Yusra Saleem 1 , Almas Iqbal 2 , Allah Ditta Ashfaq 3
Author affiliations:
Background: In resource-limited, high-volume cancer centers, even small reductions in recovery times can yield meaningful clinical and economic benefits. Therefore, local validation of emerging international findings on dexmedetomidine’s role in enhancing recovery is warranted. This study assessed whether low-dose intraoperative dexmedetomidine in day-case breast cancer surgery could shorten PACU (post-anesthesia care unit), total discharge times and increase early-discharge rates compared with standard anesthetic care.
Methodology: A prospective double blinded randomized controlled trial was conducted at the Department of Anesthesia, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, between September 2023 and July 2025. 82 women aged 18–60 years, ASA (American Society of Anesthesiologists) class I-II, scheduled for same-day elective breast surgery, were randomly allocated into two equal groups. Group A received dexmedetomidine infusion (0.6 µg/kg/h) intra-operatively, while Group B was given placebo. The primary outcome included the frequency of patients achieving early discharge (≤120 minutes).
Results: Early discharge was achieved in all patients receiving dexmedetomidine (100%) and in 37 patients from the control group (90.2%); the difference was not statistically significant (P = 0.116, Fisher’s Exact). The dexmedetomidine group demonstrated a significantly shorter PACU discharge time (35.6 ± 11.0 min vs. 51.3 ± 20.5 min; P < 0.001), chair recovery time (28.1 ± 11.0 min vs. 38.9 ± 17.6 min; P = 0.01), and total discharge time (59.6 ± 18.5 min vs. 80.7 ± 27.4 min; P < 0.001).
Conclusion: Low-dose intraoperative dexmedetomidine enhances postoperative recovery and shortens discharge time without adverse effects, supporting its role as a useful adjunct for ambulatory breast surgery anesthesia.
Keywords: Dexmedetomidine, Day-case surgery, Breast cancer, Post-anesthesia care, Ambulatory anesthesia.
Citation: Saleem Y, Iqbal A, Ashfaq AD. Effect of low dose intravenous dexmedetomidine on post anesthesia discharge after daycase breast cancer surgery: a randomized controlled trial. Anaesth. pain intensive care 2026;30(3):xxxxxxxx. DOI: 10.35975/apic.v30i3.3171
Received: November 04, 2025; Revised: January 11, 2026; Accepted: February 27, 2026
Breast cancer represents the leading cancer diagnosis for women and it makes up more than 10% of annual cancer cases worldwide.1 The disease has emerged as a major public health issue throughout Pakistan. The country maintains one of the highest South Asian age-standardized incidence rates for breast cancer while most patients seek medical help after their disease has reached advanced stages.2 The disease remains undiagnosed for extended periods because of insufficient screening programs and inadequate oncology facilities and insufficient public knowledge about the condition. The increasing number of breast cancer cases puts substantial pressure on surgical departments at tertiary care hospitals which demonstrates the necessity for efficient systems to enhance patient care quality and flow. Modern surgical and anesthetic techniques enable medical professionals to perform breast cancer operations as outpatient procedures. The day-case model allows patients to be discharged immediately after they fulfill recovery standards.3 This approach decreases hospital stay duration while minimizing the chance of acquiring hospital infections and enables patients to recover their physical and mental health more quickly.4 The success of this system heavily relies on patients achieving quick and stable anesthesia recovery with controlled vital signs and effective pain management and reduced postoperative sedation.
Dexmedetomidine, a highly selective α₂-adrenergic receptor agonist, has attracted considerable attention in this regard. It provides sedation, analgesia, and anxiolysis by stimulating α₂ receptors in the locus coeruleus and spinal cord, resulting in decreased sympathetic tone while maintaining stable respiration and inducible sedation.5 This makes it superior to traditional agents such as opioids and benzodiazepines, which are often associated with delayed discharge due to excessive sedation and respiratory depression.
Dexmedetomidine has also been studied under enhanced recovery after surgery (ERAS) pathways and has encouraging outcomes. Several studies and meta-analyses have reported reduced intraoperative opioid use, lower postoperative pain scores, fewer episodes of nausea and vomiting, and an overall improvement in recovery quality.6 It has also been linked to better hemodynamic stability and smoother emergence from anesthesia, helping patients meet discharge criteria earlier in day-case surgeries.7 Research from ambulatory dental and maxillofacial surgery settings has shown higher patient satisfaction and lower anxiety when dexmedetomidine was part of anesthesia management.8
Despite emerging worldwide evidence, corresponding local data on the action of dexmedetomidine to enhance postoperative recuperation among day-case oncological surgery patients are not available from Pakistan. At high-volume tertiary cancer centers where resources remain constrained, quite modest savings on recovery time or discharge can translate to appreciable clinical and cost benefits. Local studies thus need to confirm international results within regional surgical and recuperative settings.
The study aimed to evaluate the effect of low-dose intravenous dexmedetomidine infusion on early postoperative recovery in patients undergoing day-case breast cancer surgery. The objective was to determine whether intraoperative administration of dexmedetomidine could reduce anesthesia care unit (PACU) discharge time, shorten the total recovery period, and increase the proportion of patients suitable for early discharge compared with standard anesthetic management. This study can help provide local evidence to support approval to include the use of dexmedetomidine within standard anesthetic care pathways to optimize postoperative recovery following outpatient oncological surgery.
This randomized controlled clinical trial was conducted at the Department of Anesthesia and Pain Management, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, from September 2023 until July 2025 following approval by the Institutional Review Board (IRB-23-14). The study followed the CONSORT guidelines for randomized clinical trials (Figure 1) and was retrospectively registered on the online trial registry vide no. NCT07180810 (clinicaltrials.gov). Sample size was worked out from the earlier randomized study 9 showing an early discharge incidence of 88% in the dexmedetomidine group and 60% in the control group. Employing a statistical threshold of 80% power and a 95% confidence level, the estimation indicated a requirement of 82 participants, equated to 41 individuals assigned to each study arm.
A total of 82 female patients aged 18 to 60 years, classified as American Society of Anesthesiologists (ASA) physical Status I or II, were enrolled through non-probability consecutive sampling. All patients were scheduled for elective same-day discharge breast cancer surgery, defined as admission, operation, and discharge within the same calendar day. Patients were excluded if they had a body mass index greater than 35 kg/m², refused consent, had a known hypersensitivity to dexmedetomidine, chronic kidney disease of stage three or higher, a previous history of postoperative nausea and vomiting, chronic beta-blocker use, or cardiovascular comorbidities such as bradycardia, heart block, ischemic heart disease, or heart failure.
Randomization was done by using computer-generated randomization program. Patients in Group A received a continuous intravenous infusion of dexmedetomidine at a rate of 0.6 μg/kg/h, prepared in a 50-ml solution, starting from induction of anesthesia until skin closure. Group B received an equal volume of saline 0.9%. Participants and the anesthetist conducting the anaesthesia were blinded to the group to which the patient was assigned. General anaesthesia was standardized within each group. Propofol 2 mg/kg and atracurium 0.5 mg/kg were used to induce the anaesthesia. A supraglottic device was used to secure the airway and sevoflurane was used to maintain the anaesthesia titrated to obtain the bispectral index between 40-60. All the patients received intravenous paracetamol 1 g and ketorolac 30 mg intraoperatively to be used for analgesia. They also received prophylactic ondansetron 4 mg and dexamethasone 4 mg. Morphine 0.1 mg/kg was used as rescue analgesia if the heart rate and blood pressure increased by more than 20% from the baseline.
The main study outcome was early discharge incidence, i.e., admission-to-discharge time ≤120 minute after post-anesthesia care unit (PACU) admission. Chair recovery time, PACU discharge time, and total discharge time were the secondary outcomes. PACU discharge time was calculated from admission time to the PACU until the patient's Modified Aldrete Score reached 9 or more. Modified Aldrete Score is an instrument to evaluate the following five clinical parameters: activity, respiration, circulation, consciousness, and oxygen saturation. All parameters are scored by 0 to 2 and the maximum total therefore is 10.10 A total score of 9 or more is in accordance with readiness to be transferred from the PACU to the step-down recovery area.
Chair recovery time was identified as the time between the time to Modified Aldrete Score of 9 or more (indicating PACU discharge readiness) and the time to Modified Post-Anesthesia Discharge Scoring System score of 9 or more, indicating readiness for discharge home. The system assesses five areas under the MPADSS system: vital sign stability, ambulation, pain, nausea/vomiting, and surgical site bleeding. Each area is scored from 0 to 2, giving a total potential score of 10.11 A score of 9 or more indicates sufficient recovery to be discharged home. PACU discharge time plus chair recovery time provided the total time to discharge.
Demographic data of the patients, perioperative parameters, and postoperative variables were captured on a predesigned structured proforma. Statistical analysis using SPSS version 20 was performed. Continuous data were described on the basis of means and standard deviations and compared between the two groups by Welch's t-test. Categorical data were tested by the chi-square test or Fisher's exact test wherever applicable. Effect size and 95% confidence intervals also were estimated and the significance level to be ≤0.05.
Eighty-two patients were enrolled and randomly divided into two equal groups: Dexmedetomidine (n = 41) and Control (n = 41). All participants completed the study and were analyzed.
The two groups were similar in baseline characteristics. The mean age was 39.27 ± 8.61 years in the dexmedetomidine group and 41.90 ± 9.22 years in the control group (P = 0.19). The mean body mass index (BMI) was 27.78 ± 5.88 kg/m² and 27.21 ± 4.96 kg/m², respectively (P = 0.64). The average duration of surgery was nearly identical between groups (58.17 ± 29.88 minutes vs. 58.22 ± 23.47 minutes; P = 0.99) (Table I).
The primary outcome, early discharge within 120 minutes, was achieved by all patients in the dexmedetomidine group (100%) and by 37 patients in the control group (90.2%) (Figure 2). Although this difference was not statistically significant (Fisher’s Exact P = 0.116), the trend suggested a favorable effect of dexmedetomidine (Table 2).
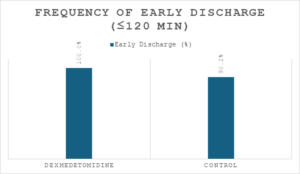
Figure 2: Bar graph showing percentage of early discharges (≤ 120 min) per group.
Among secondary outcomes, all recovery parameters showed a significant reduction in recovery time in the dexmedetomidine group. The mean PACU discharge time was 35.59 ± 11.03 minutes compared with 51.29 ± 20.50 minutes in the control group (P < 0.001). Chair recovery time was also shorter (28.14 ± 11.04 vs 38.87 ± 17.58 minutes; P = 0.01). The total discharge time, representing the overall recovery duration, was markedly reduced (59.61 ± 18.53 vs 80.68 ± 27.35 minutes; P < 0.001) (Table III) (Figure 3).
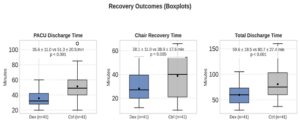
Figure 3: Boxplots comparing recovery times between dexmedetomidine and control groups, showing significantly shorter PACU, chair, and total discharge times with dexmedetomidine (P < 0.05).
No adverse events such as bradycardia, hypotension, or excessive sedation were observed in either group.
This randomized controlled trial compared the effect of intraoperative dexmedetomidine infusion on postoperative recovery and readiness for discharge in patients undergoing breast cancer surgery on a day-case basis. Findings showed that dexmedetomidine decreased PACU discharge time, chair recovery time, and total discharge time significantly, reflecting speedier and more uneventful recovery than with routine anesthetic care.
Although the rate of early discharge was higher with dexmedetomidine (100% vs. 90.2%), this difference did not reach statistical significance (P = 0.116). The 95% confidence interval for the difference (0.7%–18.9%) indicates possible clinical benefit, but the result was limited by the small number of delayed discharges and a ceiling effect in the dexmedetomidine group.
The reduction observed in the recovery time is in line with the established pharmacological profile of dexmedetomidine. Being a highly selective α₂-adrenergic receptor agonist, dexmedetomidine causes cooperative sedation and analgesia with minimal respiratory depression.5 It stabilizes intraoperative hemodynamics and decreases postoperative agitation by lowering sympathetic tone and the release of stress hormones, thus facilitating faster progression through recovery phases. These effects have been well-documented in the literature: Kaye et al. (2020) reported improved postoperative comfort and reduced opioid consumption with dexmedetomidine in enhanced recovery protocols, while Liu et al. (2024) demonstrated accelerated gastrointestinal recovery and reduced hospital stay in general anesthesia patients receiving perioperative dexmedetomidine.12,6
The present results are also congruent with Garip et al. (2022), who noted lower recovery times and greater satisfaction among dental outpatients under dexmedetomidine compared to standard sedative.13 Likewise, Das et al. (2018) reported decreased recovery time and increased discharge preparedness with low-dose dexmedetomidine used as an anesthetic adjunct in ambulatory breast surgery.9 The trend of results across these studies indicates that the sedative-analgesic synergy of the drug allows for easier emergence, decreases postoperative pain and nausea, and increases early ambulation.
In this study, dexmedetomidine did not extend anesthesia duration or extubation delay despite its sedative effects. The maintenance of arousable sedation and preserved respiratory drive may explain why all patients in the intervention group achieved early discharge. Additionally, the lack of significant hemodynamic or sedative complications attests to the safety of low-dose continuous infusion (0.6 µg/kg/h) in an outpatient oncology setting.
From a clinical perspective, the implications of these findings are that 15–20 minute shortening of both PACU and total discharge times per patient can greatly enhance operating-room turnover, increase bed availability, and decrease resource utilization in high-volume oncology centers for day-case anesthesia.
The outcomes also favor the incorporation of dexmedetomidine into Enhanced Recovery After Surgery (ERAS) protocols for breast surgery. Its opioid-sparing and anxiolytic effects complement ERAS concepts of multimodal analgesia and accelerated convalescence. With its hemodynamic stability and good safety profile, dexmedetomidine can be a useful substitute for opioids or benzodiazepines in ambulatory anesthesia regimens.
This study, however, has some limitations. First, it was performed at a single institution with a small sample size, potentially biasing generalizability. Second, exclusion of patients with more extensive cardiovascular disease limits extrapolation to higher-risk patients. Subsequent multicenter trials with greater cohorts should study the cost-effectiveness and patient report outcomes involving dexmedetomidine-facilitated recovery protocols.
Intraoperative low-dose dexmedetomidine infusion significantly shortened PACU discharge, chair recovery, and total discharge times in patients undergoing day-case breast cancer surgery. The drug was well-tolerated, with no adverse hemodynamic or sedative events. Although early-discharge frequency did not reach statistical significance, the overall recovery profile favored dexmedetomidine, supporting its use as a safe and effective adjunct for enhancing postoperative recovery and efficiency in ambulatory oncology anesthesia.
Author affiliations:
- Yusra Saleem, Dept of Anesthesiology & Intensive Care, Shaukat Khanum Memorial Cancer Hospital & Research Center, Lahore, Pakistan; Email: yusrasaleem357@gmail.com
- Almas Iqbal, Dept of Anesthesiology & Intensive Care, Shaukat Khanum Memorial Cancer Hospital & Research Center, Lahore, Pakistan; Emal: almasiqbal@skm.org.pk
- Allah Ditta Ashfaq, Dept of Anesthesiology & Intensive Care, Shaukat Khanum Memorial Cancer Hospital & Research Center, Lahore, Pakistan; Email: adashfaq@skm.org.pk
ABSTRACT
Background: In resource-limited, high-volume cancer centers, even small reductions in recovery times can yield meaningful clinical and economic benefits. Therefore, local validation of emerging international findings on dexmedetomidine’s role in enhancing recovery is warranted. This study assessed whether low-dose intraoperative dexmedetomidine in day-case breast cancer surgery could shorten PACU (post-anesthesia care unit), total discharge times and increase early-discharge rates compared with standard anesthetic care.
Methodology: A prospective double blinded randomized controlled trial was conducted at the Department of Anesthesia, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, between September 2023 and July 2025. 82 women aged 18–60 years, ASA (American Society of Anesthesiologists) class I-II, scheduled for same-day elective breast surgery, were randomly allocated into two equal groups. Group A received dexmedetomidine infusion (0.6 µg/kg/h) intra-operatively, while Group B was given placebo. The primary outcome included the frequency of patients achieving early discharge (≤120 minutes).
Results: Early discharge was achieved in all patients receiving dexmedetomidine (100%) and in 37 patients from the control group (90.2%); the difference was not statistically significant (P = 0.116, Fisher’s Exact). The dexmedetomidine group demonstrated a significantly shorter PACU discharge time (35.6 ± 11.0 min vs. 51.3 ± 20.5 min; P < 0.001), chair recovery time (28.1 ± 11.0 min vs. 38.9 ± 17.6 min; P = 0.01), and total discharge time (59.6 ± 18.5 min vs. 80.7 ± 27.4 min; P < 0.001).
Conclusion: Low-dose intraoperative dexmedetomidine enhances postoperative recovery and shortens discharge time without adverse effects, supporting its role as a useful adjunct for ambulatory breast surgery anesthesia.
Keywords: Dexmedetomidine, Day-case surgery, Breast cancer, Post-anesthesia care, Ambulatory anesthesia.
Citation: Saleem Y, Iqbal A, Ashfaq AD. Effect of low dose intravenous dexmedetomidine on post anesthesia discharge after daycase breast cancer surgery: a randomized controlled trial. Anaesth. pain intensive care 2026;30(3):xxxxxxxx. DOI: 10.35975/apic.v30i3.3171
Received: November 04, 2025; Revised: January 11, 2026; Accepted: February 27, 2026
1. INTRODUCTION
Breast cancer represents the leading cancer diagnosis for women and it makes up more than 10% of annual cancer cases worldwide.1 The disease has emerged as a major public health issue throughout Pakistan. The country maintains one of the highest South Asian age-standardized incidence rates for breast cancer while most patients seek medical help after their disease has reached advanced stages.2 The disease remains undiagnosed for extended periods because of insufficient screening programs and inadequate oncology facilities and insufficient public knowledge about the condition. The increasing number of breast cancer cases puts substantial pressure on surgical departments at tertiary care hospitals which demonstrates the necessity for efficient systems to enhance patient care quality and flow. Modern surgical and anesthetic techniques enable medical professionals to perform breast cancer operations as outpatient procedures. The day-case model allows patients to be discharged immediately after they fulfill recovery standards.3 This approach decreases hospital stay duration while minimizing the chance of acquiring hospital infections and enables patients to recover their physical and mental health more quickly.4 The success of this system heavily relies on patients achieving quick and stable anesthesia recovery with controlled vital signs and effective pain management and reduced postoperative sedation.
Dexmedetomidine, a highly selective α₂-adrenergic receptor agonist, has attracted considerable attention in this regard. It provides sedation, analgesia, and anxiolysis by stimulating α₂ receptors in the locus coeruleus and spinal cord, resulting in decreased sympathetic tone while maintaining stable respiration and inducible sedation.5 This makes it superior to traditional agents such as opioids and benzodiazepines, which are often associated with delayed discharge due to excessive sedation and respiratory depression.
Dexmedetomidine has also been studied under enhanced recovery after surgery (ERAS) pathways and has encouraging outcomes. Several studies and meta-analyses have reported reduced intraoperative opioid use, lower postoperative pain scores, fewer episodes of nausea and vomiting, and an overall improvement in recovery quality.6 It has also been linked to better hemodynamic stability and smoother emergence from anesthesia, helping patients meet discharge criteria earlier in day-case surgeries.7 Research from ambulatory dental and maxillofacial surgery settings has shown higher patient satisfaction and lower anxiety when dexmedetomidine was part of anesthesia management.8
Despite emerging worldwide evidence, corresponding local data on the action of dexmedetomidine to enhance postoperative recuperation among day-case oncological surgery patients are not available from Pakistan. At high-volume tertiary cancer centers where resources remain constrained, quite modest savings on recovery time or discharge can translate to appreciable clinical and cost benefits. Local studies thus need to confirm international results within regional surgical and recuperative settings.
The study aimed to evaluate the effect of low-dose intravenous dexmedetomidine infusion on early postoperative recovery in patients undergoing day-case breast cancer surgery. The objective was to determine whether intraoperative administration of dexmedetomidine could reduce anesthesia care unit (PACU) discharge time, shorten the total recovery period, and increase the proportion of patients suitable for early discharge compared with standard anesthetic management. This study can help provide local evidence to support approval to include the use of dexmedetomidine within standard anesthetic care pathways to optimize postoperative recovery following outpatient oncological surgery.
2. METHODOLOGY
This randomized controlled clinical trial was conducted at the Department of Anesthesia and Pain Management, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, from September 2023 until July 2025 following approval by the Institutional Review Board (IRB-23-14). The study followed the CONSORT guidelines for randomized clinical trials (Figure 1) and was retrospectively registered on the online trial registry vide no. NCT07180810 (clinicaltrials.gov). Sample size was worked out from the earlier randomized study 9 showing an early discharge incidence of 88% in the dexmedetomidine group and 60% in the control group. Employing a statistical threshold of 80% power and a 95% confidence level, the estimation indicated a requirement of 82 participants, equated to 41 individuals assigned to each study arm.
A total of 82 female patients aged 18 to 60 years, classified as American Society of Anesthesiologists (ASA) physical Status I or II, were enrolled through non-probability consecutive sampling. All patients were scheduled for elective same-day discharge breast cancer surgery, defined as admission, operation, and discharge within the same calendar day. Patients were excluded if they had a body mass index greater than 35 kg/m², refused consent, had a known hypersensitivity to dexmedetomidine, chronic kidney disease of stage three or higher, a previous history of postoperative nausea and vomiting, chronic beta-blocker use, or cardiovascular comorbidities such as bradycardia, heart block, ischemic heart disease, or heart failure.
Randomization was done by using computer-generated randomization program. Patients in Group A received a continuous intravenous infusion of dexmedetomidine at a rate of 0.6 μg/kg/h, prepared in a 50-ml solution, starting from induction of anesthesia until skin closure. Group B received an equal volume of saline 0.9%. Participants and the anesthetist conducting the anaesthesia were blinded to the group to which the patient was assigned. General anaesthesia was standardized within each group. Propofol 2 mg/kg and atracurium 0.5 mg/kg were used to induce the anaesthesia. A supraglottic device was used to secure the airway and sevoflurane was used to maintain the anaesthesia titrated to obtain the bispectral index between 40-60. All the patients received intravenous paracetamol 1 g and ketorolac 30 mg intraoperatively to be used for analgesia. They also received prophylactic ondansetron 4 mg and dexamethasone 4 mg. Morphine 0.1 mg/kg was used as rescue analgesia if the heart rate and blood pressure increased by more than 20% from the baseline.
The main study outcome was early discharge incidence, i.e., admission-to-discharge time ≤120 minute after post-anesthesia care unit (PACU) admission. Chair recovery time, PACU discharge time, and total discharge time were the secondary outcomes. PACU discharge time was calculated from admission time to the PACU until the patient's Modified Aldrete Score reached 9 or more. Modified Aldrete Score is an instrument to evaluate the following five clinical parameters: activity, respiration, circulation, consciousness, and oxygen saturation. All parameters are scored by 0 to 2 and the maximum total therefore is 10.10 A total score of 9 or more is in accordance with readiness to be transferred from the PACU to the step-down recovery area.
Chair recovery time was identified as the time between the time to Modified Aldrete Score of 9 or more (indicating PACU discharge readiness) and the time to Modified Post-Anesthesia Discharge Scoring System score of 9 or more, indicating readiness for discharge home. The system assesses five areas under the MPADSS system: vital sign stability, ambulation, pain, nausea/vomiting, and surgical site bleeding. Each area is scored from 0 to 2, giving a total potential score of 10.11 A score of 9 or more indicates sufficient recovery to be discharged home. PACU discharge time plus chair recovery time provided the total time to discharge.
Demographic data of the patients, perioperative parameters, and postoperative variables were captured on a predesigned structured proforma. Statistical analysis using SPSS version 20 was performed. Continuous data were described on the basis of means and standard deviations and compared between the two groups by Welch's t-test. Categorical data were tested by the chi-square test or Fisher's exact test wherever applicable. Effect size and 95% confidence intervals also were estimated and the significance level to be ≤0.05.
3. RESULTS
Eighty-two patients were enrolled and randomly divided into two equal groups: Dexmedetomidine (n = 41) and Control (n = 41). All participants completed the study and were analyzed.
The two groups were similar in baseline characteristics. The mean age was 39.27 ± 8.61 years in the dexmedetomidine group and 41.90 ± 9.22 years in the control group (P = 0.19). The mean body mass index (BMI) was 27.78 ± 5.88 kg/m² and 27.21 ± 4.96 kg/m², respectively (P = 0.64). The average duration of surgery was nearly identical between groups (58.17 ± 29.88 minutes vs. 58.22 ± 23.47 minutes; P = 0.99) (Table I).
| Table 1. Demographic characteristics and duration of surgery of study participants | |||
| Variable | Dexmedetomidine
(n = 41) |
Control
(n = 41) |
P-value |
| Age (years) | 39.27 ± 8.61 | 41.90 ± 9.22 | 0.19 |
| BMI (kg/m²) | 27.78 ± 5.88 | 27.21 ± 4.96 | 0.64 |
| Duration of surgery (min) | 58.17 ± 29.88 | 58.22 ± 23.47 | 0.99 |
The primary outcome, early discharge within 120 minutes, was achieved by all patients in the dexmedetomidine group (100%) and by 37 patients in the control group (90.2%) (Figure 2). Although this difference was not statistically significant (Fisher’s Exact P = 0.116), the trend suggested a favorable effect of dexmedetomidine (Table 2).

Figure 2: Bar graph showing percentage of early discharges (≤ 120 min) per group.
| Table 2. Frequency of early discharge (≤120 minutes) among study groups | ||
| Group | Early Discharge n (%) | p-value (Fisher’s Exact) |
| Dexmedetomidine | 41 (100 %) | 0.116 |
| Control | 37 (90.2 %) | |
Among secondary outcomes, all recovery parameters showed a significant reduction in recovery time in the dexmedetomidine group. The mean PACU discharge time was 35.59 ± 11.03 minutes compared with 51.29 ± 20.50 minutes in the control group (P < 0.001). Chair recovery time was also shorter (28.14 ± 11.04 vs 38.87 ± 17.58 minutes; P = 0.01). The total discharge time, representing the overall recovery duration, was markedly reduced (59.61 ± 18.53 vs 80.68 ± 27.35 minutes; P < 0.001) (Table III) (Figure 3).

Figure 3: Boxplots comparing recovery times between dexmedetomidine and control groups, showing significantly shorter PACU, chair, and total discharge times with dexmedetomidine (P < 0.05).
| Table 3. Comparison of postoperative recovery times between dexmedetomidine and control groups. | ||||
| Outcome | Dexmedetomidine
(mean ± SD) |
Control
(mean ± SD) |
Mean Difference
(95 % CI) |
P-value |
| PACU discharge (min) | 35.59 ± 11.03 | 51.29 ± 20.50 | −15.70 (−23.00 to −8.44) | < 0.001 |
| Chair recovery (min) | 28.14 ± 11.04 | 38.87 ± 17.58 | −10.73 (−18.10 to −3.36) | 0.01 |
| Total discharge (min) | 59.61 ± 18.53 | 80.68 ± 27.35 | −21.07 (−31.36 to −10.78) | < 0.001 |
No adverse events such as bradycardia, hypotension, or excessive sedation were observed in either group.
4. DISCUSSION
This randomized controlled trial compared the effect of intraoperative dexmedetomidine infusion on postoperative recovery and readiness for discharge in patients undergoing breast cancer surgery on a day-case basis. Findings showed that dexmedetomidine decreased PACU discharge time, chair recovery time, and total discharge time significantly, reflecting speedier and more uneventful recovery than with routine anesthetic care.
Although the rate of early discharge was higher with dexmedetomidine (100% vs. 90.2%), this difference did not reach statistical significance (P = 0.116). The 95% confidence interval for the difference (0.7%–18.9%) indicates possible clinical benefit, but the result was limited by the small number of delayed discharges and a ceiling effect in the dexmedetomidine group.
The reduction observed in the recovery time is in line with the established pharmacological profile of dexmedetomidine. Being a highly selective α₂-adrenergic receptor agonist, dexmedetomidine causes cooperative sedation and analgesia with minimal respiratory depression.5 It stabilizes intraoperative hemodynamics and decreases postoperative agitation by lowering sympathetic tone and the release of stress hormones, thus facilitating faster progression through recovery phases. These effects have been well-documented in the literature: Kaye et al. (2020) reported improved postoperative comfort and reduced opioid consumption with dexmedetomidine in enhanced recovery protocols, while Liu et al. (2024) demonstrated accelerated gastrointestinal recovery and reduced hospital stay in general anesthesia patients receiving perioperative dexmedetomidine.12,6
The present results are also congruent with Garip et al. (2022), who noted lower recovery times and greater satisfaction among dental outpatients under dexmedetomidine compared to standard sedative.13 Likewise, Das et al. (2018) reported decreased recovery time and increased discharge preparedness with low-dose dexmedetomidine used as an anesthetic adjunct in ambulatory breast surgery.9 The trend of results across these studies indicates that the sedative-analgesic synergy of the drug allows for easier emergence, decreases postoperative pain and nausea, and increases early ambulation.
In this study, dexmedetomidine did not extend anesthesia duration or extubation delay despite its sedative effects. The maintenance of arousable sedation and preserved respiratory drive may explain why all patients in the intervention group achieved early discharge. Additionally, the lack of significant hemodynamic or sedative complications attests to the safety of low-dose continuous infusion (0.6 µg/kg/h) in an outpatient oncology setting.
From a clinical perspective, the implications of these findings are that 15–20 minute shortening of both PACU and total discharge times per patient can greatly enhance operating-room turnover, increase bed availability, and decrease resource utilization in high-volume oncology centers for day-case anesthesia.
The outcomes also favor the incorporation of dexmedetomidine into Enhanced Recovery After Surgery (ERAS) protocols for breast surgery. Its opioid-sparing and anxiolytic effects complement ERAS concepts of multimodal analgesia and accelerated convalescence. With its hemodynamic stability and good safety profile, dexmedetomidine can be a useful substitute for opioids or benzodiazepines in ambulatory anesthesia regimens.
This study, however, has some limitations. First, it was performed at a single institution with a small sample size, potentially biasing generalizability. Second, exclusion of patients with more extensive cardiovascular disease limits extrapolation to higher-risk patients. Subsequent multicenter trials with greater cohorts should study the cost-effectiveness and patient report outcomes involving dexmedetomidine-facilitated recovery protocols.
5. CONCLUSION
Intraoperative low-dose dexmedetomidine infusion significantly shortened PACU discharge, chair recovery, and total discharge times in patients undergoing day-case breast cancer surgery. The drug was well-tolerated, with no adverse hemodynamic or sedative events. Although early-discharge frequency did not reach statistical significance, the overall recovery profile favored dexmedetomidine, supporting its use as a safe and effective adjunct for enhancing postoperative recovery and efficiency in ambulatory oncology anesthesia.
6. REFERENCES:
- Menon G, Alkabban FM, Ferguson T. Breast cancer. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482286/
- Faisal AB, Shahid F, Khalid L, Rahman MF. Advancing Immediate Breast Reconstruction Surgery in Pakistan: Bridging Literature Gaps and Meeting Patient Needs. Arch Plast Surg. 2025;52(2):116-8. [PubMed] DOI: 1055/a-2312-8945
- Shaker H, Leena NA, Mayers V, Koussa F, Deshpande A. Day-case approach to immediate breast reconstruction: pushing the boundaries of ambulatory breast surgery in the post-COVID-19 era. Ann R Coll Surg Engl. 2021;103(6):426-31. [PubMed] DOI: 1308/rcsann.2020.7152
- Kumar R, Choudhury SR, Yadav PS, Kundal R, Gupta A, Hayaran N, et al. An analysis of safety and efficacy of day-care surgery in children in a tertiary care Hospital in India. J Indian Assoc Pediatr Surg. 2021;26(3):148-52. [PubMed] DOI: 4103/jiaps.JIAPS_52_20
- Lee S. Dexmedetomidine: present and future directions. Korean J Anesthesiol. 2019;72(4):323-30. [PubMed] DOI: 4097/kja.19259
- Liu Y, Liang H, Sun Y, Liu W, Ye L, He W, et al. Effect of perioperative dexmedetomidine on recovery of postoperative gastrointestinal function in patients with general anesthesia: a systematic review and meta-analysis. BMC Anesthesiol. 2024;24(1):479. [PubMed] DOI: 1186/s12871-024-02868-0
- Ye Q, Wang F, Xu H, Wu L, Gao X. Effects of dexmedetomidine on intraoperative hemodynamics, recovery profile and postoperative pain in patients undergoing laparoscopic cholecystectomy: a randomized controlled trial. BMC Anesthesiol. 2021;21(1):63. [PubMed] DOI: 1186/s12871-021-01283-z
- Mishra N, Birmiwal KG, Pani N, Raut S, Sharma G, Rath KC. Sedation in oral and maxillofacial day care surgery: a comparative study between intravenous dexmedetomidine and midazolam. Natl J Maxillofac Surg. 2016;7(2):178-85. [PubMed] DOI: 4103/njms.NJMS_78_16
- Das R, Das RK, Sahoo S, Nanda S. Role of dexmedetomidine as an anaesthetic adjuvant in breast cancer surgery as a day-care procedure: A randomised controlled study. Indian J Anaesth. 2018;62(3):182-7. [PubMed] DOI: 4103/ija.IJA_752_17
- Dahake JS, Verma N. Comparative analysis of the modified Aldrete score and fast-track criteria for post-general anaesthesia recovery: a narrative review. Cureus. 2024;16(7):e64439. [PubMed] DOI: 7759/cureus.64439
- El Aoufy K, Forciniti C, Longobucco Y, Lucchini A, Mangli I, Magi CE, et al. A comparison among score systems for discharging patients from recovery rooms: A narrative review. Nurs Rep. 2024;14(4):2777-94. [PubMed] DOI: 3390/nursrep14040205
- Kaye AD, Chernobylsky DJ, Thakur P, Siddaiah H, Kaye RJ, Eng LK, et al. Dexmedetomidine in enhanced recovery after surgery (ERAS) protocols for postoperative pain. Curr Pain Headache Rep. 2020;24(5):21. [PubMed] DOI: 1007/s11916-020-00853-z
- Garip L, Verbist J, Stragier H, Meyns J, Mesotten D, Vundelinckx J. A comparative study of patient satisfaction about anesthesia with dexmedetomidine for ambulatory dental surgery. BMC Res Notes. 2022;15(1):376. [PubMed] DOI: 1186/s13104-022-06246-2