Doliane Mambu Nsambu Mbooto ¹ , Eric Amisi ¹ , Arriel Makembi Bunkete ²* , Gibency Mfulani ¹ , Wilfrid Mbombo ¹ , Kazi Anga Muamba ¹ , Selenge Mauwa Stéphanie ¹ , Muadi Kalenga Monique 1 , Ntambwe Mulembi Grâce ¹ , Marthe Panzu ¹ , Arsène Mulopo ⁵ , Tacite Mazoba Kpanya ⁴ , Kankonde Kalambay Dan ¹ , Kobo Patrick ¹ , Tsangu Phuati Joseph ¹ , Mavinga Nyombo José ¹ , Patrick Mukuna ¹ , Antoine Molua Aundu ⁴ , Kashongwe Murhula Innocent ³ , Médard Bulabula ¹ , Berthe Barhayiga ¹
Authors affiliations:
Background & objectives: Pulmonary ultrasound is an interesting tool for the etiological diagnosis of acute respiratory failure (ARF), but its impact has been little studied. We evaluated the impact of implementing Bedside Lung Ultrasound in Emergency (BLUE)-protocol on diagnosis and etiological treatment times, the proportion of imaging procedures performed, and mortality.
Methodology: A quasi-experimental ‘before–after’ study was conducted in the Emergency and Intensive Care Departments of the University Clinics of Kinshasa from April 1, 2023, to August 30, 2024. Pre-implementation phase entailed the prospective collection of data on patients admitted with acute respiratory failure (ARF). Data from 151 patients (99 before and 52 after the implementation of the BLUE protocol) were analyzed. Student’s t, Mann–Whitney, chi-squared, and Fisher’s exact tests were used, as well as logistic regression to identify factors associated with mortality (P < 0.05).
Results: The two groups were comparable in baseline characteristics. The time to etiological diagnosis ≤1 day increased significantly from 62.7% to 80.3% (P = 0.033). The time to etiological treatment ≤1 day also improved from 53.5% to 80.3% (P = 0.003). The use of chest X-rays decreased (48.5% vs. 9.6%, P = 0.01), while mortality remained unchanged (52.5% vs. 57.7%, P = 0.545).
Conclusion: Implementation of the BLUE protocol shortened diagnosis and treatment times and reduced ionizing imaging but did not significantly affect mortality.
Keywords: acute respiratory failure; BLUE protocol; lung ultrasound; mortality; quasi-experimental study.
Citation: Mbooto DMN, Amisi E, Bunkete AM, Mfulani G, Mbombo W, Muamba KA. et al. Impact of the implementation of the BLUE protocol on mortality in patients with acute respiratory failure: our experience at the University Clinics of Kinshasa. Anaesth. pain intensive care 2025;30(2):165-174. DOI: 10.35975/apic.v30i2.3122
Received: October 13, 2025; Revised: November 28, 2025; Accepted: November 29, 2025
Acute Respiratory Failure (ARF) is the sudden inability of the respiratory system to to ensure satisfactory haematosis.1 It is a major health problem. It accounts for 4% of hospital admissions and 10.4% of intensive care unit admissions, with mortality ranging from 35 to 40%.2 ARF management requires imaging examinations, which are often not feasible in emergency situations, or are unreliable. For several decades now, bedside lung ultrasound,3 and more specifically the BLUE protocol, has been a valuable tool in the etiological diagnosis of ARF in emergency and intensive care settings. Several studies have been carried out on its diagnostic performance.4,5,6 But very few studies been carried out on its impact on the management of AKI.3
A variety of conditions may be responsible for ARF.7 The challenge in the emergency management of ARF is not only to compensate for respiratory failure, but above all to establish a precise etiological diagnosis at an early stage.4,8 However, the imaging procedures necessary for this diagnostic process are not always feasible in emergency settings due to the potential risks associated with moving unstable patients.5,9 Chest CT and Angioscan are the preferred diagnostic modalities for investigating the underlying causes of ARF; however, these procedures often necessitate patient movement. Chest X-rays, on the other hand, can be performed at the patient's bedside, but their diagnostic value is limited in the investigation of ARF.5,9 Furthermore, the use of CT scans and chest X-rays carries the potential for patients to be exposed to ionizing radiation, a known health hazard.
Lung ultrasound was introduced late because air was seen as an obstacle to ultrasound. However, over the past two decades, lung ultrasound has become an indispensable diagnostic tool for emergency and intensive care patients. Following an examination of the ultrasound semiology of the lung, numerous authors have conducted studies on the diagnostic performance of lung ultrasound. Simplified lung ultrasound protocols have also been described with the aim of facilitating practice by clinicians at the point of care. A notable example of such a protocol is the BLUE protocol. This protocol involves the utilization of lung ultrasound in conjunction with the ultrasound of the veins in the lower limbs. The BLUE protocol is notable for its utilization of a streamlined algorithm, which enables comprehensive review of most potential etiological diagnoses of ARF. While the performance of this protocol in the etiological diagnosis of ARF has been the subject of extensive research, notably by Kankonde et al., the question of its impact on the management of ARF remains to be answered. Nevertheless, lung ultrasound, particularly the BLUE protocol, has considerable potential as a valuable tool for the etiological diagnosis of ARF in low-resource settings.
The general objective of the present study is to evaluate the impact of the implementation of the BLUE protocol on the time to etiological management, the frequency of ionizing imaging examinations and the mortality of patients admitted for acute respiratory failure to the emergency and intensive care units of the Clinique Universitaire de Kinshasa (CUK).
The objective of this quasi-experimental study was to assess whether implementing the BLUE protocol would reduce time to diagnosis and treatment, decrease use of ionizing imaging, and affect mortality among patients admitted for ARF.
We conducted a ‘before-and-after’ quasi-experimental study in the Emergency and Intensive Care Departments of the University Clinics of Kinshasa during the period from April 2023 to August 2024.
This pre-implementation phase occurred from April 1 to December 31, 2023. This phase entailed the prospective collection of data on patients admitted with ARF. The intervention consisted in implementing the BLUE protocol. This phase, which occurred from June to December 2023, encompassed several key activities, including physician training, the creation of a digital forum, protocol development and dissemination, and guided practice in lung ultrasound. The post-implementation phase ensued from January 1 to August 31, 2024. In addition to the prospective data collection, all patients selected during this phase underwent lung ultrasound according to the BLUE protocol.
The study population consisted of all patients with ARF admitted to CUK emergency departments or intensive care units. Patient recruitment was exhaustive and prospective. Patients meeting the selection criteria were included: patients with ARF, be at least 18 years old and admitted to CUK emergency and intensive care units during the periods corresponding to the two phases of the present study. The study was conducted in two phases, separated by implementation of the BLUE protocol. The implementation entailed the provision of theoretical and practical training to clinicians in the theoretical principles of US and the practice of lung ultrasound (LUS).
A 3-day seminar was organized by the Department of Anesthesia and Intensive Care in collaboration with the Department of Medical Imaging. The theoretical component of the seminar, delivered jointly by a medical imaging specialist and two anesthesia and intensive care specialists with at least one holding an ultrasound certificate, covered the basics of ultrasound, the role of ultrasound in intensive and emergency care, and LUS.
A practical workshop followed, led by an anesthesiologist with a LUS qualification. This practical training continued throughout the implementation of the BLUE protocol. The BLUE protocol was then discussed and critiqued by the attending clinicians, who presented lung ultrasound scans performed in their daily practice.
The data for this study were collected during the pre- and post-implementation phases of the BLUE protocol. LUS scans were not mandatory in the pre−implementation phase but were performed for all selected patients in the post−implementation phase. LUS scans were performed using a handheld ultrasound device consisting of a Philips-brand lumify phased array probe and a Galaxy tab S6 tablet, which held the lumify application.
The ultrasound examination of the lung was performed in the supine position for the most bedridden patients, and in the semi-seated position for the least bedridden. The thorax was divided into anterior, lateral and posterior regions. Each region was then subdivided into upper and lower quadrants, resulting in a total of six quadrants per lung. Longitudinal sections were preferred, to utilize the shadow cones of the upper and lower ribs as landmarks. The BLUE protocol was used to determine the etiological diagnosis of ARF.
In instances where patients presented with ARF accompanied by an A profile on pulmonary ultrasound, the examination was completed by a 4-point Doppler ultrasound of the lower limbs. The purpose of this Doppler ultrasound of the limbs was to look for venous thrombosis in the femoral and popliteal veins bilaterally. The presence of visible walls, hyperechoic contents, and absent or inadequate Doppler flow was indicative of venous thrombosis. This additional information was then used to either retain or exclude a diagnosis of pulmonary embolism.
The BLUE protocol algorithm was utilized to establish the etiological diagnosis of ARF (Figure 1).
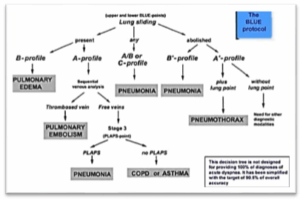
Figure 1: BLUE algorithm protocol
The following data were collected: sociodemographic data (age, sex, profession); clinical and therapeutic data (date of admission, time of admission, comorbidities, type and number of organ failures, etiological diagnosis, time of etiological diagnosis, type of imaging performed for etiological diagnosis, etiological treatment and suppletive treatment); and evolutionary data (complications, survival to D28 and length of hospital stay).
The main criterion for judgment was the etiological treatment time. Secondary criteria included the time to etiological diagnosis, reduction in the frequency of ionizing imaging examinations requested (Rx thorax and CT scan) and mortality at D28.
2.1. Operational definitions
The data collected underwent daily quality control under the responsibility of the investigator. The data collected, in particular the LUS, underwent daily quality control under the responsibility of the supervisor. This control consisted in checking the accuracy of the observations and the completeness of the forms. The data collection forms were received and subjected to a second quality control, before being encoded.
All data were collected and entered an Excel file, transferred and then analyzed using SPSS 27.0 software. The descriptive analysis performed using mean and standard deviation for quantitative data with a Gaussian distribution, median with interquartile range (IQR) for data with a non-Gaussian distribution, and relative (%) and absolute (n) proportions for categorical or qualitative data.
Pearson's Chi-squared test or Ficher's exact test was used to compare percentages. Student's t-test was used to compare means. The Mann Whitney test was used to compare medians. The Kaplan-Méier method was used to estimate patient mortality on D28 of hospitalization. The log rank test was used to compare curves. Factors associated with mortality in patients admitted for ARF were investigated using logistic regression, with calculation of Odds ratios and their 95% confidence intervals to determine the degree of risk.
For all tests performed, the threshold of statistical significance was P < 0.05.
A total of 180 patients were admitted with ARF during the two survey periods, 128 before and 52 after implementation of the BLUE protocol. Of the pre-implementation patients, 29 were excluded (Figure 2). The present study included 151 patients, 99 before and 52 after implementation.
Figure 2: Kaplan Meier curve of patient survival to 28th day of hospitalization, according to the time of implementation
Table 1 shows the general profile of the patients. It shows that 80 of the 151 patients in this study were female (53%) and 71 were male (47%). There were no gender differences between the two groups (P > 0.05). The mean age of patients was 52.3 ± 20.7 years and was comparable between the two groups (P > 0.05). Most patients had no professional activity, and this was true for both groups (P = 0.073). Most patients had one (45.7%), two (41.1%) or three (11.9%) failures. In addition to respiratory failure, circulatory failure (52 out of 151, or 34.4%) and neurological failure (36 out of 151, or 23.8%) were more common. There was no difference in the number (P = 0.675) or type (P = 0.273) of failures between the two groups.
Pulmonary embolism and pleuropneumonia were the most frequent etiological diagnoses, accounting for 21.9% and 11.3% of cases, respectively. The difference in etiologic diagnoses between the two groups was not statistically significant (P > 0.05). Conventional oxygen therapy (79.5%) was used most frequently for respiratory support. The interfaces used were nasal cannula (37.7%), single mask (6%) and high concentration mask (HCM) (31.8%). Of 151 patients admitted with ARF, 38 (25.2%) received non-invasive ventilation (NIV) and 34 (22.5%) invasive mechanical ventilation. More patients received NIV (P < 0.003) and invasive mechanical ventilation (P = 0.001) after implementation of the BLUE protocol than before.
Table 2 shows the time to etiological diagnosis, time to etiological treatment, mortality at day 28 and hospital stay for the 151 patients admitted with ARF. The time to etiological diagnosis of ARF was one day or less in 69.5% of cases, and more than one day in 30.5% of cases. Significantly more patients in the post-implementation group had their etiological diagnosis within one day of admission than in the pre-implementation group (80.3% vs. 62.7% P = 0.033).
The time to etiological treatment of acute respiratory failure was one day or less in 62.9% of cases, and more than one day in 37.5% of cases. Significantly more patients in the post-implementation group received etiological treatment within one day of admission than in the pre-implementation group (80.3% vs. 53.5% P = 0.003)
Hospital stays for patients admitted with acute respiratory failure was less than one week in 47.7% of cases, and more than one day in 52.3% of cases. Significantly more patients stayed in hospital for less than one week after implementation than before (55.8% vs. 43.4% P = 0.003).
Mortality among patients admitted for acute respiratory failure was 54.3%. It was 52.5% before and 57.7% after implementation. There was no significant difference in mortality between the pre- and post-implementation groups (P = 0.545).
Table 3 shows the imaging examinations carried out to investigate the etiology of acute respiratory failure. In descending order, the imaging examinations carried out for etiological research are pulmonary ultrasound (39.1%), chest x-ray (35.1%), cardiac ultrasound (22.5%), thoracic angioscanner (11.9%) and Doppler ultrasound of the IM Vx (4.1%).
A comparison of the number of imaging examinations performed shows a significant reduction in the number of chest X-rays performed after implementation of the BLUE protocol (48.5% vs. 9.6% P = 0.01), but a non-significant reduction in the number of thoracic angioscanner (11.1% vs. 9.6% P = 0.829), cardiac ultrasound (24.2% vs. 19.2% P = 0.642) and MI Doppler ultrasound (5.1% vs. 3.8%). It's worth noting that seven out of 99 patients (7.1%) had undergone lung ultrasound before implementation compared to 100% of patients after implementation.
Figure 3 shows the survival curves for patients enrolled before and after implementation of the BLUE protocol. It shows that survival was comparable between the pre- and post-implementation groups.
Table 4 shows the factors associated with mortality in patients admitted for acute respiratory failure. The table shows that 4 factors are associated with mortality in patients admitted for acute respiratory failure: age over 55 years, the patient not working and the presence of more than one organ failure.
ARF is defined as hypoxemia with or without hypercapnia. It is a life-threatening emergency.11-14 Early recognition is essential for optimal diagnostic and therapeutic management. The initiation of appropriate etiological treatment is closely linked to the accuracy of the etiological diagnosis. Advances in clinical, radiologic and scannographic means of lung examination have made it possible to refine the etiological investigation of ARF.15-17
X-rays are an age-old tool that can be used at the patient's bedside and are highly accessible. However, it has a number of troublesome shortcomings in life-threatening emergencies.16,18,19 CT scanning is more informative and fills in the gaps left by radiography. However, it requires patients to be moved, even if they are at risk.16,6,20,21
In the context of limited resources, in addition to the above-mentioned disadvantages, radiography and CT scans face the thorny problem of patients' financial accessibility to these investigations, which are nevertheless useful for diagnosing acute respiratory failure. Hence the interest in finding an etiological investigation method capable of solving the main problem of acute medicine: to act quickly and effectively. Ultrasound offers a fast and easy alternative. Thanks to technological advances, new ultrasound scanners allow physicians to obtain sharper images at the point of care (POC). The heart was the first example of ultrasound exploration by the clinician at the POC.22,23
Ultrasound was subsequently introduced into general emergency practice, particularly in traumatology.24-27 However, LUS was a latecomer in its development, as air was considered an ultrasound obstacle. However, over the last few decades, and following the development of ultrasound lung semiology, LUS has experienced a remarkable boom. Several studies have been carried out on the diagnostic performance of LUS,9,28-30 and simplified protocols for the clinical use of LUS have been proposed.31,32
Our study is one of the few to investigate the impact of LUS on the management of ARF.33,34 It showed that implementation of the BLUE protocol reduced the time to etiological diagnosis, the time to initiation of etiological treatment, and the number of imaging examinations, especially RT and ST, performed. However, it did not change the mortality rate of patients admitted for ARF. This is a landmark study of LUS, specifically the BLUE protocol, in emergency and intensive care units in the Democratic Republic of Congo, a low-resource country.
Our study showed that the proportion of patients admitted with ARF whose etiological diagnosis was established within one day of admission increased significantly after implementation of the BLUE protocol. It increased from 62.7% before to 80.3% after implementation of the BLUE protocol P = 0.033.
This study also showed a significant increase in the proportion of ARF patients receiving appropriate etiological treatment within 1 day of admission. We went from 53.5% receiving etiological treatment on day one before to 80.3% after implementation of the BLUE protocol P = 0.003.
Seyedhosseini et al.36 conducted a randomized study of the impact of the BLUE protocol on the time to treatment of acute respiratory distress in the emergency department. This study found a median time from admission to treatment of 17 minutes for the BLUE group, compared with 38 minutes for the control group. The difference between the two groups was statistically significant (P = 0.0001).
Our two studies show that the use of LUS, specifically the BLUE protocol, reduces the time taken to manage patients admitted to the emergency department and intensive care unit.
Volpicelli G et al. (2006) previously reported that bedside ultrasound reduced costs and saved time in the management of acute respiratory distress in the intensive care unit.35 Ünlüer and Karagöz compared lung ultrasound and radiography in the management of pneumonia. They showed that ultrasound was faster and more accurate in diagnosing pneumonia.36
Our study and that of Seyedhosseini et al. (2017) as well as several previously published studies, show that we can save time in the management of ARF by using bedside LUS. However, several methodological differences between our two-studies merit analysis. They may explain why our delay in initiating treatment is even longer than that of Seyedhosseini et al.
In fact, Seyedhisseini's study was randomized. It included patients admitted to the emergency department. The control group and the study group (BLUE protocol) received conventional imaging for the etiological investigation of acute respiratory failure. The BLUE Protocol group received LUS in addition to conventional imaging. Conventional imaging verified the accuracy of ultrasound diagnosis in the BLUE protocol group, while LUS enabled initiation of etiological treatment.
In contrast, our study was a quasi−experimental before-after study. It involved patients admitted to emergency departments and intensive care units (ICU). All patients could have benefited from conventional imaging and even LUS, depending on affordability, patient condition and availability of the technique. However, all patients admitted after the implementation of the BLUE protocol had received LUS.
The facilitators trained in LUS were used to verify the accuracy of the diagnosis.
All patients admitted with ARF after implementation benefited from LUS, whereas this examination was only performed in 7.1% of cases prior to implementation. Comparing the number of other imaging examinations performed, we recorded a significant reduction in the number of chest X-rays performed after implementation of the BLUE protocol (48.5% vs. 9.6% P = 0.01). However, the numbers of thoracic angioscanner (11.1% vs. 9.6% P = 0.829), cardiac ultrasound (24.2% vs. 19.2% P = 0.642) and lower limbs Doppler ultrasound (5.1% vs. 3.8%) were almost identical.
A retrospective study by Cornesse et al.37 found a significant decrease in chest X-rays and CT scans per ICU stay after lung ultrasound implementation, recording 10.3 ± 12 RTs before vs. 7.7 ± 10 after (P = 0.005) and 0.5 ± 1 ST before vs. 0.4 ± 1 after (P = 0.01).
Our study and that of Cornesse et al. show that the introduction of LUS reduces the number of chest X-rays performed to investigate the etiology of ARF. In our study, the number of STs was almost identical before and after implementation. This difference can be explained by the fact that thoracic CT scans, although the reference imaging technique, are rarely performed in our context, due to several obstacles, especially financial.
However, it should be noted that reducing the number of RTs and STs reduces exposure to ionizing radiation and health care costs.
This study shows that the mortality of patients admitted with ARF was 52.5% before implementation and 57.7% after, with no significant difference (P = 0.545).
Seyedhosseini et al. (2017) observed a mortality of 4.2% in the BLUE group versus 13.6% in the control group. This difference was not statistically significant (P-value: 0.34). Cornesse et al found that mortality was identical before and after implementation. Mortality was 22% and 21% respectively before and after implementation of lung ultrasound (P = 0.75).
A comparison of our study with the two studies leads to the following two observations:
The aim of this study was to evaluate the impact of implementing the Blue Protocol on the time to etiological management, the frequency of ionizing imaging examinations and the mortality of patients admitted with ARF to the Emergency and Intensive Care Units of the Clinique Universitaire de Kinshasa.
At the end of this study, we can conclude that lung ultrasound offers many advantages in the management of patients with ARF in acute care units, by reducing delays in etiological management, initiating targeted therapy, reducing ionizing examinations performed and not influencing mortality at D28 without significant difference.
However, further research is still needed to better understand the impact of ultrasound on decision-making regarding the management of patients with ARF in acute care units.
7. Ethical considerations
The protocol was approved by the Department of Anaesthesia and Intensive Care, and also by the Ethics Committee of the School of Public Health, under number ESP/CE/131/2023. Informed consent was obtained from conscious patients, and through an intermediary for patients unable to give it, in accordance with the principles of the Helsinki Convention. We have no conflict of interest in this work.
8. Competing interests
The authors declare that there was no competing interest
9. Funding
This study was completed by using institutional resources, and no external of industry funding was received .No funding received.
10. Authors' contributions
All authors took part in the study conception and design, Data collection, literature review, manuscript drafting, manuscript editing. Statistical analysis, interpretation of results, manuscript review And / or supervision,
Authors affiliations:
- Department of Anesthesia & Intensive Care, Kinshasa University Clinics, University of Kinshasa, Kinshasa, Democratic Republic of the Congo.
- Nephrology Service, Department of Internal Medicine, Kinshasa University Clinics, University of Kinshasa, Kinshasa, Democratic Republic of the Congo.
- Pulmonology Service, Department of Internal Medicine, Kinshasa University Clinics, University of Kinshasa, Kinshasa, Democratic Republic of the Congo.
- Medical Imaging Department, Kinshasa University Clinics, University of Kinshasa, Kinshasa, Democratic Republic of the Congo.
- FHI 360, Kinshasa, Democratic Republic of the Congo.
ABSTRACT
Background & objectives: Pulmonary ultrasound is an interesting tool for the etiological diagnosis of acute respiratory failure (ARF), but its impact has been little studied. We evaluated the impact of implementing Bedside Lung Ultrasound in Emergency (BLUE)-protocol on diagnosis and etiological treatment times, the proportion of imaging procedures performed, and mortality.
Methodology: A quasi-experimental ‘before–after’ study was conducted in the Emergency and Intensive Care Departments of the University Clinics of Kinshasa from April 1, 2023, to August 30, 2024. Pre-implementation phase entailed the prospective collection of data on patients admitted with acute respiratory failure (ARF). Data from 151 patients (99 before and 52 after the implementation of the BLUE protocol) were analyzed. Student’s t, Mann–Whitney, chi-squared, and Fisher’s exact tests were used, as well as logistic regression to identify factors associated with mortality (P < 0.05).
Results: The two groups were comparable in baseline characteristics. The time to etiological diagnosis ≤1 day increased significantly from 62.7% to 80.3% (P = 0.033). The time to etiological treatment ≤1 day also improved from 53.5% to 80.3% (P = 0.003). The use of chest X-rays decreased (48.5% vs. 9.6%, P = 0.01), while mortality remained unchanged (52.5% vs. 57.7%, P = 0.545).
Conclusion: Implementation of the BLUE protocol shortened diagnosis and treatment times and reduced ionizing imaging but did not significantly affect mortality.
Keywords: acute respiratory failure; BLUE protocol; lung ultrasound; mortality; quasi-experimental study.
Citation: Mbooto DMN, Amisi E, Bunkete AM, Mfulani G, Mbombo W, Muamba KA. et al. Impact of the implementation of the BLUE protocol on mortality in patients with acute respiratory failure: our experience at the University Clinics of Kinshasa. Anaesth. pain intensive care 2025;30(2):165-174. DOI: 10.35975/apic.v30i2.3122
Received: October 13, 2025; Revised: November 28, 2025; Accepted: November 29, 2025
1. INTRODUCTION
Acute Respiratory Failure (ARF) is the sudden inability of the respiratory system to to ensure satisfactory haematosis.1 It is a major health problem. It accounts for 4% of hospital admissions and 10.4% of intensive care unit admissions, with mortality ranging from 35 to 40%.2 ARF management requires imaging examinations, which are often not feasible in emergency situations, or are unreliable. For several decades now, bedside lung ultrasound,3 and more specifically the BLUE protocol, has been a valuable tool in the etiological diagnosis of ARF in emergency and intensive care settings. Several studies have been carried out on its diagnostic performance.4,5,6 But very few studies been carried out on its impact on the management of AKI.3
A variety of conditions may be responsible for ARF.7 The challenge in the emergency management of ARF is not only to compensate for respiratory failure, but above all to establish a precise etiological diagnosis at an early stage.4,8 However, the imaging procedures necessary for this diagnostic process are not always feasible in emergency settings due to the potential risks associated with moving unstable patients.5,9 Chest CT and Angioscan are the preferred diagnostic modalities for investigating the underlying causes of ARF; however, these procedures often necessitate patient movement. Chest X-rays, on the other hand, can be performed at the patient's bedside, but their diagnostic value is limited in the investigation of ARF.5,9 Furthermore, the use of CT scans and chest X-rays carries the potential for patients to be exposed to ionizing radiation, a known health hazard.
Lung ultrasound was introduced late because air was seen as an obstacle to ultrasound. However, over the past two decades, lung ultrasound has become an indispensable diagnostic tool for emergency and intensive care patients. Following an examination of the ultrasound semiology of the lung, numerous authors have conducted studies on the diagnostic performance of lung ultrasound. Simplified lung ultrasound protocols have also been described with the aim of facilitating practice by clinicians at the point of care. A notable example of such a protocol is the BLUE protocol. This protocol involves the utilization of lung ultrasound in conjunction with the ultrasound of the veins in the lower limbs. The BLUE protocol is notable for its utilization of a streamlined algorithm, which enables comprehensive review of most potential etiological diagnoses of ARF. While the performance of this protocol in the etiological diagnosis of ARF has been the subject of extensive research, notably by Kankonde et al., the question of its impact on the management of ARF remains to be answered. Nevertheless, lung ultrasound, particularly the BLUE protocol, has considerable potential as a valuable tool for the etiological diagnosis of ARF in low-resource settings.
The general objective of the present study is to evaluate the impact of the implementation of the BLUE protocol on the time to etiological management, the frequency of ionizing imaging examinations and the mortality of patients admitted for acute respiratory failure to the emergency and intensive care units of the Clinique Universitaire de Kinshasa (CUK).
The objective of this quasi-experimental study was to assess whether implementing the BLUE protocol would reduce time to diagnosis and treatment, decrease use of ionizing imaging, and affect mortality among patients admitted for ARF.
2. METHODOLOGY
We conducted a ‘before-and-after’ quasi-experimental study in the Emergency and Intensive Care Departments of the University Clinics of Kinshasa during the period from April 2023 to August 2024.
This pre-implementation phase occurred from April 1 to December 31, 2023. This phase entailed the prospective collection of data on patients admitted with ARF. The intervention consisted in implementing the BLUE protocol. This phase, which occurred from June to December 2023, encompassed several key activities, including physician training, the creation of a digital forum, protocol development and dissemination, and guided practice in lung ultrasound. The post-implementation phase ensued from January 1 to August 31, 2024. In addition to the prospective data collection, all patients selected during this phase underwent lung ultrasound according to the BLUE protocol.
The study population consisted of all patients with ARF admitted to CUK emergency departments or intensive care units. Patient recruitment was exhaustive and prospective. Patients meeting the selection criteria were included: patients with ARF, be at least 18 years old and admitted to CUK emergency and intensive care units during the periods corresponding to the two phases of the present study. The study was conducted in two phases, separated by implementation of the BLUE protocol. The implementation entailed the provision of theoretical and practical training to clinicians in the theoretical principles of US and the practice of lung ultrasound (LUS).
A 3-day seminar was organized by the Department of Anesthesia and Intensive Care in collaboration with the Department of Medical Imaging. The theoretical component of the seminar, delivered jointly by a medical imaging specialist and two anesthesia and intensive care specialists with at least one holding an ultrasound certificate, covered the basics of ultrasound, the role of ultrasound in intensive and emergency care, and LUS.
A practical workshop followed, led by an anesthesiologist with a LUS qualification. This practical training continued throughout the implementation of the BLUE protocol. The BLUE protocol was then discussed and critiqued by the attending clinicians, who presented lung ultrasound scans performed in their daily practice.
The data for this study were collected during the pre- and post-implementation phases of the BLUE protocol. LUS scans were not mandatory in the pre−implementation phase but were performed for all selected patients in the post−implementation phase. LUS scans were performed using a handheld ultrasound device consisting of a Philips-brand lumify phased array probe and a Galaxy tab S6 tablet, which held the lumify application.
The ultrasound examination of the lung was performed in the supine position for the most bedridden patients, and in the semi-seated position for the least bedridden. The thorax was divided into anterior, lateral and posterior regions. Each region was then subdivided into upper and lower quadrants, resulting in a total of six quadrants per lung. Longitudinal sections were preferred, to utilize the shadow cones of the upper and lower ribs as landmarks. The BLUE protocol was used to determine the etiological diagnosis of ARF.
In instances where patients presented with ARF accompanied by an A profile on pulmonary ultrasound, the examination was completed by a 4-point Doppler ultrasound of the lower limbs. The purpose of this Doppler ultrasound of the limbs was to look for venous thrombosis in the femoral and popliteal veins bilaterally. The presence of visible walls, hyperechoic contents, and absent or inadequate Doppler flow was indicative of venous thrombosis. This additional information was then used to either retain or exclude a diagnosis of pulmonary embolism.
The BLUE protocol algorithm was utilized to establish the etiological diagnosis of ARF (Figure 1).

Figure 1: BLUE algorithm protocol
The following data were collected: sociodemographic data (age, sex, profession); clinical and therapeutic data (date of admission, time of admission, comorbidities, type and number of organ failures, etiological diagnosis, time of etiological diagnosis, type of imaging performed for etiological diagnosis, etiological treatment and suppletive treatment); and evolutionary data (complications, survival to D28 and length of hospital stay).
The main criterion for judgment was the etiological treatment time. Secondary criteria included the time to etiological diagnosis, reduction in the frequency of ionizing imaging examinations requested (Rx thorax and CT scan) and mortality at D28.
2.1. Operational definitions
- Clinical respiratory failure was defined as hypoxemia (SpO2 less than 90% and/or PaO2 less than 60 mm Hg) associated or not with hypercapnia (Pco2) > 45 mm Hg).
- Time to etiological treatment was defined as the time between admission and the start of etiological eatment,
- Time to etiological diagnosis is defined as the time between the time of admission and the time when the etiological diagnosis is made.
The data collected underwent daily quality control under the responsibility of the investigator. The data collected, in particular the LUS, underwent daily quality control under the responsibility of the supervisor. This control consisted in checking the accuracy of the observations and the completeness of the forms. The data collection forms were received and subjected to a second quality control, before being encoded.
All data were collected and entered an Excel file, transferred and then analyzed using SPSS 27.0 software. The descriptive analysis performed using mean and standard deviation for quantitative data with a Gaussian distribution, median with interquartile range (IQR) for data with a non-Gaussian distribution, and relative (%) and absolute (n) proportions for categorical or qualitative data.
Pearson's Chi-squared test or Ficher's exact test was used to compare percentages. Student's t-test was used to compare means. The Mann Whitney test was used to compare medians. The Kaplan-Méier method was used to estimate patient mortality on D28 of hospitalization. The log rank test was used to compare curves. Factors associated with mortality in patients admitted for ARF were investigated using logistic regression, with calculation of Odds ratios and their 95% confidence intervals to determine the degree of risk.
For all tests performed, the threshold of statistical significance was P < 0.05.
3. RESULTS
A total of 180 patients were admitted with ARF during the two survey periods, 128 before and 52 after implementation of the BLUE protocol. Of the pre-implementation patients, 29 were excluded (Figure 2). The present study included 151 patients, 99 before and 52 after implementation.

Figure 2: Kaplan Meier curve of patient survival to 28th day of hospitalization, according to the time of implementation
Table 1 shows the general profile of the patients. It shows that 80 of the 151 patients in this study were female (53%) and 71 were male (47%). There were no gender differences between the two groups (P > 0.05). The mean age of patients was 52.3 ± 20.7 years and was comparable between the two groups (P > 0.05). Most patients had no professional activity, and this was true for both groups (P = 0.073). Most patients had one (45.7%), two (41.1%) or three (11.9%) failures. In addition to respiratory failure, circulatory failure (52 out of 151, or 34.4%) and neurological failure (36 out of 151, or 23.8%) were more common. There was no difference in the number (P = 0.675) or type (P = 0.273) of failures between the two groups.
| Table 1: Demographic profile of the patients | ||||
| Variables | All
(n = 151) |
Time of inclusion | P-value | |
| Before (n = 99) | After (n = )52 | |||
| Gender. n (%)
Female Male Age. year Mean ± SD Range Occupation. n (%) In occupation No occupation Number of failures One Two Three Four Type of failure Circulatory Neurological Other Diagnosis Pulmonary embolism Ac pulmonary edema Pleuropneumonia Pneumonia ARDS Sepsis Tuberculosis Other Oxygen interface Nasal cannula Single mask HCM MV Non-Invasive ventilation |
80 (53) 71 (47) 52.3 ± 20.7 18-94 26 (17.2) 125 (82.8) 69 (45.7) 62 (41.1) 18 (11.9) 2 (1.3) 52 (34.4) 36 (23.8) 13 (8.6) 33 (21.9) 12 (7.9) 6 (4) 11 (7.3) 3 (2) 27 (17.9) 8 (5.3) 8 (5.3) 57 (37.7) 9 (6) 48 (31.8) 34 (22.5) 38 (25.2) |
50 (50.5) 49 (49.5) 52.9 ± 20.3 - 21 (21.2) 78 (78.8) 44 (44.4) 40 (40.4) 13 (13.1) 2 (2) 36 (36.4) 23 (23.2) 8 (8.1) 20 (20.2) 4 (4) 5 (5.1) 4 (4) 2 (2) 16 (16.2) 8 (8.1) 6 (6.1) 34 (34.3) 8 (8.1) 29 (29.3) 15 (15.2) 15 (15.2) |
30 (57.7) 22 (42.3) 51.2 ± 21.6 - 5 (9.6) 47 (90.4) 25 (48.1) 22 (42.3) 5 (9.6) 0 16 (30.7) 13 (25) 5 (9.6) 13 (25) 8 (15.4) 1 (1.9) 7 (13.4) 1 (1.9) 11 (21.2) 0 2 (3.8) 23 (44.2) 1 (1.9) 19 (36.5) 19 (36.5) 23 (44.2) |
0.400 0.342 0.073 0.675 0.273 0.056 0.234 0.129 0.364 0.003 <0.001 |
| Data presented as mean ± SD or n (%). P < 0.05 considered as significant
HCM: High-Concentration Mask; MV: Mechanical Ventilation |
||||
Pulmonary embolism and pleuropneumonia were the most frequent etiological diagnoses, accounting for 21.9% and 11.3% of cases, respectively. The difference in etiologic diagnoses between the two groups was not statistically significant (P > 0.05). Conventional oxygen therapy (79.5%) was used most frequently for respiratory support. The interfaces used were nasal cannula (37.7%), single mask (6%) and high concentration mask (HCM) (31.8%). Of 151 patients admitted with ARF, 38 (25.2%) received non-invasive ventilation (NIV) and 34 (22.5%) invasive mechanical ventilation. More patients received NIV (P < 0.003) and invasive mechanical ventilation (P = 0.001) after implementation of the BLUE protocol than before.
Table 2 shows the time to etiological diagnosis, time to etiological treatment, mortality at day 28 and hospital stay for the 151 patients admitted with ARF. The time to etiological diagnosis of ARF was one day or less in 69.5% of cases, and more than one day in 30.5% of cases. Significantly more patients in the post-implementation group had their etiological diagnosis within one day of admission than in the pre-implementation group (80.3% vs. 62.7% P = 0.033).
| Table 2: Therapeutic and evolutionary data of patients before and after implementation | ||||
| Variables | All
(n = 151) |
Time of inclusion | P | |
| Before (n = 99) | After (n = 52) | |||
| Time between admission and etiological diagnosis
Same day One day later More than a day after Time between admission and etiological treatment Same day One day later More than a day after Hospital stay Less than a week More than a week Mortality Survivor Deaths |
74 (49) 31 (20.5) 46 (30.5) 68 (45) 27 (17.9) 56 (37.1) 72 (47.7) 79 (52.3) 69 (45.7) 82 (54.3) |
45 (45.5) 17 (17.2) 37 (37.4) 40 (40.4) 13 (13.1) 46 (46.5) 43 (43.4) 56 (56.6) 47 (47.5) 52 (52.5) |
29 (55.8) 14 (26.9) 9 (17.3) 28 (53.8) 14 (26.9) 10 (19.2) 29 (55.8) 23 (44.2) 22 (42.3) 30 (57.7) |
0.033 0.003 0.003 0.545 |
The time to etiological treatment of acute respiratory failure was one day or less in 62.9% of cases, and more than one day in 37.5% of cases. Significantly more patients in the post-implementation group received etiological treatment within one day of admission than in the pre-implementation group (80.3% vs. 53.5% P = 0.003)
Hospital stays for patients admitted with acute respiratory failure was less than one week in 47.7% of cases, and more than one day in 52.3% of cases. Significantly more patients stayed in hospital for less than one week after implementation than before (55.8% vs. 43.4% P = 0.003).
Mortality among patients admitted for acute respiratory failure was 54.3%. It was 52.5% before and 57.7% after implementation. There was no significant difference in mortality between the pre- and post-implementation groups (P = 0.545).
Table 3 shows the imaging examinations carried out to investigate the etiology of acute respiratory failure. In descending order, the imaging examinations carried out for etiological research are pulmonary ultrasound (39.1%), chest x-ray (35.1%), cardiac ultrasound (22.5%), thoracic angioscanner (11.9%) and Doppler ultrasound of the IM Vx (4.1%).
| Table 3: Paraclinical information | ||||
| Variables
|
All
n = 151 |
Time of inclusion | P-value | |
| Before (n = 99) | After (n = 52) | |||
| Chest X-ray
Not realized Realized Angioscan thorax Not realized Realized Lung ultrasound Not realized Realized Heart ultrasound Not carried out Realized Lower limb Doppler echo Not carried out Realized |
98 (64.9) 53 (35.1) 139 (92.1) 18 92 (60.9) 59 (39.1) 117 (77.5) 34 (22.5) 144 (95.4) 7 (4.6) |
51 (51.5) 48 (48.5) 92 (92.9) 11 92 (92.9) 7 (7.1) 75 (75.8) 24 (24.2) 94 (94.9) 5 (5.1) |
47 (90,4) 5 (9.6) 47 (90.4) 7 0 52 (100) 42 (80.8) 10 (19.2) 50 (96,2) 2 (3;8) |
< 0.001 0.829 < 0.001 0.642 0.921 |
A comparison of the number of imaging examinations performed shows a significant reduction in the number of chest X-rays performed after implementation of the BLUE protocol (48.5% vs. 9.6% P = 0.01), but a non-significant reduction in the number of thoracic angioscanner (11.1% vs. 9.6% P = 0.829), cardiac ultrasound (24.2% vs. 19.2% P = 0.642) and MI Doppler ultrasound (5.1% vs. 3.8%). It's worth noting that seven out of 99 patients (7.1%) had undergone lung ultrasound before implementation compared to 100% of patients after implementation.
Figure 3 shows the survival curves for patients enrolled before and after implementation of the BLUE protocol. It shows that survival was comparable between the pre- and post-implementation groups.
Table 4 shows the factors associated with mortality in patients admitted for acute respiratory failure. The table shows that 4 factors are associated with mortality in patients admitted for acute respiratory failure: age over 55 years, the patient not working and the presence of more than one organ failure.
| Table 4: Factors associated with mortality in patients admitted for ARF | ||||
| Determinants
|
Univariate analysis | Multivariate analysis | ||
| P | OR (95% CI) | P | ORa (95% CI) | |
| Age
< 55 years ≥ 55 years Profession In operation No activity No. of failures A single >1 fault |
0.011 0.027 0.003 |
1 1.6 (1.1-2.3) 1 1.5 (1.1-2.3) 1 3.3 (1.2-9.6) |
0.008 0.023 0.002 |
1 2.3 (2.1-45) 1 2.6 (2-6.4) 1 5.7 (2-20.6) |
4. DISCUSSION
ARF is defined as hypoxemia with or without hypercapnia. It is a life-threatening emergency.11-14 Early recognition is essential for optimal diagnostic and therapeutic management. The initiation of appropriate etiological treatment is closely linked to the accuracy of the etiological diagnosis. Advances in clinical, radiologic and scannographic means of lung examination have made it possible to refine the etiological investigation of ARF.15-17
X-rays are an age-old tool that can be used at the patient's bedside and are highly accessible. However, it has a number of troublesome shortcomings in life-threatening emergencies.16,18,19 CT scanning is more informative and fills in the gaps left by radiography. However, it requires patients to be moved, even if they are at risk.16,6,20,21
In the context of limited resources, in addition to the above-mentioned disadvantages, radiography and CT scans face the thorny problem of patients' financial accessibility to these investigations, which are nevertheless useful for diagnosing acute respiratory failure. Hence the interest in finding an etiological investigation method capable of solving the main problem of acute medicine: to act quickly and effectively. Ultrasound offers a fast and easy alternative. Thanks to technological advances, new ultrasound scanners allow physicians to obtain sharper images at the point of care (POC). The heart was the first example of ultrasound exploration by the clinician at the POC.22,23
Ultrasound was subsequently introduced into general emergency practice, particularly in traumatology.24-27 However, LUS was a latecomer in its development, as air was considered an ultrasound obstacle. However, over the last few decades, and following the development of ultrasound lung semiology, LUS has experienced a remarkable boom. Several studies have been carried out on the diagnostic performance of LUS,9,28-30 and simplified protocols for the clinical use of LUS have been proposed.31,32
Our study is one of the few to investigate the impact of LUS on the management of ARF.33,34 It showed that implementation of the BLUE protocol reduced the time to etiological diagnosis, the time to initiation of etiological treatment, and the number of imaging examinations, especially RT and ST, performed. However, it did not change the mortality rate of patients admitted for ARF. This is a landmark study of LUS, specifically the BLUE protocol, in emergency and intensive care units in the Democratic Republic of Congo, a low-resource country.
Our study showed that the proportion of patients admitted with ARF whose etiological diagnosis was established within one day of admission increased significantly after implementation of the BLUE protocol. It increased from 62.7% before to 80.3% after implementation of the BLUE protocol P = 0.033.
This study also showed a significant increase in the proportion of ARF patients receiving appropriate etiological treatment within 1 day of admission. We went from 53.5% receiving etiological treatment on day one before to 80.3% after implementation of the BLUE protocol P = 0.003.
Seyedhosseini et al.36 conducted a randomized study of the impact of the BLUE protocol on the time to treatment of acute respiratory distress in the emergency department. This study found a median time from admission to treatment of 17 minutes for the BLUE group, compared with 38 minutes for the control group. The difference between the two groups was statistically significant (P = 0.0001).
Our two studies show that the use of LUS, specifically the BLUE protocol, reduces the time taken to manage patients admitted to the emergency department and intensive care unit.
Volpicelli G et al. (2006) previously reported that bedside ultrasound reduced costs and saved time in the management of acute respiratory distress in the intensive care unit.35 Ünlüer and Karagöz compared lung ultrasound and radiography in the management of pneumonia. They showed that ultrasound was faster and more accurate in diagnosing pneumonia.36
Our study and that of Seyedhosseini et al. (2017) as well as several previously published studies, show that we can save time in the management of ARF by using bedside LUS. However, several methodological differences between our two-studies merit analysis. They may explain why our delay in initiating treatment is even longer than that of Seyedhosseini et al.
In fact, Seyedhisseini's study was randomized. It included patients admitted to the emergency department. The control group and the study group (BLUE protocol) received conventional imaging for the etiological investigation of acute respiratory failure. The BLUE Protocol group received LUS in addition to conventional imaging. Conventional imaging verified the accuracy of ultrasound diagnosis in the BLUE protocol group, while LUS enabled initiation of etiological treatment.
In contrast, our study was a quasi−experimental before-after study. It involved patients admitted to emergency departments and intensive care units (ICU). All patients could have benefited from conventional imaging and even LUS, depending on affordability, patient condition and availability of the technique. However, all patients admitted after the implementation of the BLUE protocol had received LUS.
The facilitators trained in LUS were used to verify the accuracy of the diagnosis.
All patients admitted with ARF after implementation benefited from LUS, whereas this examination was only performed in 7.1% of cases prior to implementation. Comparing the number of other imaging examinations performed, we recorded a significant reduction in the number of chest X-rays performed after implementation of the BLUE protocol (48.5% vs. 9.6% P = 0.01). However, the numbers of thoracic angioscanner (11.1% vs. 9.6% P = 0.829), cardiac ultrasound (24.2% vs. 19.2% P = 0.642) and lower limbs Doppler ultrasound (5.1% vs. 3.8%) were almost identical.
A retrospective study by Cornesse et al.37 found a significant decrease in chest X-rays and CT scans per ICU stay after lung ultrasound implementation, recording 10.3 ± 12 RTs before vs. 7.7 ± 10 after (P = 0.005) and 0.5 ± 1 ST before vs. 0.4 ± 1 after (P = 0.01).
Our study and that of Cornesse et al. show that the introduction of LUS reduces the number of chest X-rays performed to investigate the etiology of ARF. In our study, the number of STs was almost identical before and after implementation. This difference can be explained by the fact that thoracic CT scans, although the reference imaging technique, are rarely performed in our context, due to several obstacles, especially financial.
However, it should be noted that reducing the number of RTs and STs reduces exposure to ionizing radiation and health care costs.
This study shows that the mortality of patients admitted with ARF was 52.5% before implementation and 57.7% after, with no significant difference (P = 0.545).
Seyedhosseini et al. (2017) observed a mortality of 4.2% in the BLUE group versus 13.6% in the control group. This difference was not statistically significant (P-value: 0.34). Cornesse et al found that mortality was identical before and after implementation. Mortality was 22% and 21% respectively before and after implementation of lung ultrasound (P = 0.75).
A comparison of our study with the two studies leads to the following two observations:
- The use of lung ultrasound did not change the mortality of patients admitted with ARF in any of the three studies. Mortality in patients admitted with ARF is related to multiple factors, and reducing it requires a holistic approach that addresses the various factors associated with mortality.
- Mortality from acute respiratory failure was higher in our study than in the two studies cited above. Our study was conducted in a context of reduced health resources. The poor access of the population to health care may explain this high mortality.
5. CONCLUSION
The aim of this study was to evaluate the impact of implementing the Blue Protocol on the time to etiological management, the frequency of ionizing imaging examinations and the mortality of patients admitted with ARF to the Emergency and Intensive Care Units of the Clinique Universitaire de Kinshasa.
At the end of this study, we can conclude that lung ultrasound offers many advantages in the management of patients with ARF in acute care units, by reducing delays in etiological management, initiating targeted therapy, reducing ionizing examinations performed and not influencing mortality at D28 without significant difference.
However, further research is still needed to better understand the impact of ultrasound on decision-making regarding the management of patients with ARF in acute care units.
6. Data availability
Numerical data generated during this study is available on request.7. Ethical considerations
The protocol was approved by the Department of Anaesthesia and Intensive Care, and also by the Ethics Committee of the School of Public Health, under number ESP/CE/131/2023. Informed consent was obtained from conscious patients, and through an intermediary for patients unable to give it, in accordance with the principles of the Helsinki Convention. We have no conflict of interest in this work.
8. Competing interests
The authors declare that there was no competing interest
9. Funding
This study was completed by using institutional resources, and no external of industry funding was received .No funding received.
10. Authors' contributions
All authors took part in the study conception and design, Data collection, literature review, manuscript drafting, manuscript editing. Statistical analysis, interpretation of results, manuscript review And / or supervision,
11. REFERENCES
- Lamia B, Ougouti R, Molano LC, Cuvelier A. Acute respiratory failure. Rev Prat. 2019;69(3):e93-7.[PubMed]
- Kuo-Chin K, Hsieh MJ, Lin SW, Chuang LP, Chang CH, Hu HC, et al. Survival predictors in elderly patients with acute respiratory distress syndrome: a prospective observational cohort study. Sci Rep. 2018;8(1):13459.[PubMed] DOI: 1038/s41598-018-31811-w
- Lourenço ID, Passos RH, Flato UAP, Figueiredo EJA, Silva AA. Lung ultrasound in the management of acute respiratory distress syndrome: a comprehensive systematic review. Einstein (Sao Paulo). 2024;22(Suppl 2):eRW0598.
- Hu QJ, Shen YC, Jia LQ, Guo SJ, Long NY, Pang CS, et al. Diagnostic performance of lung ultrasound in the diagnosis of pneumonia: a bivariate meta-analysis. Int J Clin Exp Med. 2014;7(1):115-21.[PubMed]
- Alzaharani SSA, Al-Salamah MA, Al Madani WH, Elbary MA. Systematic review and meta-analysis for the use of ultrasound versus radiology in diagnosing pneumonia. Crit Ultrasound J. 2017;9(1):6.[PubMed] DOI: 1186/s13089-017-0059-y
- Kankonde D, Nsiala J, Mbombo W, Tonduangu D, Meziere G, Gizolme D, et al. Performance of Pleuropulmonary Ultrasound in the Diagnosis of Acute Respiratory Conditions in Care Patients: Case of Sens Hospital Center. Anesth Pain Res. 2023;7(2):1-5.[FreeText]
- Laribi S, Keijzers G, Van Meer O, Klim S, Motiejunaite J, Kuan WS, et al. Epidemiology of patients presenting with dyspnea to emergency departments in Europe and the Asia-Pacific region. Eur J Emerg Med. 2019;26(5):345-9.[PubMed] DOI: 1097/MEJ.0000000000000571
- Lard R. Evaluation of the interest of the "BLUE protocol" for improving the diagnosis of dyspneic patients in intra-hospital emergency medicine [dissertation]. 2020. Available from: https://dumas.ccsd.cnrs.fr/dumas-02966618
- Lichtenstein DA, Mezière GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: the BLUE protocol. Chest. 2008;134(1):117-25.[PubMed] DOI: 1378/chest.07-2800
- Lai CC, Tseng KL, Ho CH, Chiang SR, Chen CM, Chan KS, et al. Prognosis of patients with acute respiratory failure and prolonged intensive care unit stay. J Thorac Dis. 2019;11(5):2051-7.[PubMed] DOI: 21037/jtd.2019.04.84
- Papazian L, Pauly V, Hamouda I, Daviet F, Orleans V, Forel JM, et al. National incidence rate and mortality associated with acute respiratory distress syndrome in France. Anaesth Crit Care Pain Med. 2021;40(1):100795.[PubMed] DOI: 1016/j.accpm.2020.100795
- Prescott HC, Sjoding MW, Langa KM, Iwashyna TJ, McAuley DF. Late mortality after acute hypoxic respiratory failure. Thorax. 2018;73(7):618-25.[PubMed] DOI: 1136/thoraxjnl-2017-210109
- Arrivé F, Coudroy R, Thille AW. Early Identification and Diagnostic Approach in Acute Respiratory Distress Syndrome (ARDS). Diagnostics (Basel). 2021;11(12):2307.[PubMed] DOI: 3390/diagnostics11122307
- Kligfield P. The bicentennial of stethoscope 1816 to 2016. Am J Cardiol. 2016;118(10):1601-2.[PubMed] DOI: 1016/j.amjcard.2016.08.033
- Hauser H, Ody B. History of radiology in Geneva and its evolution towards medical imaging. Rev Med Suisse. 2023;19(845-846):1874-8.DOI: 53738/REVMED.2023.19.845-2.1874
- Hounsfield GN. Computerized transverse axial scanning. Br J Radiol. 1973;46(552):1016-22.[PubMed] DOI: 1259/0007-1285-46-552-1016
- Tierney DM, Huelster JS, Overgaard JD, Plunkett MB, Boland LL, St Hill CA, et al. Comparative performance of pulmonary ultrasound, chest radiograph and CT among patients with acute respiratory failure. Crit Care Med. 2020;48(2):151-7.[PubMed] DOI: 1097/CCM.0000000000004124
- Vetrugno L, Biasucci DG, Deana C, Spadaro S, Lombardi FA, Longhini F, et al. Lung ultrasound and supine chest X-ray use in modern adult intensive care: mapping 30 years of advancement (1993-2023). Ultrasound J. 2024;16(1):7.[PubMed] DOI: 1186/s13089-023-00351-4
- See KC, Ong V, Tan YL, Sahagun J, Taculod J. Chest radiography versus lung ultrasound for identification of acute respiratory distress syndrome: a retrospective observational study. Crit Care. 2018;22(1):203.[PubMed] DOI: 1186/s13054-018-2105-y
- Koenig S, Mayo P, Volpicelli G, Millington SJ. Lung Ultrasound Scanning for Respiratory Failure in Acutely Ill Patients: A Review. Chest. 2020;158(6):2511-6.[PubMed] DOI: 1016/j.chest.2020.08.2052
- Roshandel J, Alahyari S, Khazaei M, Asgari R, Moharamzad Y, Zarei E, et al. Diagnostic performance of lung ultrasound compared to CT scan in the diagnosis of pulmonary lesions of COVID-19 induced pneumonia: a preliminary study. Virusdisease. 2021;32(4):674-80.[PubMed] DOI: 1007/s13337-021-00736-w
- Chiumello D, Sferrazza Papa GF, Artigas A, Bouhemad B, Grgic A, Heunks L, et al. ERS statement on chest imaging in acute respiratory failure. Eur Respir J. 2019;54(3):1900435.[PubMed] DOI: 1183/13993003.00435-2019
- Jardin F. Ultrasound exploration in emergency medicine. In: Dubourg, editor. Paris: Masson; 1986. p. 3-154.
- Lapostolle F, Petrovic T. Prehospital ultrasound and cardiological emergencies. Ann Cardiol Angeiol (Paris). 2022;71(6):345-9.[PubMed] DOI: 1016/j.ancard.2022.09.007
- Adnet F, Galinski M, Lapostolle F. Ultrasound in trauma care for the emergency physician: from teaching to practice. Fast ultrasound in the emergency setting. Réanimation. 2004;13(8):465-70.DOI: 1016/j.reaurg.2004.09.002
- Bloom BA, Gibbons RC. Focused Assessment with Sonography for Trauma. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025.Available from: https://www.ncbi.nlm.nih.gov/books/NBK470479/
- Smith J. Focused assessment with sonography in trauma (FAST): should its role be considered? Postgrad Med J. 2010;86(1015):285-91.[PubMed] DOI: 1136/pgmj.2008.076711
- Gamberini L, Tartaglione M, Giugni A, Alban L, Allegri D, Coniglio C, et al. The role of prehospital ultrasound in reducing time to definitive care in abdominal trauma patients with moderate to severe liver and spleen injuries. Injury. 2022;53(5):1587-95.[PubMed] DOI: 1016/j.injury.2021.12.008
- Islam M, Levitus M, Eisen L, Shiloh AL, Fein D. Lung ultrasound for diagnosis and management of acute respiratory failure. Lung. 2020;198(1):1-11.[PubMed] DOI: 1007/s00408-019-00309-1
- Boumans MMA, Aerts W, Pisani L, Bos LDJ, Smit MR, Tuinman PR. Diagnostic accuracy of lung ultrasound in diagnosis of ARDS and identification of focal or non-focal ARDS subphenotypes: a systematic review and meta-analysis. Crit Care. 2024;28(1):224.[PubMed] DOI: 1186/s13054-024-04985-1
- Yuan X, Liu L, Wu W, Huang L, Chao Y, Lu X, et al. Diagnostic accuracy of lung ultrasound for ARF in critically ill patients: a systematic review and meta-analysis. Front Med (Lausanne). 2021;8:705960.[PubMed] DOI: 3389/fmed.2021.705960
- Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest. 2015;147(6):1659-70.[PubMed] DOI: 1378/chest.14-1313
- Lichtenstein D, Malbrain ML. Critical care ultrasound in cardiac arrest. Technological requirements for performing the SESAME-protocol--a holistic approach. Anaesthesiol Intensive Ther. 2015;47(5):471-81.[PubMed] DOI: 5603/AIT.a2015.0072
- Seyedhosseini J, Bashizadeh-Fakhar G, Farzaneh S, Momeni M, Karimialavijeh E. The impact of the BLUE protocol ultrasonography on the time taken to treat acute respiratory distress in the ED. Am J Emerg Med. 2017;35(12):1815-8.[PubMed] DOI: 1016/j.ajem.2017.06.007
- Volpicelli G, Mussa A, Garofalo G, Cardinale L, Casoli G, Perotto F, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med. 2006;24(6):689-96.[PubMed] DOI: 1016/j.ajem.2006.02.013
- Unluer E, Karagoz A, Senturk G, Karaman M, Olow K, Bayata S. Bedside lung ultrasonography for diagnosis of pneumonia. Hong Kong J Emerg Med. 2013;20(2):98-104.DOI: 1177/102490791302000205
- Cornesse A, Zieleskiewicz L, Brun C, Hammad E, Vigne C, Meyssignac B, et al. Impact de l’introduction de l’échographie pleuro-pulmonaire dans une unité de réanimation sur le nombre de radiographies et scanners thoraciques prescrits. Ann Fr Anesth Reanim. 2014;33(Suppl 2):A130. DOI: 1016/j.annfar.2014.07.215