Mohammed H. Mohammed 1 , Shrouk A. Hassan 2 , Ali A. Hassan 3
Authors affiliations:
Background: Lung cancer is a leading cause of cancer mortality, linked to anti-oxidant and oxidative stress and epithelial damage. Biomarkers like superoxide dismutase (SOD), glutathione peroxidase (GPx), malondialdehyde (MDA), and cytokeratin fragment 21-1 (CYFRA 21-1) may indicate tumor progression.
Objective: To assess serum levels of oxidative stress markers (SOD, GPx, MDA) and CYFRA 21-1 in lung cancer patients versus controls used to monitoring and prognosis tool for the oxidative stress status.
Methodology: This case-control study was carried out from November 2024 to April 2025 at Nassiriya Teaching Hospital in Thi-Qar, Iraq. Sixty patients with lung cancer and 60 age-matched controls were included in the study. Venous blood samples were obtained, and serum was separated and stored to determine the concentrations of SOD, Gpx, MDA and CYFRA 21-1 using enzyme-linked immunosorbent assay (ELISA).
Results: There were no significant differences in age or BMI between the two groups. Lung cancer patients had significantly elevated levels of MDA (193.2 ± 18.6 vs. 158.6 ± 21.09 nmol/mL), SOD (90.15 ± 34.9 vs. 42.07 ± 13.78 ng/mL), GPx (65.4 ± 29.4 vs. 9.98 ± 5.6 µU/mL), and CYFRA 21-1 (15.1 ± 6.5 vs. 5.9 ± 2.7 ng/mL) compared to controls (P < 0.001 for all). Oxidative stress markers and CYFRA 21-1 levels did not differ significantly among age subgroups in patients with lung cancer.
Conclusions: This study highlights a significant elevation in oxidative stress markers and CYFRA 21-1 in patients with lung cancer, supporting their potential role as diagnostic and prognostic biomarkers. Monitoring these biomarkers may enhance early detection and management of lung cancer in clinical settings.
Keywords: Lung cancer, Oxidative stress, Cytokeratin fragment 21-1
Citation: Mohammed MH, Hassan SA, Hassan AA. Evaluation of oxidative stress and diagnostic value of cytokeratin fragment 21-1 in lung cancer patients. Anaesth. pain intensive care 2026;30(3):301-307. DOI: 10.35975/apic.v30i3.3166
Received: July 20, 2025; Revised: August 28, 2025; Accepted: September 01, 2025
Lung cancer is one of the most prevalent and deadly malignancies worldwide and accounts for a significant proportion of cancer-related morbidity and mortality. It is broadly categorized into two main types: non-small cell lung cancer (NSCLC), which constitutes approximately 85% of all cases, and small-cell lung cancer (SCLC), which is less common but more aggressive.1 The primary risk factor for lung cancer is tobacco smoking, which is responsible for nearly 85% of lung cancer cases, although other factors such as environmental exposure (e.g., radon, asbestos, and air pollution) and genetic predispositions also play critical roles.2 Geographic variations in lung cancer prevalence are evident, with higher incidence rates observed in developed regions, such as North America, Europe, and Eastern Asia, largely due to smoking prevalence and environmental factors, such as air pollution.1
Oxidative stress, due to an imbalance between reactive oxygen species (ROS) generation and the antioxidant defense system, plays a crucial role in the pathogenesis and proliferation of lung cancer. ROS can produce DNA adducts, lipid peroxides, and protein carbonyls, resulting in genomic instability and oncogenic signals.3 Oxidative stress is induced by various environmental factors (e.g., tobacco smoke) and is highly expected in lung cancer, as tobacco smoke and air pollution are the dominant factors that induce ROS in the lungs.4 Superoxide dismutase is a central antioxidant enzyme that is important in biological defense against reactive oxygen species (ROS). SOD, the first line of defense in the suppression of oxidative stress, catalyzes the dismutation of superoxide anion radicals (O2-) to H2O2 and O2.5 The overproduction of ROS, including hydroxyl radicals, hydrogen peroxide, and superoxide anions, is associated with many harmful effects on various biomolecules, including DNA damage, which is one of the most dangerous threats to the integrity and function of cells.6 Superoxide dismutase plays an important role in biological defense mechanisms against the tumorigenic effects of activated oxygen and free radicals. Overexpression of SOD in cell lines has been shown to exert inhibitory effects on tumor cell growth. As increased SODs activity has shown suppressive effects on the growth of several malignant tumors, SODs may be considered tumor suppressors.7 Glutathione a tripeptide composed of glutamine, cysteine, and glycine, is the most abundant non-protein thiol in mammalian cells, present at millimolar concentrations, and serves as a critical intracellular antioxidant that regulates cellular redox balance and protects cells from damage caused by ROS, reactive nitrogen species, and xenobiotics. It antioxidative functions, GSH plays a pivotal role in signal transduction, influencing cell differentiation, proliferation, apoptosis, ferroptosis, and immune responses.8 Glutathione metabolism plays a complex role in cancer, acting as both a protector against oxidative stress and promoter of tumor growth. High GSH levels in cancer cells are linked to increased tumor progression and resistance to chemotherapy. This resistance arises because GSH can bind to and detoxify chemotherapeutic agents, reducing their effectiveness and allowing cancer cells to survive lethal treatments.9 Malondialdehyde as oxidative stress biomarkers is a reactive aldehyde and a classic biomarker of oxidative stress that is produced as a by-product of lipid peroxidation in cell membranes. MDA is a small, three-carbon molecule with the chemical formula C₃H₄O₂ with X2C units on the terminal carbon atoms. This configuration causes MDA to become highly reactive and form covalent adducts with proteins, DNA, and other cellular components, resulting in conformational and functional changes that are responsible for cellular damage. MDA production is closely related to the presence of ROS, which result in the initiation of lipid peroxidation under oxidative stress, a common trend found in numerous diseases such as cancer, cardiovascular diseases, and neurodegenerative diseases.10,11
The soluble fragment of cytokeratin 19, an epithelial cell intermediate filament-related protein, is called cytokeratin (CYFRA) fragment antigen 21-1 (CYFRA 21-1). It has been reported to be a tumor marker in many studies, especially in NSCLC, in which increased serum levels reflect the progression and poor prognosis of NSCLC.12 The biological properties of CYFRA 21-1 especially with its epithelial specificity and structural stability, justify its clinical use in oncology. Proteomics and biomarker studies have continued to evolve, and personalized cancer management has been targeted.13 Functionally CYFRA 21-1 is a surrogate marker of the turnover and apoptosis of epithelial cells. High serum and plasma concentrations of CYFRA 21-1 are frequently linked to enhanced cell proliferation, cell necrosis, and tumor burden, especially in epithelial-origin tumors.14 It is clinically useful, especially in NSCLC, and has been found to correlate with the tumor stage, prognosis, and therapeutic response. Furthermore, CYFRA 21-1 has been investigated as a potential biomarker in other malignancies (e.g., urinary bladder, cervix, and head and neck); however, its specific and sensitive applicability for different tumor types can vary.15 The objective is to evaluate serum levels of oxidative stress markers (SOD, GPx,) and MDA as anti-oxidant biomarker and CYFRA 21-1 in lung cancer patients compared to controls used to monitoring and prognosis tool for the oxidative stress status.
This case control study was conducted between November 2024 and April 2025. This study included 60 patients with lung cancer (31 males and 29 females). The patients were diagnosed by a specialist physician at the Nassiriya Teaching Hospital, ThiQar Governorate, southern Iraq. In addition, the study included 60 healthy persons as controls (31 males and 29 females), and demographic data, including age, sex, and clinical findings, were collected using a standardized questionnaire. After collecting 10 ml of venous blood from each of the 120 participants, the blood was centrifuged to obtain serum for the measurement of SOD, Gpx, MDA and CYFRA 21-1 were measured by using enzyme-linked immunosorbent assay (ELISA). SOD and GPx and CYFRA 21-1 manufactured by BT-LAB (China) with Cat.No (E0700Hu, E3921Hu, E1619Hu) respectively. MDA by Elabscience/ USA (Cat.No E-EL-0060)
Inclusion criteria include confirmed lung cancer diagnosis determined clinically and pathologically also use CT scan and Histopathological examination. Age: 28 to 81 years; both male and female. Includes smokers and non-smokers. Any disease stage (I–IV)- Patients receiving active treatment (chemotherapy, radiotherapy, immunotherapy, targeted therapy). Exclusion criteria include previous or current cancer. Use of antioxidant or vitamin D supplements. Chronic inflammatory or autoimmune disease
2.1. Statistical Analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 26, developed by IBM, SPSS Inc., USA. The study employed an independent sample t-test for parametric variables. The outcome was deemed statistically significant based on a p-value of less than or equal to 0.05.
The patients and controls in this study ranged in age from 27 to 60 years. Baseline demographic and anthropometric variables were compared between the 60 healthy controls and 60 patients with lung cancer. Table 1 exhibited no statistically significant differences in age (mean: 52.8 ± 12.7 vs.49.53 ± 10.4; P = 0.09). Notably, lung cancer patients displayed a lower mean BMI (20.5 ± 1.3 kg/m²) compared to controls (25.34 ± 3.0 kg/m²; P = 0.2), though this difference showed non-significant difference.
Regarding sex between the two groups, there was a non-significant difference between them (P - 0.85), 60 patients with lung cancer (31 males and 29 females), and 60 healthy persons as controls (32 males and 28 females). The frequency was the highest in the fourth stage 31 (51%) and lowest in the first stage 4 (6%). As shown in Figure 1.
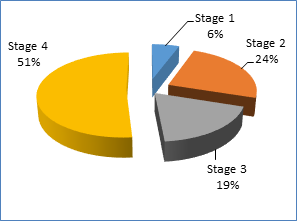
Figure 1: Prevalence of lung cancer stages among patients
Oxidative stress markers demonstrated pronounced intergroup disparities. MDA, a lipid peroxidation product, was elevated in cancer patients (193.2 ± 18.6 nmol/mL vs. 158.6 ± 21.09 nmol/mL; P < 0.001), reflecting heightened oxidative damage. Antioxidant enzymes also diverged: GPX-1 and SOD-1 levels were significantly higher in patients (65.4 ± 29.4 μU/mL vs. 9.98 ± 5.6 μU/mL and 90.15 ± 34.9 ng/mL vs. 42.07 ± 13.78 ng/mL, respectively; P < 0.001) (Table 2).
CYFRA21-1, a cytokeratin fragment associated with epithelial malignancy, was significantly elevated in lung cancer patients (15.1 ± 6.5 ng/mL vs. 5.9 ± 2.7 ng/mL; P < 0.001) (Table 2).
The levels of MDA, GPx, and SOD did not show any significant difference among age subgroup in patient with lung cancer (P - 0.95, P - 0.61, and P - 0.28, respectively), as show in Table 3.
When comparing the levels of CYFRA21-1 among the age subgroup, there was a non-significant difference (P - 0.40) among the groups, as shown in tables 2-3.
The findings of the current study showed a high level of oxidative stress markers in lung cancer patients when compared with healthy controls (Table 2). These findings are in agreement with those of Sánchez-Pozos et al. (2015), in which lung cancer patients with concomitant COPD had significantly higher levels of SOD1 and SOD2 proteins and MDA-protein adducts than patients with lung cancer and healthy controls.16
This is also consistent with a prospective comparative study by Yugraten et al. (2021), who reported higher SOD and GPx activities in both metastatic and non-metastatic lung cancer patients and higher TBARS levels (MDA equivalents) in patients than in healthy individuals.17 With reference to patients with lung cancer, in the study by Sonkar et al. (2019), serum oxidative marker analysis revealed a significant increase in TBARS (MDA surrogate).18 They also found higher SOD activity and GPx differences between the patients and controls. Superoxide dismutase is a first-line defense and antioxidant enzyme produced by the body to catalyze the dismutation of superoxide to hydrogen peroxide. High SOD activity in lung cancer patients may initially compensate for ROS production. A study by Tas et al. (2005) reported that SOD activity in the erythrocytes of non-small cell lung cancer (NSCLC) patients was markedly higher than that in the erythrocytes of normal subjects and explained these findings by the increase in oxidative stress in association with tumor metabolic changes and inflammation.19 Conversely, elevated SOD levels have been
regarded as a response to the high oxidative burden created by tumor hypoxia and mitochondrial dysfunction.20 GPx is another important antioxidant enzyme that is desirable for reducing hydrogen peroxide and lipid peroxides through the action of glutathione. Notably, some studies have described increased or decreased GPx activity in lung cancer patients, which could be related to the disease stage or
heterogeneity of tumor biology. For instance, Acharya et al. (2010) reported that the GPx activity of NSCLC patients was significantly higher,21 suggesting that higher GPx activity might be a protective mechanism for the overproduction of hydrogen peroxide. "But if stress becomes chronic, the enzyme can be used up or compromised." Our current findings are consistent with these later studies, since we observed upstream GPx, which indicates continuing oxidative stress and an active but possibly overwhelmed antioxidant defense.22 Malondialdehyde ia a product of lipid peroxidation, is an indicator of oxidative damage to cellular membranes. The marked increase in MDA levels in lung cancer patients reflects enhanced lipid peroxidation, and therefore, oxidative damage. This result is consistent with that of a study by Gackowski et al. (2008), who reported high MDA levels in the plasma and lung tissues of patients with lung cancer.23 The elevated lipid peroxidation level may be attributed to enhanced ROS generation by either tumor or immune cells, especially under conditions of chronic inflammation and exposure to tobacco smoke, a known stimulus for oxidative stress.24 Overall, these results further support the hypothesis that Mal is implicated in the pathogenesis of lung cancer via the induction of oxidative stress. These findings are in accordance with increasing evidence on the involvement of an oxidant/antioxidant imbalance in the development and progression of lung cancer.25
In the current study, increased CYFRA 21-1 level in the sera of lung cancer patients were statistically significant compared to the levels in the control group. This finding is in accordance with the report by Li et al. (26), who found significantly higher levels of CYFRA 21‑1 in malignant versus benign pulmonary nodules in their pilot study. Similarly, Molina et al. (2003) showed that CYFRA 21-1 was more significant in patients with advanced-stage NSCLC, and the level of CYFRA 21-1 was directly related to tumor progression and poor survival.27 CYFRA 21-1 is a cytokeratin 19 soluble fragment that belongs to the intermediate filament protein family in epithelial cells. In lung tumors, particularly squamous cell carcinoma, loss of cytoskeletal integrity due to tumor invasion and necrosis causes diffusion of cytokeratin fragments into the blood.28 This increase in CYFRA 21-1 is not only a result of association, but also the pathophysiological outgrowth of tumor activity. Pujol et al. (2004) found that CYFRA 21-1 had a higher sensitivity of 65%, with a low specificity of 90% for detecting NSCLC, whereas other markers, such as CEA, have a sensitivity of 30% for lung histology.29 The marked elevation detected in the present study potentially reflects the high expression of cytokeratin 19 within malignant epithelial cells and its subsequent release upon cell death. Furthermore, the lack of these high values in healthy individuals confirms the specificity of CYFRA 21-1 for neoplastic processes and not benign pulmonary diseases.30 This specificity is of vital importance, especially in clinical practice, with the need for the differential diagnosis of malignancy and inflammatory or infectious diseases of the urinary lung. Serum levels of CYFRA 21-1 differ among the different histological types of lung cancer. According to Muley et al. (2004), squamous cell carcinoma showed higher CYFRA 21-1 levels than adenocarcinoma because of the tissue-restricted expression of cytokeratin 19.31 A high baseline level is frequently observed and is closely related to poor treatment response and overall survival, indicating its applications not only in diagnostic measurement but also in monitoring treatment.32 This is consistent with the biological role of this biomarker as a marker of tumor mass and cell breakdown. High CYFRA 21-1 levels in lung cancer cases emphasize the importance of this biomarker for both diagnosis and prognosis. These results support existing evidence and suggest that CYFRA 21-1 should be included in the standard workup of patients with suspected lung cancer.33
This study highlights a significant elevation in oxidative stress markers and CYFRA 21-1 in patients with lung cancer, supporting their potential role as used to monitoring and prognosis for the oxidative stress status. Monitoring these biomarkers may enhance early detection and management of lung cancer in clinical settings.
The study has several limitations including a limited sample size, single-center study, and non-inclusion of lung cancer subtypes.
7. Availability of data
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
8. Acknowledgments
We accomplished this all on our own, with no outside financial assistance or scholarships. We would like to express our gratitude to everyone at the Nassiriya Teaching Hospital in Dhi Qar for their help with patient diagnoses and blood draws.
9. Financial support
Nil. The study did not involve any external or industry sponsorship.
10. Conflicts of interest
The authors declare no conflicts of interest.
11. Authors contribution
MKM: Conceptualization, Methodology, Formal Analysis, Original Draft Preparation, Supervision
AAH: Validation, Investigation, Data Curation, Writing – Review & Editing
SAH: Software, Resources, Visualization, Project Administration, Funding Acquisition
Authors affiliations:
- Mohammed H. Mohammed, College of Health and Medical Technologies, Southern Technical University, Sulaymaniyah, Kurdistan, Iraq; Email: mohammed.k.mohammed@fgs.stu.edu.iq 2. Shrouk A. Hassan, College of Health and Medical Technologies, Southern Technical University, Sulaymaniyah, Kurdistan, Iraq; Email: shrouk.albraheem@stu.edu.iq 3. Ali A. Hassan, Nasiriyah Technical Institute, Southern Technical University, Basrah, Basra Governorate, Iraq; Email: alialbakaa14@stu.edu
ABSTRACT
Background: Lung cancer is a leading cause of cancer mortality, linked to anti-oxidant and oxidative stress and epithelial damage. Biomarkers like superoxide dismutase (SOD), glutathione peroxidase (GPx), malondialdehyde (MDA), and cytokeratin fragment 21-1 (CYFRA 21-1) may indicate tumor progression.
Objective: To assess serum levels of oxidative stress markers (SOD, GPx, MDA) and CYFRA 21-1 in lung cancer patients versus controls used to monitoring and prognosis tool for the oxidative stress status.
Methodology: This case-control study was carried out from November 2024 to April 2025 at Nassiriya Teaching Hospital in Thi-Qar, Iraq. Sixty patients with lung cancer and 60 age-matched controls were included in the study. Venous blood samples were obtained, and serum was separated and stored to determine the concentrations of SOD, Gpx, MDA and CYFRA 21-1 using enzyme-linked immunosorbent assay (ELISA).
Results: There were no significant differences in age or BMI between the two groups. Lung cancer patients had significantly elevated levels of MDA (193.2 ± 18.6 vs. 158.6 ± 21.09 nmol/mL), SOD (90.15 ± 34.9 vs. 42.07 ± 13.78 ng/mL), GPx (65.4 ± 29.4 vs. 9.98 ± 5.6 µU/mL), and CYFRA 21-1 (15.1 ± 6.5 vs. 5.9 ± 2.7 ng/mL) compared to controls (P < 0.001 for all). Oxidative stress markers and CYFRA 21-1 levels did not differ significantly among age subgroups in patients with lung cancer.
Conclusions: This study highlights a significant elevation in oxidative stress markers and CYFRA 21-1 in patients with lung cancer, supporting their potential role as diagnostic and prognostic biomarkers. Monitoring these biomarkers may enhance early detection and management of lung cancer in clinical settings.
Keywords: Lung cancer, Oxidative stress, Cytokeratin fragment 21-1
Citation: Mohammed MH, Hassan SA, Hassan AA. Evaluation of oxidative stress and diagnostic value of cytokeratin fragment 21-1 in lung cancer patients. Anaesth. pain intensive care 2026;30(3):301-307. DOI: 10.35975/apic.v30i3.3166
Received: July 20, 2025; Revised: August 28, 2025; Accepted: September 01, 2025
1. INTRODUCTION
Lung cancer is one of the most prevalent and deadly malignancies worldwide and accounts for a significant proportion of cancer-related morbidity and mortality. It is broadly categorized into two main types: non-small cell lung cancer (NSCLC), which constitutes approximately 85% of all cases, and small-cell lung cancer (SCLC), which is less common but more aggressive.1 The primary risk factor for lung cancer is tobacco smoking, which is responsible for nearly 85% of lung cancer cases, although other factors such as environmental exposure (e.g., radon, asbestos, and air pollution) and genetic predispositions also play critical roles.2 Geographic variations in lung cancer prevalence are evident, with higher incidence rates observed in developed regions, such as North America, Europe, and Eastern Asia, largely due to smoking prevalence and environmental factors, such as air pollution.1
Oxidative stress, due to an imbalance between reactive oxygen species (ROS) generation and the antioxidant defense system, plays a crucial role in the pathogenesis and proliferation of lung cancer. ROS can produce DNA adducts, lipid peroxides, and protein carbonyls, resulting in genomic instability and oncogenic signals.3 Oxidative stress is induced by various environmental factors (e.g., tobacco smoke) and is highly expected in lung cancer, as tobacco smoke and air pollution are the dominant factors that induce ROS in the lungs.4 Superoxide dismutase is a central antioxidant enzyme that is important in biological defense against reactive oxygen species (ROS). SOD, the first line of defense in the suppression of oxidative stress, catalyzes the dismutation of superoxide anion radicals (O2-) to H2O2 and O2.5 The overproduction of ROS, including hydroxyl radicals, hydrogen peroxide, and superoxide anions, is associated with many harmful effects on various biomolecules, including DNA damage, which is one of the most dangerous threats to the integrity and function of cells.6 Superoxide dismutase plays an important role in biological defense mechanisms against the tumorigenic effects of activated oxygen and free radicals. Overexpression of SOD in cell lines has been shown to exert inhibitory effects on tumor cell growth. As increased SODs activity has shown suppressive effects on the growth of several malignant tumors, SODs may be considered tumor suppressors.7 Glutathione a tripeptide composed of glutamine, cysteine, and glycine, is the most abundant non-protein thiol in mammalian cells, present at millimolar concentrations, and serves as a critical intracellular antioxidant that regulates cellular redox balance and protects cells from damage caused by ROS, reactive nitrogen species, and xenobiotics. It antioxidative functions, GSH plays a pivotal role in signal transduction, influencing cell differentiation, proliferation, apoptosis, ferroptosis, and immune responses.8 Glutathione metabolism plays a complex role in cancer, acting as both a protector against oxidative stress and promoter of tumor growth. High GSH levels in cancer cells are linked to increased tumor progression and resistance to chemotherapy. This resistance arises because GSH can bind to and detoxify chemotherapeutic agents, reducing their effectiveness and allowing cancer cells to survive lethal treatments.9 Malondialdehyde as oxidative stress biomarkers is a reactive aldehyde and a classic biomarker of oxidative stress that is produced as a by-product of lipid peroxidation in cell membranes. MDA is a small, three-carbon molecule with the chemical formula C₃H₄O₂ with X2C units on the terminal carbon atoms. This configuration causes MDA to become highly reactive and form covalent adducts with proteins, DNA, and other cellular components, resulting in conformational and functional changes that are responsible for cellular damage. MDA production is closely related to the presence of ROS, which result in the initiation of lipid peroxidation under oxidative stress, a common trend found in numerous diseases such as cancer, cardiovascular diseases, and neurodegenerative diseases.10,11
The soluble fragment of cytokeratin 19, an epithelial cell intermediate filament-related protein, is called cytokeratin (CYFRA) fragment antigen 21-1 (CYFRA 21-1). It has been reported to be a tumor marker in many studies, especially in NSCLC, in which increased serum levels reflect the progression and poor prognosis of NSCLC.12 The biological properties of CYFRA 21-1 especially with its epithelial specificity and structural stability, justify its clinical use in oncology. Proteomics and biomarker studies have continued to evolve, and personalized cancer management has been targeted.13 Functionally CYFRA 21-1 is a surrogate marker of the turnover and apoptosis of epithelial cells. High serum and plasma concentrations of CYFRA 21-1 are frequently linked to enhanced cell proliferation, cell necrosis, and tumor burden, especially in epithelial-origin tumors.14 It is clinically useful, especially in NSCLC, and has been found to correlate with the tumor stage, prognosis, and therapeutic response. Furthermore, CYFRA 21-1 has been investigated as a potential biomarker in other malignancies (e.g., urinary bladder, cervix, and head and neck); however, its specific and sensitive applicability for different tumor types can vary.15 The objective is to evaluate serum levels of oxidative stress markers (SOD, GPx,) and MDA as anti-oxidant biomarker and CYFRA 21-1 in lung cancer patients compared to controls used to monitoring and prognosis tool for the oxidative stress status.
2. METHODOLOGY
This case control study was conducted between November 2024 and April 2025. This study included 60 patients with lung cancer (31 males and 29 females). The patients were diagnosed by a specialist physician at the Nassiriya Teaching Hospital, ThiQar Governorate, southern Iraq. In addition, the study included 60 healthy persons as controls (31 males and 29 females), and demographic data, including age, sex, and clinical findings, were collected using a standardized questionnaire. After collecting 10 ml of venous blood from each of the 120 participants, the blood was centrifuged to obtain serum for the measurement of SOD, Gpx, MDA and CYFRA 21-1 were measured by using enzyme-linked immunosorbent assay (ELISA). SOD and GPx and CYFRA 21-1 manufactured by BT-LAB (China) with Cat.No (E0700Hu, E3921Hu, E1619Hu) respectively. MDA by Elabscience/ USA (Cat.No E-EL-0060)
Inclusion criteria include confirmed lung cancer diagnosis determined clinically and pathologically also use CT scan and Histopathological examination. Age: 28 to 81 years; both male and female. Includes smokers and non-smokers. Any disease stage (I–IV)- Patients receiving active treatment (chemotherapy, radiotherapy, immunotherapy, targeted therapy). Exclusion criteria include previous or current cancer. Use of antioxidant or vitamin D supplements. Chronic inflammatory or autoimmune disease
2.1. Statistical Analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 26, developed by IBM, SPSS Inc., USA. The study employed an independent sample t-test for parametric variables. The outcome was deemed statistically significant based on a p-value of less than or equal to 0.05.
3. RESULTS
The patients and controls in this study ranged in age from 27 to 60 years. Baseline demographic and anthropometric variables were compared between the 60 healthy controls and 60 patients with lung cancer. Table 1 exhibited no statistically significant differences in age (mean: 52.8 ± 12.7 vs.49.53 ± 10.4; P = 0.09). Notably, lung cancer patients displayed a lower mean BMI (20.5 ± 1.3 kg/m²) compared to controls (25.34 ± 3.0 kg/m²; P = 0.2), though this difference showed non-significant difference.
| Table 1: Socio-demographic and anthropometric measurement characteristics among the study groups | |||
| Characteristic | Patients group
(n = 60) |
Control group
(n = 60) |
P- value |
| Age (year) | 52.8 ± 12.7 | 49.53 ± 10.4 | 0.09 NS* |
| BMI (kg/m2) | 20.5 ± 1.3 | 25.34 ± 3.0 | 0.2 NS* |
| Gender | |||
| Male | 31 (52) | 32 (53) | 0.85 |
| Female | 29 (48) | 28 (47( | |
| NS: Non-significant, *t-test;, Data given as mean ± SD or n (%) | |||
Regarding sex between the two groups, there was a non-significant difference between them (P - 0.85), 60 patients with lung cancer (31 males and 29 females), and 60 healthy persons as controls (32 males and 28 females). The frequency was the highest in the fourth stage 31 (51%) and lowest in the first stage 4 (6%). As shown in Figure 1.

Figure 1: Prevalence of lung cancer stages among patients
Oxidative stress markers demonstrated pronounced intergroup disparities. MDA, a lipid peroxidation product, was elevated in cancer patients (193.2 ± 18.6 nmol/mL vs. 158.6 ± 21.09 nmol/mL; P < 0.001), reflecting heightened oxidative damage. Antioxidant enzymes also diverged: GPX-1 and SOD-1 levels were significantly higher in patients (65.4 ± 29.4 μU/mL vs. 9.98 ± 5.6 μU/mL and 90.15 ± 34.9 ng/mL vs. 42.07 ± 13.78 ng/mL, respectively; P < 0.001) (Table 2).
CYFRA21-1, a cytokeratin fragment associated with epithelial malignancy, was significantly elevated in lung cancer patients (15.1 ± 6.5 ng/mL vs. 5.9 ± 2.7 ng/mL; P < 0.001) (Table 2).
| Table 2: Oxidative stress and anti-oxidant marker profiles and the levels of CYFRA21-1 in healthy controls and lung cancer patients | |||
| Parameters
|
Patient group
(n = 60) |
Control group
(n = 60) |
P-value |
| MDA (nmol/mL) | 193.2 ± 18.6 | 158.6 ± 21.09 | <0.001 |
| GPx (µU/mL) | 65.4 ± 29.4 | 9.98 ± 5.6 | <0.001 |
| SOD (ng/mL) | 90.15 ± 34.9 | 42.07 ± 13.78 | <0.001 |
| CYFRA21-1 (ng/ml) | 15.1 ± 6.5 | 5.9 ± 2.7 | <0.001 |
| Data presented as mean ± SD; P 0,05 considered as significant | |||
The levels of MDA, GPx, and SOD did not show any significant difference among age subgroup in patient with lung cancer (P - 0.95, P - 0.61, and P - 0.28, respectively), as show in Table 3.
| Table 3: Oxidative stress and anti-oxidant marker profiles and the levels of CYFRA21-1 in lung cancer patients according to age subgroup | ||||
| Parameters | 28-47 years | 48-67 years | > 68 years | P- value |
| MDA (nmol/mL) | 195.70 ± 15.68 | 193.89 ± 20.20 | 195.39 ± 21.15 | 0.95 |
| GPx (µU/mL) | 69. 66 ± 20.64 | 60.87 ± 28.17 | 59.78 ± 18.63 | 0.61 |
| SOD (ng/mL) | 86.96 ± 29.99 | 90.58 ± 26.16 | 94.47 ± 36.79 | 0.28 |
| CYFRA21-1 (ng/ml) | 10.188 ± 3.58 | 13.86 ± 7.93 | 15.52 ± 8.15 | 0.40 |
When comparing the levels of CYFRA21-1 among the age subgroup, there was a non-significant difference (P - 0.40) among the groups, as shown in tables 2-3.
4. DISCUSSION
The findings of the current study showed a high level of oxidative stress markers in lung cancer patients when compared with healthy controls (Table 2). These findings are in agreement with those of Sánchez-Pozos et al. (2015), in which lung cancer patients with concomitant COPD had significantly higher levels of SOD1 and SOD2 proteins and MDA-protein adducts than patients with lung cancer and healthy controls.16
This is also consistent with a prospective comparative study by Yugraten et al. (2021), who reported higher SOD and GPx activities in both metastatic and non-metastatic lung cancer patients and higher TBARS levels (MDA equivalents) in patients than in healthy individuals.17 With reference to patients with lung cancer, in the study by Sonkar et al. (2019), serum oxidative marker analysis revealed a significant increase in TBARS (MDA surrogate).18 They also found higher SOD activity and GPx differences between the patients and controls. Superoxide dismutase is a first-line defense and antioxidant enzyme produced by the body to catalyze the dismutation of superoxide to hydrogen peroxide. High SOD activity in lung cancer patients may initially compensate for ROS production. A study by Tas et al. (2005) reported that SOD activity in the erythrocytes of non-small cell lung cancer (NSCLC) patients was markedly higher than that in the erythrocytes of normal subjects and explained these findings by the increase in oxidative stress in association with tumor metabolic changes and inflammation.19 Conversely, elevated SOD levels have been
regarded as a response to the high oxidative burden created by tumor hypoxia and mitochondrial dysfunction.20 GPx is another important antioxidant enzyme that is desirable for reducing hydrogen peroxide and lipid peroxides through the action of glutathione. Notably, some studies have described increased or decreased GPx activity in lung cancer patients, which could be related to the disease stage or
heterogeneity of tumor biology. For instance, Acharya et al. (2010) reported that the GPx activity of NSCLC patients was significantly higher,21 suggesting that higher GPx activity might be a protective mechanism for the overproduction of hydrogen peroxide. "But if stress becomes chronic, the enzyme can be used up or compromised." Our current findings are consistent with these later studies, since we observed upstream GPx, which indicates continuing oxidative stress and an active but possibly overwhelmed antioxidant defense.22 Malondialdehyde ia a product of lipid peroxidation, is an indicator of oxidative damage to cellular membranes. The marked increase in MDA levels in lung cancer patients reflects enhanced lipid peroxidation, and therefore, oxidative damage. This result is consistent with that of a study by Gackowski et al. (2008), who reported high MDA levels in the plasma and lung tissues of patients with lung cancer.23 The elevated lipid peroxidation level may be attributed to enhanced ROS generation by either tumor or immune cells, especially under conditions of chronic inflammation and exposure to tobacco smoke, a known stimulus for oxidative stress.24 Overall, these results further support the hypothesis that Mal is implicated in the pathogenesis of lung cancer via the induction of oxidative stress. These findings are in accordance with increasing evidence on the involvement of an oxidant/antioxidant imbalance in the development and progression of lung cancer.25
In the current study, increased CYFRA 21-1 level in the sera of lung cancer patients were statistically significant compared to the levels in the control group. This finding is in accordance with the report by Li et al. (26), who found significantly higher levels of CYFRA 21‑1 in malignant versus benign pulmonary nodules in their pilot study. Similarly, Molina et al. (2003) showed that CYFRA 21-1 was more significant in patients with advanced-stage NSCLC, and the level of CYFRA 21-1 was directly related to tumor progression and poor survival.27 CYFRA 21-1 is a cytokeratin 19 soluble fragment that belongs to the intermediate filament protein family in epithelial cells. In lung tumors, particularly squamous cell carcinoma, loss of cytoskeletal integrity due to tumor invasion and necrosis causes diffusion of cytokeratin fragments into the blood.28 This increase in CYFRA 21-1 is not only a result of association, but also the pathophysiological outgrowth of tumor activity. Pujol et al. (2004) found that CYFRA 21-1 had a higher sensitivity of 65%, with a low specificity of 90% for detecting NSCLC, whereas other markers, such as CEA, have a sensitivity of 30% for lung histology.29 The marked elevation detected in the present study potentially reflects the high expression of cytokeratin 19 within malignant epithelial cells and its subsequent release upon cell death. Furthermore, the lack of these high values in healthy individuals confirms the specificity of CYFRA 21-1 for neoplastic processes and not benign pulmonary diseases.30 This specificity is of vital importance, especially in clinical practice, with the need for the differential diagnosis of malignancy and inflammatory or infectious diseases of the urinary lung. Serum levels of CYFRA 21-1 differ among the different histological types of lung cancer. According to Muley et al. (2004), squamous cell carcinoma showed higher CYFRA 21-1 levels than adenocarcinoma because of the tissue-restricted expression of cytokeratin 19.31 A high baseline level is frequently observed and is closely related to poor treatment response and overall survival, indicating its applications not only in diagnostic measurement but also in monitoring treatment.32 This is consistent with the biological role of this biomarker as a marker of tumor mass and cell breakdown. High CYFRA 21-1 levels in lung cancer cases emphasize the importance of this biomarker for both diagnosis and prognosis. These results support existing evidence and suggest that CYFRA 21-1 should be included in the standard workup of patients with suspected lung cancer.33
5. CONCLUSION
This study highlights a significant elevation in oxidative stress markers and CYFRA 21-1 in patients with lung cancer, supporting their potential role as used to monitoring and prognosis for the oxidative stress status. Monitoring these biomarkers may enhance early detection and management of lung cancer in clinical settings.
6. LIMITATION
The study has several limitations including a limited sample size, single-center study, and non-inclusion of lung cancer subtypes.
7. Availability of data
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
8. Acknowledgments
We accomplished this all on our own, with no outside financial assistance or scholarships. We would like to express our gratitude to everyone at the Nassiriya Teaching Hospital in Dhi Qar for their help with patient diagnoses and blood draws.
9. Financial support
Nil. The study did not involve any external or industry sponsorship.
10. Conflicts of interest
The authors declare no conflicts of interest.
11. Authors contribution
MKM: Conceptualization, Methodology, Formal Analysis, Original Draft Preparation, Supervision
AAH: Validation, Investigation, Data Curation, Writing – Review & Editing
SAH: Software, Resources, Visualization, Project Administration, Funding Acquisition
12. REFERENCES
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-49. DOI: 3322/caac.21660
- Bade BC, Dela Cruz CS. Lung cancer 2020: Epidemiology, etiology, and prevention. Clin Chest Med. 2020;41(1):1-24. DOI: 1016/j.ccm.2019.10.001
- Sies H, Belousov VV, Chandel NS, Davies MJ, Jones DP, Mann GE, et al. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat Rev Mol Cell Biol. 2022;23(7):499-515. DOI: 1038/s41580-022-00456-z
- Zhang H, Forman HJ, Choi YJ. Oxidative stress response and Nrf2 signaling in aging. Free Radic Biol Med. 2020;88(Pt B):314-36. PMCID: PMC4628850 DOI: 1016/j.freeradbiomed.2015.05.036
- Zheng M, Liu Y, Zhang G, Yang Z, Xu W, Chen Q. The applications and mechanisms of superoxide dismutase in medicine, food, and cosmetics. Antioxidants (Basel). 2023;12(9):1675. PMCID: PMC10525108 DOI: 3390/antiox12091675
- Ayubi N, Wibawa JC, Ming JW. Analyzing superoxide dismutase as a molecular signal transducer that is beneficial for health during physical exercise: A systematic review. Al-Kindy Coll Med J. 2024;20(2):82-8.
- Adachi Y, Nojima M, Mori M, Yamano HO, Sasaki Y, Nakase H, et al. Association of serum superoxide dismutase activity and the incidence of colorectal cancer in a nested case-control study. Cancer Epidemiol. 2023;87:102455. DOI: 1016/j.canep.2023.102455
- Kennedy L, Sandhu JK, Harper ME, Cuperlovic-Culf M. Role of glutathione in cancer: From mechanisms to therapies. Biomolecules. 2020;10(10):1429. PMCID: PMC7600400 DOI: 3390/biom10101429
- Bansal A, Simon MC. Glutathione metabolism in cancer progression and treatment resistance. J Cell Biol. 2018;217(7):2291-8.
- Ayala A, Muñoz MF, Argüelles S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev. 2014;2014:360438.
- Del Rio D, Stewart AJ, Pellegrini N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc Dis. 2005;15(4):316-28.
- Stieber P, Hatz R, Holdenrieder S. CYFRA 21-1 as a biomarker in lung cancer: Current evidence and future perspectives. Clin Chem Lab Med. 2020;58(5):701-12.
- Wang L, Chen Y, Li H. Advances in the application of CYFRA 21-1 in cancer diagnostics and prognosis. Biomark Med. 2022;16(4):245-56.
- Tang J, Shu HY, Sun T, Zhang LJ, Kang M, Ying P, et al. Potential factors of cytokeratin fragment 21-1 and cancer embryonic antigen for mediastinal lymph node metastasis in lung cancer. Front Genet. 2022;13:1009141.
- Holdenrieder S, Wehnl B, Hettwer K, Simon K, Uhlig S, Dayyani F. Carcinoembryonic antigen and cytokeratin-19 fragments for assessment of therapy response in non-small cell lung cancer: A systematic review and meta-analysis. Br J Cancer. 2017;116(8):1037-45.
- Sánchez Pozos R, Torres-Del Real JP, Ortega-López JD, Jiménez-Capdeville ME. Redox imbalance in lung cancer of patients with underlying chronic respiratory conditions. Mol Med. 2015;21:123-32.
- Yugraten B, Özdemir İH, Demir MÖ, Tuncel T. Evaluation of oxidative stress biomarkers in brain metastatic and non-metastatic lung cancer patients with different cell types. Eur J Respir Med. 2021;2021(2):45-53.
- Sonkar AB, Verma A, Yadav S, Kumar R, Singh J, Keshari AK, et al. Oxidative stress in lung cancer patients is associated with altered serum markers of lipid metabolism. Cancer Cell Int. 2019;19(1):219.
- Tas F, Hansel H, Belce A, Ilvan S, Argon A, Camlica H, et al. Oxidative stress in breast cancer. Med Oncol. 2005;22(1):11-5.
- Kinnula VL, Crapo JD. Superoxide dismutases in malignant cells and human tumors. Free Radic Biol Med. 2004;36(6):718-44.
- Acharya A, Das I, Singh S, Saha T. Chemopreventive properties of indole-3-carbinol, a cruciferous phytochemical, in combating cancer by targeting multiple signaling pathways. Anticancer Agents Med Chem. 2010;10(3):249-58.
- Nowakowska E, Główka F, Kowalczyk Pachel D, Mrozek Wilczkiewicz A. Are antioxidant enzymes essential markers in the diagnosis and monitoring of cancer patients? J Med Life. 2021;14(4):287-96.
- Gackowski D, Banaszkiewicz Z, Rozalski R, Jawien A, Olinski R. Persistent oxidative stress in lung cancer patients after surgical treatment is accompanied by decreased level of 8-oxo-7,8-dihydro-2'-deoxyguanosine repair in DNA. Mutat Res. 2008;645(1-2):67-73.
- Bartsch H, Nair J. Chronic inflammation and oxidative stress in the genesis and perpetuation of cancer: Role of lipid peroxidation, DNA damage, and repair. Lung Cancer. 2006;54 Suppl 2:S31-8. DOI: 1007/s00423-006-0073-1
- Klaunig JE, Wang Z, Pu X, Zhou S. Oxidative stress and oxidative damage in chemical carcinogenesis. Toxicol Appl Pharmacol. 2010;254(2):86-99. DOI: 1016/j.taap.2009.11.028
- Li X, Zhang Q, Jin X, Lihua Cao. Combining serum miRNAs, CEA, and CYFRA 21-1. World J Surg Oncol. 2017;15:107. PMCID: PMC5445450 DOI: 1186/s12957-017-1171-y
- Molina R, Filella X, Auge JM, Zanon G, Marín JL, Farrús B, et al. Tumor markers (CEA, CA 125, CYFRA 21-1, SCC and NSE) in patients with non-small cell lung cancer as indicators of tumor stage and histology. Tumour Biol. 2003;24(4):209-18.
- Barlesi F, Gimenez C, Torre JP, Doddoli C, Mancini J, Kleisbauer JP, et al. Prognostic value of combination of CYFRA 21-1, neuron-specific enolase, and carcinoembryonic antigen levels in patients with advanced non-small cell lung cancer. Cancer. 2004;100(9):1997-2003.
- Pujol JL, Molinier O, Ebert W, Daures JP, Barlesi F. CYFRA 21-1 is a prognostic determinant in non-small-cell lung cancer: Results of a meta-analysis in 2063 patients. Br J Cancer. 2004;90(11):2097-105.
- Nakao M, Kinoshita R, Kuriyama M, Kiyotoshi H, Sugihara M, Takeda N, et al. Serum CYFRA 21-1 level as a prognostic marker for extensive disease small cell lung cancer. Int J Anticancer Res. 2024. DOI: 21873/anticanres.16877
- Muley T, Dienemann H, Ebert W, Meister M. CYFRA 21-1 in the differentiation of pleural effusions due to lung cancer and benign diseases. Br J Cancer. 2004;91(2):332-5.
- Fu L, Wang R, Yin L, Shang X, Zhang R, Zhang P. CYFRA 21-1 tests in the diagnosis of non-small cell lung cancer: A meta-analysis. Int J Biol Markers. 2019. DOI: 1177/1724600819868234
- Dall'Olio FG, Abbati F, Facchinetti F, Massucci M, Melotti B, Squadrilli A, et al. CEA and CYFRA 21-1 as prognostic biomarker and as a tool for treatment monitoring in advanced NSCLC treated with immune checkpoint inhibitors. Ther Adv Med Oncol. 2020 Oct 31;12:1758835920952994.. PMCID: PMC7607728 DOI: 1177/1758835920952994