigate the long-term effects of dexmedetomidine in postoperative anesthesia.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
The study utilized the hospital resources only.
10. Acknowledgements
We express our sincere gratitude to the healthcare workers and the anesthesia department for their crucial support and help with the data collecting.
11. Authors’ contribution
RM conceived and designed the study, conducted research, provided research materials, collected and organized data, also analyzed and interpreted data; drafted the initial and final draft SS, AS reviewed the article and TS provided logistical support. SS also acts as a supervisor. All authors have critically reviewed and approved the final draft.
12. REFERENCES
- Hussain A, Hussain A, Feroze R, Akram M, Saleem MA, Hussain AAA, et al. Comparison of bupivacaine and dexmedetomidine versus bupivacaine alone in transversus abdominis plane block for post-operative analgesia. J Rawalpindi Med Coll. 2021;25(1). DOI: 37939/jrmc.v25i1.1552
- Almarakbi WA, Kaki AM. Addition of dexmedetomidine to bupivacaine in transversus abdominis plane block potentiates post-operative pain relief among abdominal hysterectomy patients: A prospective randomized controlled trial. Saudi J Anaesth. 2014;8(2):161-6. [PubMed] DOI: 4103/1658-354X.130683
- Eldegwy M, Alfkey R. Ultrasound-guided TAP Block with or without dexmedetomidine vs. local infiltration of the wound after open herniorrhaphy. A randomized prospective controlled study. J Anesth Clin Res. 2018;9(1). DOI: 4172/2155-6148.1000800
- Aksu R, Patmano G, Biçer C, Emek E, Çoruh AE. Efficiency of bupivacaine and association with dexmedetomidine in transversus abdominis plane block ultrasound guided in postoperative pain of abdominal surgery. Rev Bras Anestesiol. 2018;68(1):49-56. [PubMed] DOI: 1016/j.bjan.2017.04.021
- Horn R, Hendrix JM, Kramer J. Postoperative pain control. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK544298/
- Chou R, Gordon DB, de Leon-Casasola OA, Rosenberg JM, Bickler S, Brennan T, et al. Management of Postoperative Pain: a clinical practice guideline from the American pain society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists' committee on regional anesthesia, executive committee, and administrative council. J Pain. 2016;17(2):131-57. [PubMed] DOI: 1016/j.jpain.2015.12.008
- Karasu D, Yilmaz C, Ozgunay SE, Yalcin D, Ozkaya G. Ultrasound-guided transversus abdominis plane block for postoperative analgesia in laparoscopic cholecystectomy: A retrospective study. North Clin Istanb. 2021;8(1):88-94. [PubMed] DOI: 14744/nci.2020.84665
- Abdel Bar Salem A. Radiofrequency denervation for management of chronic sacroiliac joint pain. Al-Azhar Sci J. 2019;30(1). DOI: 21608/esj.2019.5491.1070
- Ramya Parameswari A, Udayakumar P. Comparison of efficacy of bupivacaine with dexmedetomidine versus bupivacaine alone for transversus abdominis plane block for post-operative analgesia in patients undergoing elective caesarean section. J Obstet Gynaecol India. 2018;68(2):98-103. [PubMed] DOI: 1007/s13224-017-0990-7
- Brady R, Ventham N, Roberts D, Graham C, Daniel T. Open transversus abdominis plane block and analgesic requirements in patients following right hemicolectomy. Ann R Coll Surg Engl. 2012;94(5):327-30. [PubMed] DOI: 1308/003588412X13171221589856
- Qian H, Zhang Q, Zhu P, Zhang X, Tian L, Feng J, et al. Ultrasound-guided transversus abdominis plane block using ropivacaine and dexmedetomidine in patients undergoing caesarian sections to relieve post-operative analgesia: A randomized controlled clinical trial. Exp Ther Med. 2020;20(2):1163-8. [PubMed] DOI: 3892/etm.2020.8781
- El Kenany S, Elshehawi ME, Farid AM, Eid MI. Efficacy of adding midazolam to bupivacaine for transversus abdominis plane block on postoperative analgesia after hysterectomy: a randomized controlled study. Anesth Essays Res. 2019;13(3):522-7. [PubMed] DOI: 4103/aer.AER_95_19
- Fuladi N, Deshmukh S, Bhure A. Comparative study of bupivacaine 0.25% versus ropivacaine 0.5% in transversus abdominis plane block for postoperative analgesia in lower abdominal surgeries: a randomised controlled trial. J Evol Med Dent Sci. 2014;3(17):4569-76. [FreeText]
- Hua F, Sun Q, Zhao T, Chen X, He H. Reporting quality of randomised controlled trial abstracts presented at the SLEEP Annual Meetings: a cross-sectional study. BMJ Open. 2019;9(7):e029270. [PubMed] DOI: 1136/bmjopen-2019-029270
- Hariton E, Locascio JJ. Randomised controlled trials—the gold standard for effectiveness research. BJOG. 2018;125(13):1716. [PubMed] DOI: 1111/1471-0528.15199
- Bhiwal A, Sharma K, Rathore V, Patel C, Chhabra A, Jaitawat S. Comparison of two different doses of dexmedetomidine (0.25 mcg/kg and 0.5 mcg/kg) in prolonging duration of spinal anaesthesia and postoperative analgesia in patients undergoing transurethral resection of prostate: A prospective randomized double blinded. Int J Res Med Sci. 2021;9:1569-76. DOI: 18203/2320-6012.ijrms20211930
- Manne GR, Upadhyay MR, Swadia VN. Effects of low dose dexmedetomidine infusion on haemodynamic stress response, sedation and post-operative analgesia requirement in patients undergoing laparoscopic cholecystectomy. Indian J Anaesth. 2014;58(6):726-31. [PubMed] DOI: 4103/0019-5049.147164
- Ganai S, Lee KF, Merrill A, Lee MH, Bellantonio S, Brennan M, et al. Adverse outcomes of geriatric patients undergoing abdominal surgery who are at high risk for delirium. Arch Surg. 2007;142(11):1072-8. [PubMed] DOI: 1001/archsurg.142.11.1072
- Wang Q, Chen C, Wang L. Efficacy and safety of dexmedetomidine in maintaining hemodynamic stability in pediatric cardiac surgery: a systematic review and meta-analysis. J Pediatr (Rio J). 2022;98(1):15-25. [PubMed] DOI: 1016/j.jped.2021.05.008
- Zhao L, Sun Z, Shen W, Zeng Y. Clinical application of dexmedetomidine combined with dezocine in local anesthesia for endoscopic dacryocystorhinostomy. Ear Nose Throat J. 2022;101(9):NP391-5. [PubMed] DOI: 1177/01455613221108366
- Zhong W-G, Ge X-Y, Zhu H, Liang X, Gong H-X, Zhong M, et al. Dexmedetomidine for antiemesis in gynecologic surgery: a meta-analysis of randomized controlled trials. Int J Clin Exp Med. 2015;8(9):14566-76. [PubMed]
- Imani F, Zaman B, De Negri P. Postoperative pain management: Role of dexmedetomidine as an adjuvant. Anesth Pain Med. 2021;10(6):e112176. [PubMed] DOI: 5812/aapm.112176