Ahmed Elsherbeny 1 . Ibrahim AbdelAal 2 . Ahmad Abuzaid 3 . Ahmed Abdalwahab 4
Authors affiliations:
Background & objective: Despite an international effort lead by the World Health Organization (WHO) many cardiac centers still continue the practice of empirical blood product transfusion. Current guidelines and published evidence strongly support the use of transfusion algorithms that are guided by point-of-care coagulation testing. The aim of this study is to determine the proportion of patients with normal coagulation parameters who still received empirical blood products during cardiac surgery. Secondary end point is the occurrence of transfusion-related adverse events.
Methodology: This is a retrospective observational study of 100 pediatric patients scheduled for cardiac surgery. As part of the rotational thromboelastometry (ROTEM) introduction to the operating theatre, two ROTEM samples were collected, before and after blood products. The empirical blood products practice continued as usual and did not change based on those ROTEM samples.
Results: Among patients who received empirical blood products, 16% had normal fibrinogen levels (did not need Cryoprecipitates), 24% had normal clotting factors (did not need fresh frozen plasma) and 60% had normal platelets assessment. No significant adverse events attributable to blood products were observed.
Conclusion A proportion of pediatric patients had normal coagulation parameters prior to the administration of empirical blood products. The study highlights the value of viscoelastic point-of-care testing to avoid unnecessary transfusion during cardiac surgery. It underscores the urgent need to transition institutional practice towards algorithm-based approach and appropriate use of blood components.
Abbreviations: FFP: Fresh frozen plasma. ROTEM: rotational thromboelastometry, WHO: World Health Organization, TACO: Transfusion-associated circulatory overload, TRALI: Transfusion-related lung injury
Keywords: Blood coagulation; Cardiac surgery; Cardiopulmonary bypass; Fresh frozen plasma; Point of care; : Rotational thromboelastometry.
Citation: Elsherbeny A, AbdelAal I, Abuzaid A, Abdalwahab A. A retrospective study of empirical transfusion of blood products during cardiac surgery measured by thromboelastometry; Is it time to change? Anaesth. pain intensive care 2026;30(3):314-318. DOI: 10.35975/apic.v30i3.3168
Received: December 23, 2025; Revised: March 06, 2026; Accepted: March 11, 2026
“The first step in successful institutional change is establishing a sense of urgency, this awareness of a crisis is important to motivate individuals”, wrote Harvard professor John Kotter in his 1995 article “ Leading change, why transformation efforts fail”.1 Despite the international effort led by the World Health Organization (WHO) to spread the culture of patient blood management,2 many cardiac centers continue long-standing practice of empirical transfusion of blood products.
Although evidence supports the use of transfusion algorithms guided by point-of-care coagulation testing,3-5 and despite numerous publications describing the transfusion-related adverse outcome in cardiac surgery,6-10 the actual change of institutional culture needs both time and effort.
As our centre prepared to introduce algorithm-based transfusion practice, we collected coagulation data using point-of-care testing (Rotational thromboelastometry ROTEM®, Tem International GmbH) while the existing empirical practice were still going. The aim of this study is to determine the proportion of patients with normal coagulation parameters who still received empirical blood products during cardiac surgery.
This is a retrospective observational study of patients scheduled for cardiac surgery. The study was approved by the institutional review board (No. 1690), which granted a waiver of written inform consent due to the strict observational design of this study. Data from 100 pediatric patients who underwent cardiac surgery over six months period (corresponding to the initial availabilty of ROTEM in the operating theatre) were analyzed. Inclusion criteria were any pediatric patient undergoing cardiac surgery who received empirical blood products after cardiopulmonary bypass weaning. Exclusion criteria were patients with pre-existing coagulopathy including renal and hepatic dysfunction, patients on anticoagulants or antiplatelets, off-pump cardiac surgery and patients receiving blood products before starting the cardiopulmonary bypass.
ROTEM measurements
With the initial introduction of ROTEM in theatre, the usual practice of empirical blood products during cardiac surgery continued unchanged. As part of the initiation phase, two ROTEM samples were collected:
According to the existing practice, all patients had a routine order of 10 ml/kg of FFP, 10 ml/kg of Platelets and 5 ml/kg of Cryoprecipitates on admission to operating room. Blood products are then transfused after weaning.
A second set of blood products may be ordered and transfused if there is diffuse bleeding based on the surgical team assessment. This empirical practice did not change based on the ROTEM samples, throughout the study period.
Adverse events attributable to blood product transfusion were recorded, this included: Transfusion reaction, Transfusion-associated circulatory overload (TACO), Transfusion-related lung injury (TRALI) and venous thrombosis.
The primary endpoint is the percentage of patients with normal coagulation even before the empirical blood products transfusion, based on the first ROTEM sample. Secondary endpoint was the incidence of adverse events related to blood product transfusion.
Statistical analysis
Data were coded and entered using the statistical package for the Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA). Data was summarized using median and interquartile range. Data was double checked for normality using normality plots and Shapiro Wilk test and proved to be deviated from normal distribution. For comparison of paired measurements (before and after) within each patient, the non-parametric Wilcoxon signed rank test was used. P-values less than 0.05 were considered as statistically significant. The sample size was calculated based on previously published data by Ichikawa et al. (2018) they found an incidence of normal ROTEM parameters for FFP after CPB of 43%, accordingly a calculated sample of 95 patients is needed with a confidence interval of 95% and a precision of ± 10%.11
Data collected from 100 pediatric cardiac surgery patients were analysed. Demographic and perioperative data are presented in (Table 1).
The comparison between ROTEM values before and after empiric products is presented in (Table 2), a clinically significant improvement in all parameters can be observed, except for INTEM CT/HEPTEM CT ratio, which remained unchanged before and after products. The percentage of patients with normal coagulation in pre-transfusion ROTEM samples are shown in (Figure 1). 16% had normal fibrinogen levels (did not need Cryoprecipitates), 24% had normal clotting factors (did not need fresh frozen plasma) and 60% had normal platelets assessment. No significant adverse events attributable to blood product transfusion were reported.
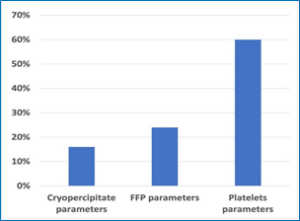
Figure 1: Normal ROTEM before blood products
In this retrospective study, the authors found a high percentage of patients with normal ROTEM parameters prior to receiving empirical blood product transfusion. As expected, there was a numerical improvement in most of the coagulation parameters observed after empirical treatment. The prevalence of normal pre-transfusion ROTEM parameters varied according to the specific coagulation factor observed.
Implementing evidence-based transfusion guidelines comes in the heart of patient blood management (PBM) polices.2 One of the pillars PBM is to minimize blood loss and optimize coagulation. The world Health Organization (WHO) has issued a policy brief in 2021 “The urgent need to implement patient blood management”.2 The brief describes an urgent need to adopt of PBM as a standard of care. Intraoperative point-of-care coagulation testing was described as an important component in a PBM programmes and can reduce unnecessary transfusions and enhance recovery after cardiac surgery.12-15 One of the main barriers against implementation of PBM policies is the established culture within an institution.12 Our center, and probably many other centers, use of empirical blood product transfusion in cardiac surgery continued for decades. A switch to algorithm-based transfusion practice needs to bring different stakeholders on board. The results of the current study will be used to highlight the urgent need for change within the institution.
Among the study group, we found the main deficient clotting parameter was Fibrinogen. Depletion of fibrinogen in pediatric cardiac surgery is well known.16-18 In a retrospective study, Lindhart et al. (2022) recorded thromboelsatography (TEG) parameters in pediatric patients at the end of CPB and found that 84% of study population have low TEG parameters for fibrinogen, this matches our findings using ROTEM.17 Gautam et al [2017] demonstrated a drop of fibrinogen plasma level in pediatric population from (241± 68 mg/dl) before bypass to (176± 51 mg/dl) after bypass, demonstrating the magnitude of the issue, and the value of point-of-care testing to monitor such deficiency.19 The results did not reveal adverse events related to blood product transfusion, however, a group of patients (15%) received PRCs due to the hemodilution after blood products. This was reported by previous metanalysis, comparing FFP group vs non-FFP group, with a tendency for more blood transfusion in the FFP group (odds ratio of 2.57).20
This study has several limitations. First, the small sample size, as the study was mainly designed to initiate change of culture in the institution. Second, the retrospective design. Additionally, ROTEM lacks the ability of platelet function mapping, which would have demonstrated more need for platelet transfusion in our cohort.
A proportion of pediatric patients had normal coagulation parameters prior to the administration of empirical blood products. The study highlights the value of viscoelastic point-of-care testing to avoid unnecessary transfusion during cardiac surgery. It underscores the urgent need to transition institutional practice towards algorithm-based approach and appropriate use of blood components.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
All authors took part in the concept, conduct of the study, data search and manuscript editing
Authors affiliations:
- Ahmed Elsherbeny. MBBCh. Cardiac Anesthesia Department, Prince Sultan Cardiac Centre, Riyadh, Saudi Arabia; Email: aalsherbeny@pscc.med.sa; https://orcid.org/0009-0002-4721-6901
- Ahmad Abuzaid. MBBCh. Cardiac Anesthesia Department, Prince Sultan Cardiac Centre, Riyadh, Saudi Arabia; Email: AAbuZaid@pscc.med.sa; https://orcid.org/0009-0008-4616-9123
- Ahmed Abdalwahab. MBBCh. Cardiac Anesthesia Department, Prince Sultan Cardiac Centre, Riyadh, Saudi Arabia; Email: dr.ahmedabdalwahab@gmail.com;
ABSTRACT
Background & objective: Despite an international effort lead by the World Health Organization (WHO) many cardiac centers still continue the practice of empirical blood product transfusion. Current guidelines and published evidence strongly support the use of transfusion algorithms that are guided by point-of-care coagulation testing. The aim of this study is to determine the proportion of patients with normal coagulation parameters who still received empirical blood products during cardiac surgery. Secondary end point is the occurrence of transfusion-related adverse events.
Methodology: This is a retrospective observational study of 100 pediatric patients scheduled for cardiac surgery. As part of the rotational thromboelastometry (ROTEM) introduction to the operating theatre, two ROTEM samples were collected, before and after blood products. The empirical blood products practice continued as usual and did not change based on those ROTEM samples.
Results: Among patients who received empirical blood products, 16% had normal fibrinogen levels (did not need Cryoprecipitates), 24% had normal clotting factors (did not need fresh frozen plasma) and 60% had normal platelets assessment. No significant adverse events attributable to blood products were observed.
Conclusion A proportion of pediatric patients had normal coagulation parameters prior to the administration of empirical blood products. The study highlights the value of viscoelastic point-of-care testing to avoid unnecessary transfusion during cardiac surgery. It underscores the urgent need to transition institutional practice towards algorithm-based approach and appropriate use of blood components.
Abbreviations: FFP: Fresh frozen plasma. ROTEM: rotational thromboelastometry, WHO: World Health Organization, TACO: Transfusion-associated circulatory overload, TRALI: Transfusion-related lung injury
Keywords: Blood coagulation; Cardiac surgery; Cardiopulmonary bypass; Fresh frozen plasma; Point of care; : Rotational thromboelastometry.
Citation: Elsherbeny A, AbdelAal I, Abuzaid A, Abdalwahab A. A retrospective study of empirical transfusion of blood products during cardiac surgery measured by thromboelastometry; Is it time to change? Anaesth. pain intensive care 2026;30(3):314-318. DOI: 10.35975/apic.v30i3.3168
Received: December 23, 2025; Revised: March 06, 2026; Accepted: March 11, 2026
1. INTRODUCTION
“The first step in successful institutional change is establishing a sense of urgency, this awareness of a crisis is important to motivate individuals”, wrote Harvard professor John Kotter in his 1995 article “ Leading change, why transformation efforts fail”.1 Despite the international effort led by the World Health Organization (WHO) to spread the culture of patient blood management,2 many cardiac centers continue long-standing practice of empirical transfusion of blood products.
Although evidence supports the use of transfusion algorithms guided by point-of-care coagulation testing,3-5 and despite numerous publications describing the transfusion-related adverse outcome in cardiac surgery,6-10 the actual change of institutional culture needs both time and effort.
As our centre prepared to introduce algorithm-based transfusion practice, we collected coagulation data using point-of-care testing (Rotational thromboelastometry ROTEM®, Tem International GmbH) while the existing empirical practice were still going. The aim of this study is to determine the proportion of patients with normal coagulation parameters who still received empirical blood products during cardiac surgery.
2. METHODOLOGY
This is a retrospective observational study of patients scheduled for cardiac surgery. The study was approved by the institutional review board (No. 1690), which granted a waiver of written inform consent due to the strict observational design of this study. Data from 100 pediatric patients who underwent cardiac surgery over six months period (corresponding to the initial availabilty of ROTEM in the operating theatre) were analyzed. Inclusion criteria were any pediatric patient undergoing cardiac surgery who received empirical blood products after cardiopulmonary bypass weaning. Exclusion criteria were patients with pre-existing coagulopathy including renal and hepatic dysfunction, patients on anticoagulants or antiplatelets, off-pump cardiac surgery and patients receiving blood products before starting the cardiopulmonary bypass.
ROTEM measurements
With the initial introduction of ROTEM in theatre, the usual practice of empirical blood products during cardiac surgery continued unchanged. As part of the initiation phase, two ROTEM samples were collected:
- Immediately after protamine administration (before blood products),and
- After administration of empirical blood products.
According to the existing practice, all patients had a routine order of 10 ml/kg of FFP, 10 ml/kg of Platelets and 5 ml/kg of Cryoprecipitates on admission to operating room. Blood products are then transfused after weaning.
A second set of blood products may be ordered and transfused if there is diffuse bleeding based on the surgical team assessment. This empirical practice did not change based on the ROTEM samples, throughout the study period.
Adverse events attributable to blood product transfusion were recorded, this included: Transfusion reaction, Transfusion-associated circulatory overload (TACO), Transfusion-related lung injury (TRALI) and venous thrombosis.
The primary endpoint is the percentage of patients with normal coagulation even before the empirical blood products transfusion, based on the first ROTEM sample. Secondary endpoint was the incidence of adverse events related to blood product transfusion.
Statistical analysis
Data were coded and entered using the statistical package for the Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA). Data was summarized using median and interquartile range. Data was double checked for normality using normality plots and Shapiro Wilk test and proved to be deviated from normal distribution. For comparison of paired measurements (before and after) within each patient, the non-parametric Wilcoxon signed rank test was used. P-values less than 0.05 were considered as statistically significant. The sample size was calculated based on previously published data by Ichikawa et al. (2018) they found an incidence of normal ROTEM parameters for FFP after CPB of 43%, accordingly a calculated sample of 95 patients is needed with a confidence interval of 95% and a precision of ± 10%.11
3. RESULTS
Data collected from 100 pediatric cardiac surgery patients were analysed. Demographic and perioperative data are presented in (Table 1).
| Table 1: Demographic and perioperative data. | |
| Parameter | Result |
| Age | 5 (3,10) months |
| Weight (Kg) | 10.3 ± 4 |
| Bypass time (minutes) | 187 ± 93 |
| Clamp time (minutes) | 83 ± 57 |
| Heparin/Protamine ratio | 0.88 ± 0.12 |
| Volume of FFP (ml) | 176 ± 226 |
| Volume of Platelets (ml) | 76 ± 65 |
| Volume of Cryoprecipitate (ml) | 23.3 ± 20.5 |
| Transfusion reaction | 0 |
| TACO | 0 |
| TRALI | 0 |
| Thrombosis (CVA, arterial or venous thrombosis) | 2 (2%) |
| PRCs due to associated hemodilution | 15 (15%) |
| Abbreviations: FFP: Fresh frozen plasma. TACO: TRALI: CVA: PRCs: Data presented as median (Q1,Q3) , mean ± standard deviation or number (percentage). n = 100 | |
The comparison between ROTEM values before and after empiric products is presented in (Table 2), a clinically significant improvement in all parameters can be observed, except for INTEM CT/HEPTEM CT ratio, which remained unchanged before and after products. The percentage of patients with normal coagulation in pre-transfusion ROTEM samples are shown in (Figure 1). 16% had normal fibrinogen levels (did not need Cryoprecipitates), 24% had normal clotting factors (did not need fresh frozen plasma) and 60% had normal platelets assessment. No significant adverse events attributable to blood product transfusion were reported.
| Table 2: ROTEM parameters before and after blood products transfusion (n=100) | |||||||
| Before | After | P value | |||||
| Median | 1st quartile | 3rd quartile | Median | 1st quartile | 3rd quartile | ||
| CT INTEM | 697.50 | 496.00 | 1069.00 | 314.00 | 253.00 | 353.00 | <0.001 |
| CT HEPTEM | 616.00 | 404.00 | 1004.00 | 315.00 | 247.00 | 352.00 | <0.001 |
| CT INT/ CT HEP | 1.04 | 1.00 | 1.08 | 1.00 | 0.92 | 1.02 | 0.078 |
| A5 FIBTEM | 5.00 | 3.00 | 10.00 | 13.00 | 10.00 | 15.00 | <0.001 |
| A5 EXTEM | 27.00 | 23.00 | 40.00 | 41.00 | 37.00 | 45.00 | <0.001 |
| CT EXTEM | 99.00 | 94.00 | 117.00 | 76.00 | 70.00 | 90.00 | <0.001 |

Figure 1: Normal ROTEM before blood products
4. DISCUSSION
In this retrospective study, the authors found a high percentage of patients with normal ROTEM parameters prior to receiving empirical blood product transfusion. As expected, there was a numerical improvement in most of the coagulation parameters observed after empirical treatment. The prevalence of normal pre-transfusion ROTEM parameters varied according to the specific coagulation factor observed.
Implementing evidence-based transfusion guidelines comes in the heart of patient blood management (PBM) polices.2 One of the pillars PBM is to minimize blood loss and optimize coagulation. The world Health Organization (WHO) has issued a policy brief in 2021 “The urgent need to implement patient blood management”.2 The brief describes an urgent need to adopt of PBM as a standard of care. Intraoperative point-of-care coagulation testing was described as an important component in a PBM programmes and can reduce unnecessary transfusions and enhance recovery after cardiac surgery.12-15 One of the main barriers against implementation of PBM policies is the established culture within an institution.12 Our center, and probably many other centers, use of empirical blood product transfusion in cardiac surgery continued for decades. A switch to algorithm-based transfusion practice needs to bring different stakeholders on board. The results of the current study will be used to highlight the urgent need for change within the institution.
Among the study group, we found the main deficient clotting parameter was Fibrinogen. Depletion of fibrinogen in pediatric cardiac surgery is well known.16-18 In a retrospective study, Lindhart et al. (2022) recorded thromboelsatography (TEG) parameters in pediatric patients at the end of CPB and found that 84% of study population have low TEG parameters for fibrinogen, this matches our findings using ROTEM.17 Gautam et al [2017] demonstrated a drop of fibrinogen plasma level in pediatric population from (241± 68 mg/dl) before bypass to (176± 51 mg/dl) after bypass, demonstrating the magnitude of the issue, and the value of point-of-care testing to monitor such deficiency.19 The results did not reveal adverse events related to blood product transfusion, however, a group of patients (15%) received PRCs due to the hemodilution after blood products. This was reported by previous metanalysis, comparing FFP group vs non-FFP group, with a tendency for more blood transfusion in the FFP group (odds ratio of 2.57).20
5. LIMITATIONS
This study has several limitations. First, the small sample size, as the study was mainly designed to initiate change of culture in the institution. Second, the retrospective design. Additionally, ROTEM lacks the ability of platelet function mapping, which would have demonstrated more need for platelet transfusion in our cohort.
6. CONCLUSION
A proportion of pediatric patients had normal coagulation parameters prior to the administration of empirical blood products. The study highlights the value of viscoelastic point-of-care testing to avoid unnecessary transfusion during cardiac surgery. It underscores the urgent need to transition institutional practice towards algorithm-based approach and appropriate use of blood components.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
All authors took part in the concept, conduct of the study, data search and manuscript editing
11. REFERENCES
- Kotter JP. Leading change: why transformation efforts fail. Harvard business review 1995; May-June. Available from https://hbr.org/1995/05/leading-change-why-transformation-efforts-fail-2
- World Health Organization. 2021. The urgent need to implement patient blood management: policy brief. World Health Organization. Available from https://iris.who.int/handle/10665/346655.
- Kuiper GJAJM, van Egmond LT, Henskens YMC, Roekaerts PM, Maessen JG, Ten Cate H et al. Shifts of transfusion demand in cardiac surgery after implementation of rotational thromboelastometry-guided transfusion protocols: analysis of the HEROES-CS (HEmostasis Registry of patiEntS in Cardiac Surgery) observational, prospective open cohort database. Journal of Cardiothoracic and Vascular Anesthesia 2019; 33 (2) 307–317. DOI: 1053/j.jvca.2018.08.203
- Görlinger K, Pérez-Ferrer A, Dirkmann D, Saner F, Maegele M, Calatayud ÁAP et al. The role of evidence-based algorithms for rotational thromboelastometry-guided bleeding management. Korean Journal of anaesthesiology 2019; Aug;72(4):297-322. PMCID: PMC6676023 DOI: 4097/kja.19169
- Nakayama Y, Nakajima Y, Tanaka KA, Sessler DI, Maeda S, Iida J et al. Thromboelastometry-guided intraoperative haemostatic management reduces bleeding and red cell transfusion after paediatric cardiac surgery. Br J Anaesth 2015; 114:91–102. DOI: 1093/bja/aeu339
- Gulack BC, Kirkwood KA, Shi W, Smith PK, Alexander JH, Burks SG et al. Secondary surgical-site infection after coronary artery bypass grafting: A multi-institutional prospective cohort study. J Thorac Cardiovasc Surg 2018; 155:1555-62.
- Shaw RE, Johnson CK, Ferrari G, Brizzio ME, Sayles K, Rioux N et al. Blood transfusion in cardiac surgery does increase the risk of 5-year mortality: results from a contemporary series of 1714 propensity-matched patients. Transfusion 2014; 54:1106-13. DOI: 1111/trf.12364
- Horvath KA, Acker MA, Chang H, Bagiella E, Smith PK, Iribarne A et al. Blood transfusion and infection after cardiac surgery. Ann Thorac Surg 2013; 95:2194-201. PMCID: PMC3992887 DOI: 1016/j.athoracsur.2012.11.078
- Mikkola R, Gunn J, Heikkinen J, Wistbacka JO, Teittinen K, Kuttila K et al. Use of blood products and risk of stroke after coronary artery bypass surgery. Blood Transfus 2012; 10(4):490-501. PMCID: PMC3496224 DOI: 2450/2012.0119-11
- Stone GW, Clayton TC, Mehran R, Dangas G, Parise H, Fahy M et al. Impact of major bleeding and blood transfusions after cardiac surgery: analysis from the Acute Catheterization and Urgent Intervention Triage strategy (ACUITY) trial. American heart journal 2012; 163:522-9. DOI: 1016/j.ahj.2011.11.016
- Ichikawa J, Marubuchi T, Nishiyama K, Kodaka M, Görlinger K, Ozaki M et al. Introduction of thromboelastometry-guided administration of fresh-frozen plasma is associated with decreased allogeneic blood transfusions and post-operative blood loss in cardiopulmonary-bypass surgery. Blood Transfus. 2018; 16(3):244-252. PMCID: PMC5919836 DOI: 2450/2017.0265-16
- World Health Organization. Educational modules on clinical use of blood. World Health Organization. Available from https://iris.who.int/handle/10665/350246.
- Grant MC, Crisafi C, Alvarez A, Arora RC, Brindle ME, Chatterjee S et al. Perioperative Care in Cardiac Surgery: A Joint Consensus Statement by the Enhanced Recovery After Surgery (ERAS) Cardiac Society, ERAS International Society, and The Society of Thoracic Surgeons (STS). Ann Thorac Surg 2024; 117(4):669-689. DOI: 1016/j.athoracsur.2023.12.006
- Boer C, Meesters MI, Milojevic M, Benedetto U, Bolliger D, von Heymann C et al. Task Force on Patient Blood Management for Adult Cardiac Surgery of the European Association for Cardio-Thoracic Surgery (EACTS) and the European Association of Cardiothoracic Anaesthesiology (EACTA). J Cardiothorac Vasc Anesth 2018; 32(1):88-120. DOI: 1053/j.jvca.2017.06.026
- Tibi P, McClure RS, Huang J, Baker RA, Fitzgerald D, Mazer CD et al. STS/SCA/AmSECT/SABM Update to the Clinical Practice Guidelines on Patient Blood Management. Ann Thorac Surg 2021; 112(3):981-1004. DOI: 1016/j.athoracsur.2021.03.033
- Dennhardt N, Sümpelmann R, Horke A, Keil O, Nickel K, Heiderich S et al. Prevention of postoperative bleeding after complex pediatric cardiac surgery by early administration of fibrinogen, prothrombin complex and platelets: a prospective observational study. BMC Anesthesiology 2020; 20:302. PMCID: PMC7747387 DOI: 1186/s12871-020-01217-1
- Lindhardt RB, Kronborg JR, Wanscher M, Andersen LW, Gjedsted J, Ravn HB. Evaluation of Thromboelastography 6s prognostication of fibrinogen supplementation in pediatric cardiac surgery. Acta Anaesthesiol Scand 2022; 66(10):1166-1173. PMCID: PMC9826011 DOI: 1111/aas.14144
- Gautam NK, Cai C, Pawelek O, Rafique MB, Cattano D, Pivalizza EG. Performance of functional fibrinogen thromboelastography in children undergoing congenital heart surgery. Paediatr Anaesth 2017; 27(2):181-189. DOI: 1111/pan.13048
- Gautam NK, Cai C, Pawelek O, Rafique MB, Cattano D, Pivalizza EG. Performance of functional fibrinogen thromboelastography in children undergoing congenital heart surgery. Paediatr Anaesth 2017; 27(2):181-189. DOI: 1111/pan.13048
- Desborough MJR, Sandu R, Brunskill SJ, Doree C, Trivella M, Montedori A et al. Fresh frozen plasma for cardiovascular surgery. Cochrane Database of Systematic Reviews 2015; Issue 7. Art. No.: CD007614.