Tresna Kusumah Natapraja 1 , Raka Jati Prasetya *2
Authors affiliations:
Background: ACTH4-10Pro8-Gly9-Pro10, a synthetic adrenocorticotropic hormone analogue lacking hormonal effects, was investigated for its neuroprotective effects against ketamine-induced neurotoxicity in neonatal rats.
Methodology: This experimental study employed a post-test-only control group design with simple randomization and double blinding. Forty-two male Sprague-Dawley rats (7 days old, 15–20 g) were divided into three groups (n=14): control, ketamine (40 mg/kg BW subcutaneously), and ACTH4-10Pro8-Gly9-Pro10 (50 mcg/kg BW intranasally) + ketamine. BDNF expression in the cortex and hippocampus tissues and serum BDNF levels were analyzed.
Results: ACTH4-10Pro8-Gly9-Pro10 pretreatment significantly increased the percentage of BDNF-expressing cells compared to the ketamine group (31.0±7.9%, P = 0.001). Serum BDNF levels were higher in the treatment group, but the difference was not statistically significant (P = 0.723).
Conclusion: ACTH4-10Pro8-Gly9-Pro10 demonstrates neuroprotective potential against ketamine-induced neurotoxicity in neonatal rats by increasing BDNF expression, although not serum levels.
Keywords: ACTH4-10Pro8-Gly9-Pro10; BDNF; Ketamine-induced neurotoxicity; Apoptosis
Citation: Natapraja TK, Prasetya RJ. Neuroprotective effects of ACTH4-10PRO8-GLY9-PRO10 on the ketamine-induced neurotoxicity in neonatal rats: increased BDNF expression and BDNF serum concentrations. Anaesth. pain intensive care 2026;30(1):75-80 DOI: 10.35975/apic.v30i1.3105
Received: July 21, 2025; Revised: September 01, 2024; Accepted: September 05, 2025
Advancements in pediatric surgical techniques have led to more prolonged and intricate anesthetic procedures, potentially resulting in anesthetic-induced neurotoxicity in children. Over the last ten years, an increasing body of animal studies and clinical evidence has indicated a link between the duration of anesthetic exposure, early brain development during the growth spurt period, and the possibility of lasting cognitive impairment.1
The central nervous system (CNS) experiences a growth spurt with extensive synaptogenesis during early childhood, rendering the brain highly vulnerable to external stimuli. Studies on the use of general anesthesia, particularly ketamine, during childbirth have revealed adverse effects on the neurological development of infants, including autism, developmental disorders, and mental health issues.2 Subsequently, numerous primate and rodent studies have demonstrated the detrimental effects of anesthetics. When commonly used anesthetic drugs are administered to neonatal animals, the results indicate long-term neurological damage and behavioral disturbances. This phenomenon has been documented by various research groups across multiple species, including non-primate animals. Evidence suggests that prolonged exposure, concurrent administration of multiple anesthetics, and repeated exposure cause histological damage. The extent of this damage appears to correlate with the observed behavioral changes.3
A cohort study conducted in 2011 in Rochester, Minnesota, involving over 8000 children, included 350 subjects exposed to anesthesia before the age of two. These children, who were exposed to multiple types of anesthetic drugs and surgical procedures, exhibited a significantly higher incidence of learning disabilities compared to unexposed controls.4 In Western Australia, researchers examined cognitive changes following anesthesia in nearly 3000 children. In this study, 321 children were exposed to anesthetic drugs before the age of three years. At ten years old, the children underwent individual neuropsychological tests to assess their behaviour. This cohort demonstrated deficits in language and abstract reasoning by the age of ten. However, no significant differences were observed in fine motor or gross function assessments, nor in the standardized questionnaire completed by parents to evaluate behavioural problems.5
Human studies are currently limited to retrospective cohort studies, with inconclusive results due to substantial confounding factors that are challenging to identify. Consequently, further research is necessary to investigate the toxicity of anesthetic agents that potentially cause brain damage during development.6
Numerous animal studies have demonstrated that repeated ketamine exposure leads to compromised brain function linked to neuroapoptotic damage in developing brains. This discovery has prompted medical professionals to reassess the safety of ketamine anesthesia in pediatric patients1. The United States Food and Drug Administration has cautioned against the use of anesthetic drugs administered repeatedly or for durations exceeding 3 hours during surgeries or procedures on children under three years of age or women in their third trimester of pregnancy, citing potential adverse effects on the child's brain development.7
ACTH4-10Pro8-Gly9-Pro10, a synthetic analogue of a short adrenocorticotropic hormone (ACTH) fragment, lacks hormonal effects but exhibits neuromodulatory activity. This drug has been shown to possess immune-modulating and neurotrophic activities, balancing anti-inflammatory and trophic factors (IL-IO, TNF-α, TGF-Pl, BDNF, and NGF) against proinflammatory factors (IL-Ip, IL-8, CRP, and LE). It also enhances anti-apoptotic defense (by elevating Bcl-2) and reduces peroxidation processes (by increasing SOD activity).8 The neuromodulatory characteristics of ACTH4-10Pro8-Gly9-Pro10 enable it to function as a neuroprotector by inhibiting apoptotic processes. By modulating BDNF levels, ACTH4-10Pro8-Gly9-Pro10 impedes apoptosis. Research conducted by Gusev and Skvortsova (2000) revealed that ischemic stroke patients treated with ACTH4-10Pro8-Gly9-Pro10 exhibited increased BDNF levels, reduced mortality rates, and shorter hospital stays.8,9
Given these findings, we investigated the impact of ACTH4-10Pro8-Gly9-Pro10 administration on ketamine-induced neurotoxicity in neonatal rats by evaluating BDNF levels.
This investigation employed a legitimate experimental research methodology utilizing a posttest-only control group design. A simple randomization technique with double blinding was implemented for treatment allocation. The analyst assigned random numerical labels (1-42) to all samples. The rodents were randomly segregated into three groups of 14. The sample distribution included a negative-positive control (subcutaneous ketamine 40 mg/kg BW) and a treatment group (intranasal ACTH4-10Pro8-Gly9-Pro10 at 50 mcg/kg BW and subcutaneous ketamine at 40 mg/kg BW). The Faculty of Medicine Health Research Ethics Committee of the University of North Sumatra approved this study (381/KEP/USU/2020). The Experimental Animal Care Unit Laboratory at Brawijaya Malang's Faculty of Medicine facilitated the treatment of experimental animals and examination of BDNF serum levels using an enzyme-linked immunosorbent assay (ELISA). BDNF was also assessed through IHC staining to identify specific proteins in tissues or cells using antibodies. The study was conducted in January 2021 using seven-day-old male Rattus norvegicus rats of the Sprague-Dawley strain, weighing 15-20 grams, sourced from the Experimental Animal Care Unit. The exclusion criteria encompassed aggressive behavior towards other groups and failure to survive the entire study period. The research adhered to the 3R principles (reduction, replacement, and refinement) and the 5 F principles of animal welfare. Participants were withdrawn if they became ill or died before completing the research procedure.
ACTH4-10Pro8-Gly9-Pro10 therapy, marketed as Semax®, was produced by The Institute of Molecular Genetics Russian Academy of Sciences. Ketamine-Hameln®, manufactured by PT. Combiphar Indonesia, was the ketamine used. Statistical analysis of BDNF serum levels in control and treated rats was performed using SPSS 25.0, with a 5% significance level and a 95% confidence interval (α= 0.05). This study was registered at ClinicalTrials.gov (no. NCT05648526).
This investigation utilized 14 male Rattus norvegicus per group, aged seven days, weighing 15-20 grams, of the Sprague-Dawley strain, sourced from the Experimental Animal Care Unit. During the course of the study, mortality rates varied across groups: two rats in the negative control group, one in the treatment group, nine in the positive control group, and 21 in the treatment group.
Table 1 shows the BDNF expression in neonatal rats administered ketamine and ACTH4-10Pro8-Gly9-Pro10 prior to ketamine. The findings revealed that the group receiving ACTH4-10Pro8-Gly9-Pro10 before ketamine administration exhibited an increased percentage of cells expressing BDNF compared to the positive control group (subcutaneous ketamine). The distribution percentage was 31.0±7.9%, which was statistically significant (P = 0.001) (Table 1) (Figure 1).
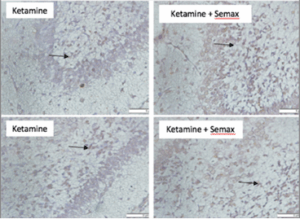
Figure 1. Distribution of BDNF staining in the brains of Rattus norvegicus Sprague Dawley strain. The preparations were observed using a microscope with a magnification of 400x, and image analysis was performed using ImageJ 1.53c software
The results of the study presented in Table 1 show the effects of treatment with ACTH4-10Pro8-Gly9-Pro10 prior to ketamine administration on BDNF expression in neonatal rats. The analysis compared two experimental groups: a positive control group (P1) treated with subcutaneous ketamine and a treatment group (P2) that received ACTH4-10Pro8-Gly9-Pro10 before ketamine administration. The key parameters analysed were the expression activity of Brain-Derived Neurotrophic Factor (BDNF) and the serum levels of BDNF.
The first notable observation was that the expression activity, represented by the percentage distribution of cells expressing BDNF, was significantly higher in the treatment group (P2) than in the control group (P1). The distribution percentage in P1 was 19.9 ± 4.9%, while in P2 it was 31.0 ± 7.9%, with a p-value of 0.001. This result indicates that the administration of ACTH4-10Pro8-Gly9-Pro10 before ketamine significantly enhanced BDNF expression in the cells, suggesting a potential neuroprotective effect or a mechanism that enhances neuroplasticity in response to the combination of ACTH4-10Pro8-Gly9-Pro10 and ketamine.
Despite the statistically significant difference in expression activity, serum BDNF levels did not differ significantly (P = 0.723) (Table 1). Nevertheless, there was a tendency for higher serum BDNF levels in the semax and ketamine groups.
This finding is important because BDNF is a key protein involved in the growth, maintenance, and survival of neurones, and its expression is often associated with improved cognitive function and neuronal health. The increased expression of BDNF in the treatment group may suggest that the peptide ACTH4-10Pro8-Gly9-Pro10 plays a crucial role in amplifying ketamine’s neurogenic effects, which could have therapeutic implications, particularly in treating neurological disorders in which BDNF levels are typically reduced.
However, when serum BDNF levels were measured, no significant difference was observed between the treatment (P2) and control (P1) groups. The serum BDNF levels in P1 were 2.6 ± 0.36 ng/mL, while in P2, they were 2.5 ± 0.4 ng/mL, with a p-value of 0.723. This lack of statistical significance suggests that while
ACTH4-10Pro8-Gly9-Pro10 enhances the local expression of BDNF in cells, it does not significantly alter the overall serum concentration of BDNF in the system. This observation could imply that the observed changes in BDNF expression might be more localized within the brain or tissues rather than reflecting a systemic change in the circulating levels of BDNF.
Interestingly, although the serum levels did not differ significantly, there was a slight trend toward higher serum BDNF levels in the semax and ketamine treatment groups, although this did not reach statistical significance. This could indicate that there are other factors or treatment combinations that may influence serum BDNF, which were not fully captured in this study, or that a more refined experimental design could reveal these subtle differences.
The findings of this study suggest that ACTH4-10Pro8-Gly9-Pro10, when administered prior to ketamine, significantly enhances BDNF expression, suggesting a beneficial effect on neuronal activity and plasticity. However, the treatment did not significantly affect serum BDNF levels, indicating that the peptide may influence local BDNF expression in the brain without altering its systemic concentrations. Further studies with more refined measurement techniques could explore the underlying mechanisms of localized BDNF upregulation and its potential therapeutic applications in neurological disorders.
This investigation aimed to demonstrate that administering ACTH4-10Pro8-Gly9 Pro10 can mitigate the occurrence of neurotoxicity caused by ketamine administration in Rattus norvegicus Sprague Dawley strain PND 7. The levels of serum caspase 3, BDNF, and Bcl-2 in the brain were examined. The study involved 42 rats, which were allocated into three groups: negative control, positive control, and treatment.
Ketamine use in pediatric patients remains limited, primarily for surgical procedures and diagnostic tests. Despite its continued use, substantial evidence from animal studies indicates that ketamine can induce neuronal death and disrupt neurogenesis following injection. Conversely, ACTH4-10Pro8-Gly9-Pro10, a peptide compound with neuroprotective qualities, is considered relatively safe and has been shown to alleviate stress response, hypoxia, and ischemia in the brain. These properties make it a promising candidate for neuroprotection against ketamine-induced neurotoxicity.10–12
Initially, ketamine interacts with the NMDA receptor at an appropriate concentration. NMDA receptor activation is crucial in the hippocampus during learning and memory processes. Inhibiting NMDA receptors in the brain compromises synaptic plasticity, ultimately interfering with learning and memory functions.13 This demonstrates that temporary NMDA receptor inhibition during the brain's growth spurt or synaptogenesis period triggers neuro-apoptosis.14
Subsequently, prolonged inhibition of NMDA receptors by ketamine leads to an increase in the number of NMDA receptors after ketamine administration ceases. This increase renders these receptors on neurones more vulnerable to the excitotoxic effects of endogenous glutamate, resulting in neuronal accumulation of toxic intracellular Ca2+ ions. The excessive accumulation of Ca2+ in the mitochondria elevates ROS levels, causes depolarization of the mitochondrial membrane, and activates the apoptotic cascade. 15,16
Ketamine enhances NMDA receptors and induces neurotoxicity through the GABA signal transduction pathway. This occurs due to the impairment of mitochondrial morphogenesis, integrity, and function during peak synaptogenesis. This process triggers neurotoxicity by increasing mitochondrial permeability and reducing the mitochondrial membrane potential. Consequently, this leads to reduced expression of the anti-apoptotic protein Bcl-2, increased BCL2-associated X protein (Bax) expression, and stimulated cytochrome C release. The mitochondrial apoptosis activation pathway is initiated via caspase-9 and caspase-3, resulting in DNA fragmentation and neuronal death.17 NMDA receptor inhibition on internal GABA interneurons immobilizes glutamatergic neurones owing to reduced GABAergic inhibitors. This inhibition releases glutamate, primarily forming AMPA and kainate receptors. Although the combined effect of NMDA receptor blockade and AMPA activation is brief due to ketamine's short half-life, it is believed to be sufficient to elicit the antidepressant effect of ketamine. Furthermore, AMPA receptor activation stimulates BDNF expression and release, which in turn activates TrkB.18
Despite the mechanism suggesting ketamine's potential to cause apoptosis and neurotoxicity, it remains widely accepted as a potent anesthetic and analgesic agent. Its short duration of action and ability to maintain hemodynamic status and respiratory stabilization contribute to its widespread use.19 Researchers have explored neuroprotective agents to prevent histological and developmental abnormalities caused by an-aesthetic drugs. Perianesthetic treatment with ROS scavengers or mitochondrial protection has shown promise in preventing anesthesia-induced cognitive impairment. The administration of neuropeptides is another approach being investigated to mitigate neurotoxicity.20
A separate study involving experimental animals with cerebral artery occlusion treated with ACTH4-10Pro8-Gly9-Pro10 demonstrated reduced neurological deficits (p <0.05, compared to placebo). Initial clinical trials of ACTH4-10Pro8-Gly9-Pro10 in patients with acute focal brain ischemia have been conducted at the Department of Neurology, Russian State University. This study aimed to assess the safety and efficacy of ACTH4-10Pro8-Gly9-Pro10 neuroprotective effects in patients with acute ischemic stroke. This randomized, blinded study compared intranasal ACTH4-10Pro8-Gly9-Pro10 administration with placebo. Results indicated a more substantial reduction in neurological deficits in the ACTH4-10Pro8-Gly9-Pro10 group.8
No additional studies have been conducted on the impact of ACTH4-10Pro8-Gly9-Pro10 on ketamine-induced neurotoxicity in humans. Such research has been hindered by the challenges of obtaining neurones and performing developmental toxicity tests in pediatric and foetal groups. It is not feasible to postpone surgery for a child or administer anesthetics to healthy children solely for the purpose of investigating ketamine's neurotoxic effects.21 Numerous investigations using growing animal models, such as rodents and other primates experiencing growth spurts, have demonstrated that ketamine neurotoxicity occurs through an apoptotic mechanism. This neurotoxicity can be identified by the expression of caspase, Bcl-2, and BDNF in the brain.
A comparative investigation in Russia examined the effects of ACTH4-10Pro8-Gly9-Pro10 administration in healthy animals and humans. The findings revealed that this neuropeptide could enhance memory and attention, boost hypoxia resistance, and improve cerebral blood flow over an extended period. Notably, in healthy human subjects, the administration of ACTH4-10Pro8-Gly9-Pro10 during extreme conditions or activities led to significant improvements in memory and attention. Overall, these neuropeptides exhibited positive effects without negative side effects. This neuropeptide shows promise for novel applications in the medical field.22
Administration of ACTH4-10Pro8-Gly9-Pro10 demonstrated neuroprotective effects against ketamine-induced neurotoxicity in newborn rats. This compound significantly enhanced the proportion of cells expressing BDNF, although it did not lead to a substantial increase in serum BDNF levels.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
TKN: Conception and design. provision of study materials or patients.
RJP: Conception and design, administrative support
10. REFERENCES
Authors affiliations:
- Tresna Kusumah Natapraja, Department of Anesthesiology & Intensive Care, Faculty of Medicine, Universitas Padjadjaran, Indonesia; Email: t9725703@gmail.com
- Raka Jati Prasetya, Department of Anesthesiology & Intensive Care, Faculty of Medicine, Universitas Sumatera Utara, Indonesia; Email: rakajati@usu.ac.id
ABSTRACT
Background: ACTH4-10Pro8-Gly9-Pro10, a synthetic adrenocorticotropic hormone analogue lacking hormonal effects, was investigated for its neuroprotective effects against ketamine-induced neurotoxicity in neonatal rats.
Methodology: This experimental study employed a post-test-only control group design with simple randomization and double blinding. Forty-two male Sprague-Dawley rats (7 days old, 15–20 g) were divided into three groups (n=14): control, ketamine (40 mg/kg BW subcutaneously), and ACTH4-10Pro8-Gly9-Pro10 (50 mcg/kg BW intranasally) + ketamine. BDNF expression in the cortex and hippocampus tissues and serum BDNF levels were analyzed.
Results: ACTH4-10Pro8-Gly9-Pro10 pretreatment significantly increased the percentage of BDNF-expressing cells compared to the ketamine group (31.0±7.9%, P = 0.001). Serum BDNF levels were higher in the treatment group, but the difference was not statistically significant (P = 0.723).
Conclusion: ACTH4-10Pro8-Gly9-Pro10 demonstrates neuroprotective potential against ketamine-induced neurotoxicity in neonatal rats by increasing BDNF expression, although not serum levels.
Keywords: ACTH4-10Pro8-Gly9-Pro10; BDNF; Ketamine-induced neurotoxicity; Apoptosis
Citation: Natapraja TK, Prasetya RJ. Neuroprotective effects of ACTH4-10PRO8-GLY9-PRO10 on the ketamine-induced neurotoxicity in neonatal rats: increased BDNF expression and BDNF serum concentrations. Anaesth. pain intensive care 2026;30(1):75-80 DOI: 10.35975/apic.v30i1.3105
Received: July 21, 2025; Revised: September 01, 2024; Accepted: September 05, 2025
1. INTRODUCTION
Advancements in pediatric surgical techniques have led to more prolonged and intricate anesthetic procedures, potentially resulting in anesthetic-induced neurotoxicity in children. Over the last ten years, an increasing body of animal studies and clinical evidence has indicated a link between the duration of anesthetic exposure, early brain development during the growth spurt period, and the possibility of lasting cognitive impairment.1
The central nervous system (CNS) experiences a growth spurt with extensive synaptogenesis during early childhood, rendering the brain highly vulnerable to external stimuli. Studies on the use of general anesthesia, particularly ketamine, during childbirth have revealed adverse effects on the neurological development of infants, including autism, developmental disorders, and mental health issues.2 Subsequently, numerous primate and rodent studies have demonstrated the detrimental effects of anesthetics. When commonly used anesthetic drugs are administered to neonatal animals, the results indicate long-term neurological damage and behavioral disturbances. This phenomenon has been documented by various research groups across multiple species, including non-primate animals. Evidence suggests that prolonged exposure, concurrent administration of multiple anesthetics, and repeated exposure cause histological damage. The extent of this damage appears to correlate with the observed behavioral changes.3
A cohort study conducted in 2011 in Rochester, Minnesota, involving over 8000 children, included 350 subjects exposed to anesthesia before the age of two. These children, who were exposed to multiple types of anesthetic drugs and surgical procedures, exhibited a significantly higher incidence of learning disabilities compared to unexposed controls.4 In Western Australia, researchers examined cognitive changes following anesthesia in nearly 3000 children. In this study, 321 children were exposed to anesthetic drugs before the age of three years. At ten years old, the children underwent individual neuropsychological tests to assess their behaviour. This cohort demonstrated deficits in language and abstract reasoning by the age of ten. However, no significant differences were observed in fine motor or gross function assessments, nor in the standardized questionnaire completed by parents to evaluate behavioural problems.5
Human studies are currently limited to retrospective cohort studies, with inconclusive results due to substantial confounding factors that are challenging to identify. Consequently, further research is necessary to investigate the toxicity of anesthetic agents that potentially cause brain damage during development.6
Numerous animal studies have demonstrated that repeated ketamine exposure leads to compromised brain function linked to neuroapoptotic damage in developing brains. This discovery has prompted medical professionals to reassess the safety of ketamine anesthesia in pediatric patients1. The United States Food and Drug Administration has cautioned against the use of anesthetic drugs administered repeatedly or for durations exceeding 3 hours during surgeries or procedures on children under three years of age or women in their third trimester of pregnancy, citing potential adverse effects on the child's brain development.7
ACTH4-10Pro8-Gly9-Pro10, a synthetic analogue of a short adrenocorticotropic hormone (ACTH) fragment, lacks hormonal effects but exhibits neuromodulatory activity. This drug has been shown to possess immune-modulating and neurotrophic activities, balancing anti-inflammatory and trophic factors (IL-IO, TNF-α, TGF-Pl, BDNF, and NGF) against proinflammatory factors (IL-Ip, IL-8, CRP, and LE). It also enhances anti-apoptotic defense (by elevating Bcl-2) and reduces peroxidation processes (by increasing SOD activity).8 The neuromodulatory characteristics of ACTH4-10Pro8-Gly9-Pro10 enable it to function as a neuroprotector by inhibiting apoptotic processes. By modulating BDNF levels, ACTH4-10Pro8-Gly9-Pro10 impedes apoptosis. Research conducted by Gusev and Skvortsova (2000) revealed that ischemic stroke patients treated with ACTH4-10Pro8-Gly9-Pro10 exhibited increased BDNF levels, reduced mortality rates, and shorter hospital stays.8,9
Given these findings, we investigated the impact of ACTH4-10Pro8-Gly9-Pro10 administration on ketamine-induced neurotoxicity in neonatal rats by evaluating BDNF levels.
2. METHODOLOGY
This investigation employed a legitimate experimental research methodology utilizing a posttest-only control group design. A simple randomization technique with double blinding was implemented for treatment allocation. The analyst assigned random numerical labels (1-42) to all samples. The rodents were randomly segregated into three groups of 14. The sample distribution included a negative-positive control (subcutaneous ketamine 40 mg/kg BW) and a treatment group (intranasal ACTH4-10Pro8-Gly9-Pro10 at 50 mcg/kg BW and subcutaneous ketamine at 40 mg/kg BW). The Faculty of Medicine Health Research Ethics Committee of the University of North Sumatra approved this study (381/KEP/USU/2020). The Experimental Animal Care Unit Laboratory at Brawijaya Malang's Faculty of Medicine facilitated the treatment of experimental animals and examination of BDNF serum levels using an enzyme-linked immunosorbent assay (ELISA). BDNF was also assessed through IHC staining to identify specific proteins in tissues or cells using antibodies. The study was conducted in January 2021 using seven-day-old male Rattus norvegicus rats of the Sprague-Dawley strain, weighing 15-20 grams, sourced from the Experimental Animal Care Unit. The exclusion criteria encompassed aggressive behavior towards other groups and failure to survive the entire study period. The research adhered to the 3R principles (reduction, replacement, and refinement) and the 5 F principles of animal welfare. Participants were withdrawn if they became ill or died before completing the research procedure.
ACTH4-10Pro8-Gly9-Pro10 therapy, marketed as Semax®, was produced by The Institute of Molecular Genetics Russian Academy of Sciences. Ketamine-Hameln®, manufactured by PT. Combiphar Indonesia, was the ketamine used. Statistical analysis of BDNF serum levels in control and treated rats was performed using SPSS 25.0, with a 5% significance level and a 95% confidence interval (α= 0.05). This study was registered at ClinicalTrials.gov (no. NCT05648526).
3. RESULTS
This investigation utilized 14 male Rattus norvegicus per group, aged seven days, weighing 15-20 grams, of the Sprague-Dawley strain, sourced from the Experimental Animal Care Unit. During the course of the study, mortality rates varied across groups: two rats in the negative control group, one in the treatment group, nine in the positive control group, and 21 in the treatment group.
Table 1 shows the BDNF expression in neonatal rats administered ketamine and ACTH4-10Pro8-Gly9-Pro10 prior to ketamine. The findings revealed that the group receiving ACTH4-10Pro8-Gly9-Pro10 before ketamine administration exhibited an increased percentage of cells expressing BDNF compared to the positive control group (subcutaneous ketamine). The distribution percentage was 31.0±7.9%, which was statistically significant (P = 0.001) (Table 1) (Figure 1).
| Table 1: Expression of BDNF in positive control and treatment groups | |||
| BDNF | P1 (Control)
(n = 11) |
P2 (Treatment)
(n = 10) |
P-value |
| Expression activity (distribution percentage) | 19.9 ± 4.9 | 31.0 ± 7.9 | 0.001 |
| Serum level (ng/mL) | 2.6 ± 0.6 | 2.5 ± 0.4 | 0.723 |
| P < 0.05 considered as significant | |||

Figure 1. Distribution of BDNF staining in the brains of Rattus norvegicus Sprague Dawley strain. The preparations were observed using a microscope with a magnification of 400x, and image analysis was performed using ImageJ 1.53c software
The results of the study presented in Table 1 show the effects of treatment with ACTH4-10Pro8-Gly9-Pro10 prior to ketamine administration on BDNF expression in neonatal rats. The analysis compared two experimental groups: a positive control group (P1) treated with subcutaneous ketamine and a treatment group (P2) that received ACTH4-10Pro8-Gly9-Pro10 before ketamine administration. The key parameters analysed were the expression activity of Brain-Derived Neurotrophic Factor (BDNF) and the serum levels of BDNF.
The first notable observation was that the expression activity, represented by the percentage distribution of cells expressing BDNF, was significantly higher in the treatment group (P2) than in the control group (P1). The distribution percentage in P1 was 19.9 ± 4.9%, while in P2 it was 31.0 ± 7.9%, with a p-value of 0.001. This result indicates that the administration of ACTH4-10Pro8-Gly9-Pro10 before ketamine significantly enhanced BDNF expression in the cells, suggesting a potential neuroprotective effect or a mechanism that enhances neuroplasticity in response to the combination of ACTH4-10Pro8-Gly9-Pro10 and ketamine.
Despite the statistically significant difference in expression activity, serum BDNF levels did not differ significantly (P = 0.723) (Table 1). Nevertheless, there was a tendency for higher serum BDNF levels in the semax and ketamine groups.
This finding is important because BDNF is a key protein involved in the growth, maintenance, and survival of neurones, and its expression is often associated with improved cognitive function and neuronal health. The increased expression of BDNF in the treatment group may suggest that the peptide ACTH4-10Pro8-Gly9-Pro10 plays a crucial role in amplifying ketamine’s neurogenic effects, which could have therapeutic implications, particularly in treating neurological disorders in which BDNF levels are typically reduced.
However, when serum BDNF levels were measured, no significant difference was observed between the treatment (P2) and control (P1) groups. The serum BDNF levels in P1 were 2.6 ± 0.36 ng/mL, while in P2, they were 2.5 ± 0.4 ng/mL, with a p-value of 0.723. This lack of statistical significance suggests that while
ACTH4-10Pro8-Gly9-Pro10 enhances the local expression of BDNF in cells, it does not significantly alter the overall serum concentration of BDNF in the system. This observation could imply that the observed changes in BDNF expression might be more localized within the brain or tissues rather than reflecting a systemic change in the circulating levels of BDNF.
Interestingly, although the serum levels did not differ significantly, there was a slight trend toward higher serum BDNF levels in the semax and ketamine treatment groups, although this did not reach statistical significance. This could indicate that there are other factors or treatment combinations that may influence serum BDNF, which were not fully captured in this study, or that a more refined experimental design could reveal these subtle differences.
The findings of this study suggest that ACTH4-10Pro8-Gly9-Pro10, when administered prior to ketamine, significantly enhances BDNF expression, suggesting a beneficial effect on neuronal activity and plasticity. However, the treatment did not significantly affect serum BDNF levels, indicating that the peptide may influence local BDNF expression in the brain without altering its systemic concentrations. Further studies with more refined measurement techniques could explore the underlying mechanisms of localized BDNF upregulation and its potential therapeutic applications in neurological disorders.
4. DISCUSSION
This investigation aimed to demonstrate that administering ACTH4-10Pro8-Gly9 Pro10 can mitigate the occurrence of neurotoxicity caused by ketamine administration in Rattus norvegicus Sprague Dawley strain PND 7. The levels of serum caspase 3, BDNF, and Bcl-2 in the brain were examined. The study involved 42 rats, which were allocated into three groups: negative control, positive control, and treatment.
Ketamine use in pediatric patients remains limited, primarily for surgical procedures and diagnostic tests. Despite its continued use, substantial evidence from animal studies indicates that ketamine can induce neuronal death and disrupt neurogenesis following injection. Conversely, ACTH4-10Pro8-Gly9-Pro10, a peptide compound with neuroprotective qualities, is considered relatively safe and has been shown to alleviate stress response, hypoxia, and ischemia in the brain. These properties make it a promising candidate for neuroprotection against ketamine-induced neurotoxicity.10–12
Initially, ketamine interacts with the NMDA receptor at an appropriate concentration. NMDA receptor activation is crucial in the hippocampus during learning and memory processes. Inhibiting NMDA receptors in the brain compromises synaptic plasticity, ultimately interfering with learning and memory functions.13 This demonstrates that temporary NMDA receptor inhibition during the brain's growth spurt or synaptogenesis period triggers neuro-apoptosis.14
Subsequently, prolonged inhibition of NMDA receptors by ketamine leads to an increase in the number of NMDA receptors after ketamine administration ceases. This increase renders these receptors on neurones more vulnerable to the excitotoxic effects of endogenous glutamate, resulting in neuronal accumulation of toxic intracellular Ca2+ ions. The excessive accumulation of Ca2+ in the mitochondria elevates ROS levels, causes depolarization of the mitochondrial membrane, and activates the apoptotic cascade. 15,16
Ketamine enhances NMDA receptors and induces neurotoxicity through the GABA signal transduction pathway. This occurs due to the impairment of mitochondrial morphogenesis, integrity, and function during peak synaptogenesis. This process triggers neurotoxicity by increasing mitochondrial permeability and reducing the mitochondrial membrane potential. Consequently, this leads to reduced expression of the anti-apoptotic protein Bcl-2, increased BCL2-associated X protein (Bax) expression, and stimulated cytochrome C release. The mitochondrial apoptosis activation pathway is initiated via caspase-9 and caspase-3, resulting in DNA fragmentation and neuronal death.17 NMDA receptor inhibition on internal GABA interneurons immobilizes glutamatergic neurones owing to reduced GABAergic inhibitors. This inhibition releases glutamate, primarily forming AMPA and kainate receptors. Although the combined effect of NMDA receptor blockade and AMPA activation is brief due to ketamine's short half-life, it is believed to be sufficient to elicit the antidepressant effect of ketamine. Furthermore, AMPA receptor activation stimulates BDNF expression and release, which in turn activates TrkB.18
Despite the mechanism suggesting ketamine's potential to cause apoptosis and neurotoxicity, it remains widely accepted as a potent anesthetic and analgesic agent. Its short duration of action and ability to maintain hemodynamic status and respiratory stabilization contribute to its widespread use.19 Researchers have explored neuroprotective agents to prevent histological and developmental abnormalities caused by an-aesthetic drugs. Perianesthetic treatment with ROS scavengers or mitochondrial protection has shown promise in preventing anesthesia-induced cognitive impairment. The administration of neuropeptides is another approach being investigated to mitigate neurotoxicity.20
A separate study involving experimental animals with cerebral artery occlusion treated with ACTH4-10Pro8-Gly9-Pro10 demonstrated reduced neurological deficits (p <0.05, compared to placebo). Initial clinical trials of ACTH4-10Pro8-Gly9-Pro10 in patients with acute focal brain ischemia have been conducted at the Department of Neurology, Russian State University. This study aimed to assess the safety and efficacy of ACTH4-10Pro8-Gly9-Pro10 neuroprotective effects in patients with acute ischemic stroke. This randomized, blinded study compared intranasal ACTH4-10Pro8-Gly9-Pro10 administration with placebo. Results indicated a more substantial reduction in neurological deficits in the ACTH4-10Pro8-Gly9-Pro10 group.8
No additional studies have been conducted on the impact of ACTH4-10Pro8-Gly9-Pro10 on ketamine-induced neurotoxicity in humans. Such research has been hindered by the challenges of obtaining neurones and performing developmental toxicity tests in pediatric and foetal groups. It is not feasible to postpone surgery for a child or administer anesthetics to healthy children solely for the purpose of investigating ketamine's neurotoxic effects.21 Numerous investigations using growing animal models, such as rodents and other primates experiencing growth spurts, have demonstrated that ketamine neurotoxicity occurs through an apoptotic mechanism. This neurotoxicity can be identified by the expression of caspase, Bcl-2, and BDNF in the brain.
A comparative investigation in Russia examined the effects of ACTH4-10Pro8-Gly9-Pro10 administration in healthy animals and humans. The findings revealed that this neuropeptide could enhance memory and attention, boost hypoxia resistance, and improve cerebral blood flow over an extended period. Notably, in healthy human subjects, the administration of ACTH4-10Pro8-Gly9-Pro10 during extreme conditions or activities led to significant improvements in memory and attention. Overall, these neuropeptides exhibited positive effects without negative side effects. This neuropeptide shows promise for novel applications in the medical field.22
5. CONCLUSION
Administration of ACTH4-10Pro8-Gly9-Pro10 demonstrated neuroprotective effects against ketamine-induced neurotoxicity in newborn rats. This compound significantly enhanced the proportion of cells expressing BDNF, although it did not lead to a substantial increase in serum BDNF levels.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
TKN: Conception and design. provision of study materials or patients.
RJP: Conception and design, administrative support
10. REFERENCES
- Kokane SS, Lin Q. Impact of early life ketamine exposure on the developing brain and cognitive sequelae: a discussion of apoptotic neurodegeneration mechanisms. Neuropathol Drug Addict Subst Misuse. 2016; 28:581–92.
- Hattori R, Desimaru M, Nagayama I, Inoue K. Autistic, and developmental disorders after general anesthetic delivery. Lancet. 1991;337(8753):1357–8. DOI: 1016/0140-6736(91)93045-b
- Montana MC, Evers AS. Anesthetic neurotoxicity: New findings and future directions. J Pediatr. 2017; 181:279–85. DOI: 1016/j.jpeds.2016.10.049
- Flick RP, Katusic SK, Colligan RC, Wilder RT, Voigt RG, Olson MD, et al. Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics. 2011;128(5):e1053-61. PMCID: PMC3307194 DOI: 10.1542/peds.2011-0351
- Ing C, DiMaggio C, Whitehouse A, Hegarty MK, Brady J, Von Ungern-Sternberg BS, et al. Long-term differences in language and cognitive function after childhood exposure to anesthesia. Pediatrics. 2012;130(3): e476-85. DOI: 1542/peds.2011-3822
- Van Biesen S, Van de Velde M, Rex S. Anesthesia and neurotoxicity in the developing brain: A non-systematic review. Acta Anaesthesiol Belg. 2015;66(3):67–79.
- Jaganath D, Johnson K, Tschudy MM, Topel K, Stackhouse B, Solomon BS. Desirability of Clinic-Based Financial Services in Urban Pediatric Primary Care.. J Pediatr. 2018; 9:285–90. DOI: 1016/j.jpeds.2018.05.055
- Gusev E, Skvortsova VI. Secondary neuroprotection. In: Brain Ischemia. New York: Springerlink; 2003. p. 298.
- Adan RAH, Gispen WH. Melanocortins and the brain: From effects via receptors to drug targets. Eur J Pharmacol. 2000; 405:13–24.
- Ashmarin IP, Stukalov PV. Neurochemistry. Institute of Biological and Medical Chemistry of Russian Scientific Academy Publishing House, Moscow (in Russian). 1996.
- Kaplan AY, Koshelev VB, Nezavibatko VN, Ashmarin IP. Increased resistance to hypoxia affected by the neuropeptide preparation Semax. Fiziol Cheloveka. 1992;18(5):104–7.
- Marino MW, Dunn A, Grail D, Inglese M, Noguchi Y, Richards E, et al. Characterization of tumor necrosis factor-deficient mice. Proc Natl Acad Sci. 1997.
- Peng S, Zhang Y, Wang H, Ren B, Zhang J. Anesthetic ketamine counteracts repetitive mechanical stress-induced learning and memory impairment in developing mice. Mol Biol Rep. 2011;38:4347–51.
- Fredriksson A, Archer T, Alm H, Gordh T, Eriksson P. Neurofunctional deficits and potentiated apoptosis by neonatal NMDA antagonist administration. Behav Brain Res. 2004;153:367–76.
- Wang C, Slikker W. Strategies and experimental models for evaluating anesthetics: Effects on the developing nervous system. In: Anesthesia and Analgesia. 2008. p. 1643–58.
- Yan J, Li YR, Zhang Y, Lu Y, Jiang H. Repeated exposure to anesthetic ketamine can negatively impact neurodevelopment in infants: A prospective preliminary clinical study. J Child Neurol. 2014;29(10):1333–8. DOI: 1177/0883073813517508
- Braun S, Gaza N, Werdehausen R, Hermanns H, Bauer I, Durieux ME, et al. Ketamine induces apoptosis via the mitochondrial pathway in human lymphocytes and neuronal cells. Br J Anaesth. 2010; 105:347–54. DOI: 1093/bja/aeq169
- Murck H. Ketamine, magnesium and major depression: From pharmacology to pathophysiology and back. J Psychiatr Res. 2013; 47:955–65.
- Green SM, Roback MG, Kennedy RM, Krauss B. Clinical practice guideline for emergency department ketamine dissociative sedation: 2011 update. Ann Emerg Med. 2011; 57:449–61.
- Durga P, Yalamanchili V. Basic cellular and molecular mechanisms of anesthetic-induced developmental neurotoxicity: Potential strategies for alleviation. J Neuroanaesth Crit Care. 2016;3(1):015–24.
- Bosnjak JZ, Yan Y, Canfield SY, Muravyeva M, Kikuchi C, Wells CW, et al. Ketamine induces toxicity in human neurons differentiated from embryonic stem cells via mitochondrial apoptosis pathway. Curr Drug Saf. 2012;7(2):106–19.
- Yakovleva EV, Kuzenkov VS, Fedorov VN, Skvortsova VI, Koshelev VB, Gusev EI, et al. In vivo efficiency of Semax in global cerebral ischemia. Byulleten’ Eksp Biol i Meditsiny. 1999;128(8):172–4.