Nancy Boules 1 , Ramin Rahimian 2 , Taizoon Dhoon 3
Authors affiliations:
Spinal epidural hematoma (SEH) is a rare complication of neuraxial anesthesia. We present a case of a 28-year-old G2P1001 at 28 weeks' gestation underwent an urgent cesarean delivery due to preterm premature rupture of membranes, pre-eclampsia with severe features, and breech presentation. The patient received a spinal anesthetic, which required two attempts. Postoperatively, she developed progressive lower extremity weakness and sensory changes within 48 hours. An MRI revealed a 14 × 10 × 82 mm SEH extending from L3 to S1, necessitating an emergent laminectomy that resulted in favorable outcomes.
We conclude that the anesthesiologists should remain vigilant for SEH, as early detection is vital for optimal maternal outcomes.
Abbreviations: NSVD: Normal Spontaneous Vaginal Delivery, PPROM: preterm premature rupture of membranes, SEH: spinal epidural hematoma, SSS: single shot spinal
Teaching Points:
Citation: Boules N, Rahimian R, Dhoon T. Neurological emergency after cesarean section: a case of atypical spinal epidural hematoma. Anaesth. pain intensive care 2026;30(1):274-278. DOI: 10.35975/apic.v30i1.3138
Received: November 11, 2025; Revised: January 04, 2026; Accepted: January 06, 2026
Spinal Epidural Hematoma (SEH) is a rare but severe complication of neuraxial anesthesia, occurring in approximately 1 in 200,000 obstetric cases.1 While SEH is typically associated with disorders like coagulopathy, recent anticoagulation, or thrombocytopenia, it can occur in low-risk patients as well.2,3 Accumulation of blood within the epidural space can exert significant pressure on the spinal cord and nerve roots, resulting in neurological perturbations such sensory and motor deficits, bowel and bladder dysfunction, and paralysis.2,3
We present the case of a 28-year-old woman, G2P1001, who developed a SEH following a single shot spinal (SSS) anesthetic. Progression of neurological symptoms 36 hours postpartum raised concerns for SEH. Emergent surgical decompression was performed following Magnetic Resonance Imaging (MRI) which demonstrated a massive hematoma extending from L3 to S1.
This report focuses on the early identification of neurological symptoms, workup, and management, including timely surgical decompression and hematoma evacuation.
A 28-year-old G2P1001 at 28 weeks and 1 day was scheduled for an urgent cesarean section due to preterm premature rupture of membranes (PPROM), pre-eclampsia with severe features consisting of elevated liver function tests and persistent headache, and breech presentation. Her past obstetric history includes one prior normal spontaneous vaginal delivery (NSVD) with labor epidural. At the time of surgery, the patient was receiving a magnesium sulfate infusion and was not on anticoagulation therapy. Her platelet count was 134,000/μL on the day prior to surgery and 144,000/μL on the morning of the procedure.
Spinal anesthesia was performed at the L3-L4 level after two attempts using a 25-gauge needle. The first attempt caused transient left-sided buttock paresthesia, which resolved immediately after needle withdrawal. A second attempt at neuraxial placement at the same interspace was successful and well tolerated, resulting in an effective sensory block up to the T4 dermatome. The cesarean delivery was complicated by uterine atony, which was managed with intravenous oxytocin (60 units infused over 60 minutes), intramuscular carboprost tromethamine (250 micrograms), intravenous tranexamic acid (1 gram), and placement of a modified B-Lynch suture to achieve uterine compression. Following these interventions, the total estimated blood loss was 465 mL. Postoperatively, magnesium sulfate infusion was continued for seizure prophylaxis.
On postoperative day (POD) 1, the patient experienced intermittent left sided buttock paresthesia and paresthesia of the left ankle. She was able to urinate and ambulate with assistance. On physical exam, the patient exhibited preserved bilateral plantarflexion and dorsiflexion, along with intact hip flexion and extension. Sensory examination revealed intact sensation to light touch and cold bilaterally, with preservation up to the T10 dermatomal level. No saddle anesthesia was noted, and there were no clinical signs of hematoma or infection at the lower back. The perineal numbness was initially thought to be related to residual effects of spinal anesthesia and left ankle paresthesia due to a nerve irritation from the first spinal attempt; both symptoms appeared to be resolving on POD 1.
On POD 2, the patient continued to experience persistent bilateral buttock paresthesias and reported new-onset perineal numbness. She also described worsening paresthesia and newly developed motor weakness in the left ankle and foot as well as left thigh and calf tightness. On physical exam, the patient exhibited diminished left-sided plantarflexion and dorsiflexion, reduced flexion and extension of the left toes, and intact hip flexion and extension. Sensory examination revealed intact sensation to light touch and cold bilaterally, with preservation up to the T10 dermatomal level. Urinary and bowel function remained at baseline.
An emergent MRI was obtained, and neurology and neurosurgical consultation was initiated. Complete blood count (CBC) and coagulation profiles were within normal limits, with a platelet count of 124,000/μL. MRI revealed a 14 mm x 10 mm x 82 mm epidural hematoma extending from L3 to S1, resulting in significant mass effect on the spinal canal and severe spinal canal stenosis, possibly reflecting cauda equina syndrome [Figure 1]. Neurosurgery was immediately consulted, and the patient underwent emergent L3-L5 laminectomy for evacuation of epidural hematoma, approximately 48 hours after the initial SSS. Preoperatively, the patient's complete blood count showed a white blood cell count of 9.9 ×10⁹/L, hemoglobin of 12.5 g/dL, hematocrit of 35.5%, and platelet count of 144,000/µL. Coagulation studies were not obtained.
At one-year follow-up, the patient's left leg paresthesia and motor deficits had fully resolved. However, she continued to experience intermittent right-sided perineal paresthesia, which she described as noticeable but not bothersome. Her bowel and bladder function remained stable and unchanged from baseline.
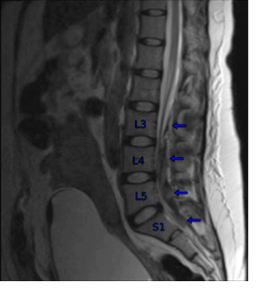
Figure 1: Sagittal MRI images of the lumbar spine showing a 14 x 10 x 82 mm heterogeneous enhancement of the dorsal aspect of the thecal sac extending from L3 to S1, with significant mass effect on thecal sac and compression on the nerve roots
3. DISCUSSION
SEH, though rare, can occur following spinal and epidural techniques if a blood vessel is punctured by a needle or catheter.6 SEH is associated with patients who have coagulation disorders or are on anticoagulation but can also occur in low-risk patients.2,3 The incidence of SEH is significantly lower in obstetric patients, with retrospective studies reporting an incidence between 0 and 0.6 per 100,000 epidural catheterizations.10 For spinal anesthetics, the incidence has been estimated to be less than 1 in 220,000 in the obstetric population.1 The veins, surrounded by loose areolar tissue, are prone to rupture, especially in pregnancy due to increased venous pressure from uterine enlargement and hemodynamic changes.1,2 While venous bleeding is widely accepted as the source of SEH, some debate persists over an arterial origin, as arterial pressure may be more capable of compressing the spinal cord than lower-pressure venous bleeding.1,2
Diagnosing SEH is challenging due to its concealed hemorrhage and nonspecific clinical presentation, necessitating a high index of suspicion. 1The differential diagnosis includes prolonged or exaggerated neuraxial block, disc herniation, exacerbation or unmasking of underlying neurologic conditions, transient neurologic syndrome, epidural abscess, cauda equina syndrome, and anterior spinal artery syndrome.7,9
Several patient- and procedure-related factors can increase the risk of spinal epidural hematoma following neuraxial anesthesia. These include advanced age, underlying coagulopathies such as thrombocytopenia or platelet dysfunction, and the use of antithrombotic or anticoagulant medications.1,2,3 Pre-existing spinal pathologies, including vascular malformations or ankylosing spondylitis, may also predispose patients to this complication. Additional risk factors include extremes of body habitus, diabetes mellitus, and pregnancy.1 Technical challenges during spinal or epidural placement further elevate the risk.6,11
Common symptoms include progressive motor and sensory block, as well as bowel or bladder dysfunction, and localized back pain that may or may not have a radicular component.1,9 The return of sensory or motor deficits several hours after a spinal or epidural block has worn off (with or without back pain) is highly pathognomonic and should be treated as spinal or epidural hematoma until proven otherwise.1 The most common presenting sign is lower extremity weakness, which can be progressive and either unilateral or bilateral.2,9 Back pain is often amplified by percussion over the spine and by maneuvers that increase intraspinal pressure, such as coughing, straining, or sneezing (Table 1).1,9
Unlike the classic presentation of spinal epidural hematoma, which typically manifests within hours of neuraxial anesthesia or catheter removal, some cases demonstrate a delayed onset of symptoms, emerging several days or even weeks later.5,11 These atypical presentations may include a fluctuating pattern of motor weakness or paresthesia, altered spinal reflexes, and, notably, the absence of back pain.11 Additionally, symptoms may appear remote from the puncture site, as hematomas can develop at spinal levels distant from the site of neuraxial intervention, such as cervical or thoracic regions following a lumbar puncture.5,11 These features can obscure timely diagnosis and highlight the importance of maintaining a high index of suspicion in patients presenting with new or evolving neurological symptoms after neuraxial procedures (Table 1).1
An SEH typically presents within 24–48 hours after a procedure, making prompt evaluation critical for any new or worsening neurologic symptoms to rule out space-occupying lesions.4,6 If neurologic deficits occur during epidural analgesia, the infusion should be stopped while leaving the catheter in place to assess whether the anesthetic is the cause; motor and sensory function should return if so.4,8 When SEH is suspected, a thorough physical exam, complete blood count with platelets, and coagulation profile should be obtained.1,4,5 A stat MRI with and without contrast is essential to evaluate the hematoma’s extent, cord compression, and age.1,11 Additionally, neurology and neurosurgery consultations should be initiated promptly.4 Small, non-compressive hematomas may be managed conservatively with close monitoring, but urgent surgical decompression is indicated for hematomas causing neurologic deficits.5
Recovery from SEH depends on the severity and duration of neurologic deficits and the timing of surgery, with delays worsening the prognosis.1,4 Surgical intervention within 8 hours between the development of paralysis and surgical intervention increases the likelihood of complete neurologic recovery.1 Recovery can occur if surgery and decompression are performed within 36 hours of a complete motor deficit and within 48 hours of a partial deficit.4
In our case, the patient may have been at an elevated risk for hematoma formation due to preeclampsia, which can cause both quantitative and qualitative defects in platelet function. The patient's platelet levels remained stable above 100,000/μL over multiple days; however, qualitative defects may have been present due to preeclampsia. During the procedure, the patient exhibited signs of uterine atony. Although the quantitative blood loss was 485 ml, the patient may have experienced subclinical disseminated intravascular coagulation (DIC) during this period. In our scenario, the source of hemorrhage was an epidural artery rather than an epidural vein, given the extent of bleeding within the epidural space. The hemorrhage likely occurred after the SSS and progressively caused spinal cord compression.
During the SSS, the first attempt was slightly off midline, causing transient left buttock paresthesia that resolved upon needle removal. Initially, the patient had no issues with back pain, ambulation, motor deficits, or bowel or bladder dysfunction, and fluctuating paresthesia in the buttocks and left ankle/foot made the clinical picture ambiguous. However, once the patient began to exhibit motor deficits, the situation became clearer and more concerning, prompting expedited MRI imaging, neurosurgery consultation, and surgical decompression. A high degree of clinical suspicion, communication with a multidisciplinary team, and prompt management led to a favorable outcome.
Maintaining a high degree of clinical vigilance and a strong index of suspicion, particularly in cases of atypical SEH, is essential for timely diagnosis and effective management. Early identification of clinical signs and prompt therapeutic intervention are critical in optimizing patient outcomes and minimizing the risk of irreversible neurological deficits. Ongoing education and heightened awareness among healthcare professionals are imperative to mitigate the impact of this rare yet potentially devastating complication.
RR: conduct of study work
TD: literature search, manuscript editing
Authors affiliations:
- Nancy Boules, 1501 Violet St. Colton, CA 92324, USA; Email: nancy.boules@md.cusm.edu
- Ramin Rahimian, 101 The City Dr., S., Orange, CA, 92868, USA; Email: rrahimia@hs.uci.edu
- Taizoon Dhoon, 101 the city drive, Orange, CA, USA; Email: tdhoon@hs.uci.edu
ABSTRACT
Spinal epidural hematoma (SEH) is a rare complication of neuraxial anesthesia. We present a case of a 28-year-old G2P1001 at 28 weeks' gestation underwent an urgent cesarean delivery due to preterm premature rupture of membranes, pre-eclampsia with severe features, and breech presentation. The patient received a spinal anesthetic, which required two attempts. Postoperatively, she developed progressive lower extremity weakness and sensory changes within 48 hours. An MRI revealed a 14 × 10 × 82 mm SEH extending from L3 to S1, necessitating an emergent laminectomy that resulted in favorable outcomes.
We conclude that the anesthesiologists should remain vigilant for SEH, as early detection is vital for optimal maternal outcomes.
Abbreviations: NSVD: Normal Spontaneous Vaginal Delivery, PPROM: preterm premature rupture of membranes, SEH: spinal epidural hematoma, SSS: single shot spinal
Teaching Points:
- Be vigilant for delayed and fluctuating symptoms: SEH can present days to weeks after anesthesia with symptoms like motor weakness or paresthesia that may fluctuate, even without back pain.
- Prioritize early diagnosis with MRI: Any new or evolving neurological symptoms in high-risk patients warrant immediate consideration of SEH. MRI is the diagnostic tool of choice due to its accuracy; CT scans can miss early or small hematomas.
- Prompt surgical decompression is crucial: The best neurological outcomes are linked to early surgical intervention, especially in patients experiencing significant or worsening neurological deficits.
Citation: Boules N, Rahimian R, Dhoon T. Neurological emergency after cesarean section: a case of atypical spinal epidural hematoma. Anaesth. pain intensive care 2026;30(1):274-278. DOI: 10.35975/apic.v30i1.3138
Received: November 11, 2025; Revised: January 04, 2026; Accepted: January 06, 2026
1. INTRODUCTION
Spinal Epidural Hematoma (SEH) is a rare but severe complication of neuraxial anesthesia, occurring in approximately 1 in 200,000 obstetric cases.1 While SEH is typically associated with disorders like coagulopathy, recent anticoagulation, or thrombocytopenia, it can occur in low-risk patients as well.2,3 Accumulation of blood within the epidural space can exert significant pressure on the spinal cord and nerve roots, resulting in neurological perturbations such sensory and motor deficits, bowel and bladder dysfunction, and paralysis.2,3
We present the case of a 28-year-old woman, G2P1001, who developed a SEH following a single shot spinal (SSS) anesthetic. Progression of neurological symptoms 36 hours postpartum raised concerns for SEH. Emergent surgical decompression was performed following Magnetic Resonance Imaging (MRI) which demonstrated a massive hematoma extending from L3 to S1.
This report focuses on the early identification of neurological symptoms, workup, and management, including timely surgical decompression and hematoma evacuation.
2. CASE REPORT
A 28-year-old G2P1001 at 28 weeks and 1 day was scheduled for an urgent cesarean section due to preterm premature rupture of membranes (PPROM), pre-eclampsia with severe features consisting of elevated liver function tests and persistent headache, and breech presentation. Her past obstetric history includes one prior normal spontaneous vaginal delivery (NSVD) with labor epidural. At the time of surgery, the patient was receiving a magnesium sulfate infusion and was not on anticoagulation therapy. Her platelet count was 134,000/μL on the day prior to surgery and 144,000/μL on the morning of the procedure.
Spinal anesthesia was performed at the L3-L4 level after two attempts using a 25-gauge needle. The first attempt caused transient left-sided buttock paresthesia, which resolved immediately after needle withdrawal. A second attempt at neuraxial placement at the same interspace was successful and well tolerated, resulting in an effective sensory block up to the T4 dermatome. The cesarean delivery was complicated by uterine atony, which was managed with intravenous oxytocin (60 units infused over 60 minutes), intramuscular carboprost tromethamine (250 micrograms), intravenous tranexamic acid (1 gram), and placement of a modified B-Lynch suture to achieve uterine compression. Following these interventions, the total estimated blood loss was 465 mL. Postoperatively, magnesium sulfate infusion was continued for seizure prophylaxis.
On postoperative day (POD) 1, the patient experienced intermittent left sided buttock paresthesia and paresthesia of the left ankle. She was able to urinate and ambulate with assistance. On physical exam, the patient exhibited preserved bilateral plantarflexion and dorsiflexion, along with intact hip flexion and extension. Sensory examination revealed intact sensation to light touch and cold bilaterally, with preservation up to the T10 dermatomal level. No saddle anesthesia was noted, and there were no clinical signs of hematoma or infection at the lower back. The perineal numbness was initially thought to be related to residual effects of spinal anesthesia and left ankle paresthesia due to a nerve irritation from the first spinal attempt; both symptoms appeared to be resolving on POD 1.
On POD 2, the patient continued to experience persistent bilateral buttock paresthesias and reported new-onset perineal numbness. She also described worsening paresthesia and newly developed motor weakness in the left ankle and foot as well as left thigh and calf tightness. On physical exam, the patient exhibited diminished left-sided plantarflexion and dorsiflexion, reduced flexion and extension of the left toes, and intact hip flexion and extension. Sensory examination revealed intact sensation to light touch and cold bilaterally, with preservation up to the T10 dermatomal level. Urinary and bowel function remained at baseline.
An emergent MRI was obtained, and neurology and neurosurgical consultation was initiated. Complete blood count (CBC) and coagulation profiles were within normal limits, with a platelet count of 124,000/μL. MRI revealed a 14 mm x 10 mm x 82 mm epidural hematoma extending from L3 to S1, resulting in significant mass effect on the spinal canal and severe spinal canal stenosis, possibly reflecting cauda equina syndrome [Figure 1]. Neurosurgery was immediately consulted, and the patient underwent emergent L3-L5 laminectomy for evacuation of epidural hematoma, approximately 48 hours after the initial SSS. Preoperatively, the patient's complete blood count showed a white blood cell count of 9.9 ×10⁹/L, hemoglobin of 12.5 g/dL, hematocrit of 35.5%, and platelet count of 144,000/µL. Coagulation studies were not obtained.
At one-year follow-up, the patient's left leg paresthesia and motor deficits had fully resolved. However, she continued to experience intermittent right-sided perineal paresthesia, which she described as noticeable but not bothersome. Her bowel and bladder function remained stable and unchanged from baseline.

Figure 1: Sagittal MRI images of the lumbar spine showing a 14 x 10 x 82 mm heterogeneous enhancement of the dorsal aspect of the thecal sac extending from L3 to S1, with significant mass effect on thecal sac and compression on the nerve roots
3. DISCUSSION
SEH, though rare, can occur following spinal and epidural techniques if a blood vessel is punctured by a needle or catheter.6 SEH is associated with patients who have coagulation disorders or are on anticoagulation but can also occur in low-risk patients.2,3 The incidence of SEH is significantly lower in obstetric patients, with retrospective studies reporting an incidence between 0 and 0.6 per 100,000 epidural catheterizations.10 For spinal anesthetics, the incidence has been estimated to be less than 1 in 220,000 in the obstetric population.1 The veins, surrounded by loose areolar tissue, are prone to rupture, especially in pregnancy due to increased venous pressure from uterine enlargement and hemodynamic changes.1,2 While venous bleeding is widely accepted as the source of SEH, some debate persists over an arterial origin, as arterial pressure may be more capable of compressing the spinal cord than lower-pressure venous bleeding.1,2
Diagnosing SEH is challenging due to its concealed hemorrhage and nonspecific clinical presentation, necessitating a high index of suspicion. 1The differential diagnosis includes prolonged or exaggerated neuraxial block, disc herniation, exacerbation or unmasking of underlying neurologic conditions, transient neurologic syndrome, epidural abscess, cauda equina syndrome, and anterior spinal artery syndrome.7,9
Several patient- and procedure-related factors can increase the risk of spinal epidural hematoma following neuraxial anesthesia. These include advanced age, underlying coagulopathies such as thrombocytopenia or platelet dysfunction, and the use of antithrombotic or anticoagulant medications.1,2,3 Pre-existing spinal pathologies, including vascular malformations or ankylosing spondylitis, may also predispose patients to this complication. Additional risk factors include extremes of body habitus, diabetes mellitus, and pregnancy.1 Technical challenges during spinal or epidural placement further elevate the risk.6,11
Common symptoms include progressive motor and sensory block, as well as bowel or bladder dysfunction, and localized back pain that may or may not have a radicular component.1,9 The return of sensory or motor deficits several hours after a spinal or epidural block has worn off (with or without back pain) is highly pathognomonic and should be treated as spinal or epidural hematoma until proven otherwise.1 The most common presenting sign is lower extremity weakness, which can be progressive and either unilateral or bilateral.2,9 Back pain is often amplified by percussion over the spine and by maneuvers that increase intraspinal pressure, such as coughing, straining, or sneezing (Table 1).1,9
Unlike the classic presentation of spinal epidural hematoma, which typically manifests within hours of neuraxial anesthesia or catheter removal, some cases demonstrate a delayed onset of symptoms, emerging several days or even weeks later.5,11 These atypical presentations may include a fluctuating pattern of motor weakness or paresthesia, altered spinal reflexes, and, notably, the absence of back pain.11 Additionally, symptoms may appear remote from the puncture site, as hematomas can develop at spinal levels distant from the site of neuraxial intervention, such as cervical or thoracic regions following a lumbar puncture.5,11 These features can obscure timely diagnosis and highlight the importance of maintaining a high index of suspicion in patients presenting with new or evolving neurological symptoms after neuraxial procedures (Table 1).1
| Table 1: Overview of Spinal Epidural Hematoma presentation and management. | |
| Onset | Typically appears within 24–48 hours post-procedure |
| Risk Factors | · Advanced age · Underlying coagulopathy (including thrombocytopenia or platelet dysfunction) · Use of antithrombotic or anticoagulant medications · Pre-existing spinal pathology (such as vascular malformations or ankylosing spondylitis) · Extremes of body habitus (obesity) · Diabetes mellitus · Difficult spinal or epidural placement · Abnormal coagulation profile or thrombocytopenia · Pregnancy |
| Typical Presentation | · Return of blockade >1h after block resolution · Last epidural dose given >3h ago · Classic triad of acute back or radicular pain, sensory changes, and motor weakness · Focal neurological deficit (bilateral or unilateral) · Bowel or bladder dysfunction · Neurologic deficit in the absence of expected level of sympathetic blockade · Back pain aggravated by palpation of the procedural site or by Valsalva maneuver |
| Atypical Presentation | · Delayed onset of symptoms · Fluctuating pattern of motor weakness or paresthesia · Absence of back pain · Altered spinal reflexes · Symptoms remote from the puncture site |
| Management | · Stop epidural infusion · Neurochecks q15 min · Emergent Neurosurgery and Neurology consultation · Stat MRI spine · Stat CBC and coagulation panel · Reversal or coagulation or platelet dysfunction · Emergent surgical decompression, if needed |
An SEH typically presents within 24–48 hours after a procedure, making prompt evaluation critical for any new or worsening neurologic symptoms to rule out space-occupying lesions.4,6 If neurologic deficits occur during epidural analgesia, the infusion should be stopped while leaving the catheter in place to assess whether the anesthetic is the cause; motor and sensory function should return if so.4,8 When SEH is suspected, a thorough physical exam, complete blood count with platelets, and coagulation profile should be obtained.1,4,5 A stat MRI with and without contrast is essential to evaluate the hematoma’s extent, cord compression, and age.1,11 Additionally, neurology and neurosurgery consultations should be initiated promptly.4 Small, non-compressive hematomas may be managed conservatively with close monitoring, but urgent surgical decompression is indicated for hematomas causing neurologic deficits.5
Recovery from SEH depends on the severity and duration of neurologic deficits and the timing of surgery, with delays worsening the prognosis.1,4 Surgical intervention within 8 hours between the development of paralysis and surgical intervention increases the likelihood of complete neurologic recovery.1 Recovery can occur if surgery and decompression are performed within 36 hours of a complete motor deficit and within 48 hours of a partial deficit.4
In our case, the patient may have been at an elevated risk for hematoma formation due to preeclampsia, which can cause both quantitative and qualitative defects in platelet function. The patient's platelet levels remained stable above 100,000/μL over multiple days; however, qualitative defects may have been present due to preeclampsia. During the procedure, the patient exhibited signs of uterine atony. Although the quantitative blood loss was 485 ml, the patient may have experienced subclinical disseminated intravascular coagulation (DIC) during this period. In our scenario, the source of hemorrhage was an epidural artery rather than an epidural vein, given the extent of bleeding within the epidural space. The hemorrhage likely occurred after the SSS and progressively caused spinal cord compression.
During the SSS, the first attempt was slightly off midline, causing transient left buttock paresthesia that resolved upon needle removal. Initially, the patient had no issues with back pain, ambulation, motor deficits, or bowel or bladder dysfunction, and fluctuating paresthesia in the buttocks and left ankle/foot made the clinical picture ambiguous. However, once the patient began to exhibit motor deficits, the situation became clearer and more concerning, prompting expedited MRI imaging, neurosurgery consultation, and surgical decompression. A high degree of clinical suspicion, communication with a multidisciplinary team, and prompt management led to a favorable outcome.
4. CONCLUSION
Maintaining a high degree of clinical vigilance and a strong index of suspicion, particularly in cases of atypical SEH, is essential for timely diagnosis and effective management. Early identification of clinical signs and prompt therapeutic intervention are critical in optimizing patient outcomes and minimizing the risk of irreversible neurological deficits. Ongoing education and heightened awareness among healthcare professionals are imperative to mitigate the impact of this rare yet potentially devastating complication.
- Conflict of interest
- Funding
- Ethical considerations
- Authors’ contribution
RR: conduct of study work
TD: literature search, manuscript editing
9. REFERENCES
- Nelson A, Benzon HT, Jabri RS. Diagnosis and Management of Spinal and Peripheral Nerve Hematoma. NYSORA. Available from: https://www.nysora.com/topics/complications/diagnosis-management-spinal-peripheral-nerve-hematoma/. Accessed April 16, 2025.
- Yi S, Yoon DH, Kim KN, Kim SH, Shin HC. Postoperative spinal epidural hematoma: risk factor and clinical outcome. Yonsei Med J. 2006;47(3):326-32.[PubMed] DOI: 3349/ymj.2006.47.3.326
- Benzon HT, Jabri RS, Zundert T. Neuraxial Anesthesia & Peripheral Nerve Blocks in Patients on Anticoagulants. In: Hadzic A, editor. Hadzic's Textbook of Regional Anesthesia and Acute Pain Management. 2nd ed. McGraw-Hill Education; 2017.
- Lawton MT, Porter RW, Heiserman JE, Jacobowitz R, Sonntag VK, Dickman CA. Surgical management of spinal epidural hematoma: relationship between surgical timing and neurological outcome. J Neurosurg. 1995;83(1):1-7.[PubMed] DOI: 3171/jns.1995.83.1.0001
- Henry JB, Messerer M, Thomas V, Diabira S, Morandi X, Hamlat A. Nontraumatic spinal epidural hematoma during pregnancy: diagnosis and management concerns. Spinal Cord. 2012;50(9):655-60.[PubMed] DOI: 1038/sc.2012.48
- Coviello A, Iacovazzo C, Frigo MG, et al. Technical aspects of neuraxial analgesia during labor and maternity care: an updated overview. J Anesth Analg Crit Care. 2025;5:6.[PubMed] DOI: 1186/s44158-025-00224-3
- D'Angelo R, Smiley RM, Riley ET, Segal S. Serious complications related to obstetric anesthesia: the serious complication repository project of the Society for Obstetric Anesthesia and Perinatology. Anesthesiology. 2014;120(6):1505-12.[PubMed] DOI: 1097/ALN.0000000000000253
- Bateman BT, Mhyre JM, Ehrenfeld J, et al. The risk and outcomes of epidural hematomas after perioperative and obstetric epidural catheterization: a report from the Multicenter Perioperative Outcomes Group Research Consortium. Anesth Analg. 2013;116(6):1380-5.[PubMed] DOI: 1213/ANE.0b013e318251daed
- Palot M, Visseaux H, Botmans C, Pire JC. Epidemiology of complications of obstetrical epidural analgesia. Cah Anesthesiol. 1994;42(2):229-33.[PubMed]
- Rosero EB, Joshi GP. Nationwide incidence of serious complications of epidural analgesia in the United States. Acta Anaesthesiol Scand. 2016;60(6):810-20.[PubMed] DOI: 1111/aas.12702
- Makris A, Gkliatis E, Diakomi M, Karmaniolou I, Mela A. Delayed spinal epidural hematoma following spinal anesthesia, far from needle puncture site. Spinal Cord. 2014;52(Suppl 1):S14-6.[PubMed] DOI: 1038/sc.2013.174