Nahlah F. Makki 1 , Mustafa M. Khalil 2
Background & objective: Coronary artery disease (CAD) remains a major cause of global morbidity and mortality. Among the regulatory peptides involved in vascular homeostasis, apelin and calcitonin gene–related peptide (CGRP) play critical roles in vasodilation, endothelial protection, and cardiac function, highlighting their potential value as diagnostic biomarkers. This study aimed to evaluate the diagnostic utility of serum apelin and CGRP levels in patients with CAD.
Methods: A total of 90 male participants were enrolled in this case–control study, including 45 CAD patients and 45 age-matched healthy controls. Routine biochemical parameters were assessed using standard colorimetric assays, while serum apelin and CGRP levels were measured using ELISA. Anthropometric indices and atherogenic ratios were calculated using standard formulas.
Results: CAD patients demonstrated significantly higher levels of triglycerides, total cholesterol, LDL, VLDL, atherogenic indices (AIP, AC, CRI-I, CRI-II), troponin, CRP, calcium, and body fat percentage, while HDL, magnesium, apelin, and CGRP levels were markedly reduced (P < 0.001). ROC analysis showed that apelin exhibited excellent diagnostic accuracy with an AUC of 0.894 and an optimal cutoff of 2.98 ng/mL (sensitivity: 88.9%; specificity: 82.2%). CGRP showed moderate diagnostic performance, with an AUC of 0.691 and a cutoff of 30.5 pg/mL.
Conclusion: Reduced serum apelin and CGRP levels are strongly associated with CAD, with apelin demonstrating superior diagnostic value compared to CGRP. Apelin may serve as a promising independent biomarker for early CAD detection. Further large-scale studies are recommended to validate its clinical applicability.
Abbreviations: CAD-coronary artery disease; T2DM-type2 diabetes mellitus; ACS- acute coronary syndrome; HDL – high-density lipoprotein; TG - triglyceride; BMI - body mass index; SBP - systolic blood pressure; DBP - diastolic blood pressure; CRP- C-reactive protein; BF%-Body fat percentage; BMP7-Bone Morphogenetic Protein 7; TRPM7-Transient Receptor Potential Melastatin 7.
Keywords: CRI. Apelin. CRP. Troponin. WHtR.
Citation: Makki NF, Khalil MM. Apelin and CGRP as diagnostic biomarkers in coronary artery disease: a case–control study. Anaesth. pain intensive care 2026;30(1):55-61. DOI: 10.35975/apic.v30i1.3102
Received: December 19, 2025; Revised: December 26, 2025; Accepted: January 01, 2026
Coronary artery disease (CAD) remains one of the leading causes of morbidity and mortality worldwide. It results from atherosclerotic narrowing of the coronary arteries and may present clinically as stable angina or acute coronary syndrome (ACS).1,2 ACS encompasses unstable angina, ST-segment elevation myocardial infarction (STEMI), and non-ST-segment elevation myocardial infarction (NSTEMI). Although the terms ACS, coronary heart disease (CHD), and CAD are often used interchangeably, they are not synonymous. ACS is a subcategory of CAD, while CHD is generally considered a consequence of CAD progression.3 Advances in noninvasive coronary imaging have significantly improved the detection of subclinical disease.4 Nevertheless, despite improvements in diagnosis and management, the molecular mechanisms underlying CAD remain inadequately understood, emphasizing the need to identify novel biomarkers and therapeutic targets.5Among the molecules of emerging interest is apelin, a small adipokine primarily secreted by adipose tissue but also expressed in the brain, heart, lungs, kidneys, and stomach.6-9 As the endogenous ligand of the APJ receptor, apelin plays essential roles in regulating blood pressure, fluid balance, cardiac contractility, angiogenesis, and energy homeostasis. Dysregulation of the apelin–APJ signaling pathway has been linked to several pathological conditions, including heart failure, obesity, and diabetes.9Another key mediator is calcitonin gene-related peptide (CGRP), a potent endogenous vasodilator widely distributed throughout the cardiovascular and nervous systems. CGRP is released predominantly from perivascular sensory nerves and acts through G-protein–coupled receptor complexes, increasing cyclic adenosine monophosphate (cAMP) and nitric oxide production to promote vascular smooth muscle relaxation. In addition to its vasodilatory effects, CGRP contributes to blood pressure regulation, suppression of vascular inflammation, and preservation of endothelial function. Emerging evidence suggests that CGRP may exert a compensatory protective role in conditions characterized by vascular dysfunction, such as hypertension, atherosclerosis, and ischemic heart disease.10-12 These attributes highlight CGRP as a potential biomarker and therapeutic target in cardiovascular disorders.Therefore, this study aimed to assess serum apelin and CGRP levels in patients with CAD compared with healthy controls, to explore their potential roles as circulating biomarkers and their relevance as possible therapeutic targets.
2.1. Sampling
A 5 mL venous blood sample was obtained from each participant following a 12-hour overnight fast. One milliliter of the sample was placed into an EDTA tube for HbA1c measurement. The remaining 4 mL was transferred into a gel-separator tube, incubated at 37 °C for 10 minutes to allow clot formation, and subsequently centrifuged at 3000 rpm (~1000 × g) for 10–15 minutes to separate the serum. The resulting serum was aliquoted into sterile Eppendorf tubes and stored at −80 °C until further analysis.
2.2. Study design
A case–control study was conducted at Al-Sadder Teaching Hospital and the Endocrinology Centre. The study enrolled 45 male patients diagnosed with coronary
artery disease (CAD) and 45 age-matched healthy male controls, aged 45–65 years. Sample collection took place from November 2023 to the end of April 2024. Exclusion criteria included obesity, cancer, thyroid dysfunction, a history of liver cirrhosis, myocardial infarction within the past 1–4 weeks, recent unstable angina or acute coronary syndrome (ACS), severe heart failure, significant valvular or congenital heart disease, active infection or inflammatory disorders, and severe renal or hepatic impairment. All participants provided written informed consent before enrollment. The study protocol was approved by the Institutional Review Board (IRB) of the University of Kufa (IRB reference number: 7547 R/2023). The IRB adheres to international guidelines for the protection of human research participants and complies with the principles of the Declaration of Helsinki.
2.3. Methods
The levels of Apelin and CGRP were determined using the double-antibody sandwich ELISA technique (BioVendor, United States). Serum CRP levels were measured using a commercial ELISA kit (Demeditec, Germany). Lipid profiles, including total cholesterol (T.C.), triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C), were measured using standard enzymatic methods (Spinreact, Spain), as well as, magnesium and calcium concentrations were determined using kits from Biolabo (France). High-sensitivity troponin (hs-Tn) levels were measured using the VIDAS instrument (BioMerieux, France). Low-density lipoprotein cholesterol (LDL-C), very low-density lipoprotein cholesterol (VLDL-C) are calculated via equations.13,14 The atherogenic index of plasma (AIP) was calculated as: AIP=LOG(TG/(HDL_C )).15 The atherogenic coefficient (AC) was determined using: AC=(TC- HDL_C)/HDL_C.16 Castelli’s Risk Indexes (CRI-I and CRI-II), also referred to as cardiac risk indexes, were calculated using:
CRI_I=TC/HDL_C, CRI-II=LDL-C/HDL_C.17 Body mass index (BMI), waist-to-hip ratio (WHR), and waist-to-height ratio (WHtR) were calculated mathematically.18-20 Whereas the formula of body fat percentage (BF%) is as follows: 1.2 x BMI+0.23 x Age-10.8 x Gender-5.4, where sex = ‘1’ for men and ‘0’ for women.21
2.4. Statistical analysis
Data were entered and managed using Microsoft Excel, and statistical analyses were carried out using SPSS version 25. The Kolmogorov–Smirnov test was used to assess the normality of the variables. Quantitative data are presented as means ± standard deviations. Continuous variables were compared using the independent samples t-test. Pearson’s correlation coefficients and linear regression analyses were employed to evaluate associations between variables. The receiver operating characteristic (ROC) curve was applied to determine the cutoff values with optimal sensitivity and specificity for disease diagnosis. Statistical significance was considered at P < 0.05. The sample size was calculated using G*Power version 3.1.9.7. Based on an effect size of 0.3, a significance level of 0.05, a power of 0.8, and two groups in an analysis of covariance, a total sample size of 90 participants was recommended. Accordingly, 90 individuals were recruited, including 45 patients in the experimental group and 45 healthy controls.
The anthropometry results of the CAD and healthy control groups are presented in Table 1.
The analysis revealed no significant differences between the groups in terms of age and body mass index (BMI). However, significant increases were observed in WHR, WHtR, BF%, systolic blood pressure (SBP), and diastolic blood pressure (DBP) in the CAD group compared to controls. The results of the clinical data are displayed in Table 2, including TG, CHOL, HDL, LDL, VLDL, AIP, AC, CRI-I, CRI-II, Troponin, CRP, Ca, Mg, Apelin, and CGRP, which revealed highly significant values (p<0.05).
Receiver operating characteristic (ROC) curve analysis was carryout to evaluate the sensitivity and specificity of the selected biomarkers in distinguishing male CAD patients from healthy controls. The ROC coordinates and corresponding cutoff values, Area Under the Curve (AUC), optimal sensitivity and specificity are presented in Table 3.
The results presented in Table 1 show no significant differences in age or BMI between CAD patients and controls, consistent with previous findings reporting no significant BMI alterations in similar cohorts.22 However, indices of central obesity, including WHR and WHtR, were significantly higher in the CAD group, indicating that visceral adiposity is more closely associated with CAD risk than general obesity. Central fat accumulation contributes to dyslipidemia, hypertension, and atherosclerosis independently of BMI.23 In addition, individuals with CAD frequently exhibit hypertension, insulin resistance, and metabolic syndrome, all of which promote visceral fat deposition, leading to increased waist circumference and WHR. The resulting hyperinsulinemia contributes to elevated blood pressure, primarily through enhanced renal sodium retention and other pressor mechanisms.24 Body fat percentage (BF%) also demonstrated a significant elevation in CAD patients. BF% is a recognized determinant of vascular health, as higher adiposity is linked with increased cardiovascular risk, hypertension, and subclinical vascular inflammation. Importantly, elevated BF% has been shown to predict vascular dysfunction even in individuals with normal BMI, reinforcing its role as a key cardiometabolic risk indicator.25 Table 2 further demonstrates a pronounced reduction in HDL levels, along with significant elevations in total cholesterol, LDL, TG, VLDL, AC, CRI-I, CRI-II, and AIP. These findings align with established evidence identifying dyslipidemia—particularly elevated LDL as a central contributor to CAD pathogenesis. Cholesterol, being hydrophobic, circulates bound to lipoproteins such as HDL, LDL, VLDL, and chylomicrons. Under oxidative stress, LDL undergoes conversion to oxidized LDL (oxLDL), which is subsequently taken up by macrophages to form foam cells, initiating the atherosclerotic cascade.26
Markers of inflammation and myocardial injury also showed notable elevations. Both hs-CRP and hs-troponin were significantly higher in CAD patients. Inflammation plays a central role in atherogenesis, and elevated hs-CRP is known to predict up to a 70% increase in the risk of future cardiovascular events.27
Similarly, in chronic CAD, elevated hs-troponin indicates ongoing low-grade myocardial injury and is a powerful prognostic marker.28 A significant reduction in magnesium (Mg) was observed in the CAD group. Magnesium is crucial for cardiovascular homeostasis through its effects on coagulation, lipid metabolism, nitric oxide release, and synergistic action with antihypertensive agents. Mg deficiency promotes vascular calcification by dysregulating proteins such as osteopontin, Bone Morphogenetic Protein 7 (BMP7), and Transient Receptor Potential Melastatin 7 (TRPM7).29 Additionally, it contributes to chronic inflammation (elevated CRP), mitochondrial dysfunction, reduced ATP synthesis, diminished antioxidant capacity, and increased ROS generation.30 Elevated serum calcium levels, a common feature of cardiovascular disease, further reflect vascular injuryand calcification, correlating strongly with arterial plaque burden.31
As shown in Table 2 and Fig. 1, apelin and CGRP levels were significantly reduced in CAD patients compared to controls. Apelin is a potent vasodilatory and cardioprotective peptide, and decreased circulating levels may reflect impaired vascular function or a diminished compensatory response to ischemia.32 Similarly, CGRP—a neuropeptide with strong
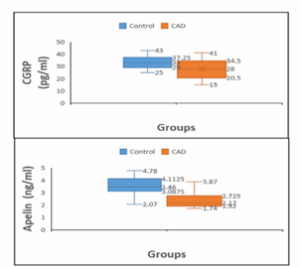
Figure 1: Comparison of serum Apelin and CGRP between CAD men patients and men control group
vasodilatory and anti-inflammatory actions was also reduced, suggesting weakened endogenous vascular protection in CAD patients.33 The diagnostic performance of these biomarkers was further evaluated using ROC analysis. Apelin demonstrated outstanding diagnostic utility, with an AUC of 0.894 and an optimal cutoff of 2.98 ng/mL, yielding high sensitivity (88.9%) and specificity (82.2%). In contrast, CGRP showed only moderate diagnostic accuracy (AUC: 0.691) with lower sensitivity (66.7%) and specificity (55.6%) at a cutoff of 30.5 pg/mL. These findings indicate that apelin is a more robust and reliable biomarker for CAD diagnosis, whereas CGRP alone offers limited discrimination. ROC metrics reinforce that AUC values approaching 1 denote excellent diagnostic ability, whereas values below 0.5 lack diagnostic significance.34
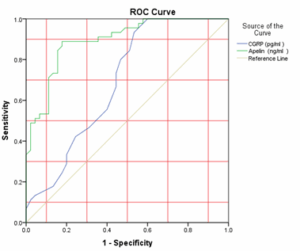
Figure 2: ROC curve analysis of CGRP and Apelin for determining diagnostic cutoff values in CAD patients
This study demonstrated that patients with coronary artery disease exhibit significantly reduced serum levels of apelin, CGRP, and magnesium, alongside marked elevations in lipid profile components (except HDL), atherogenic indices, body fat percentage, and calcium levels. ROC analysis revealed that apelin provides the highest diagnostic accuracy for CAD, with a cutoff value of 2.98 ng/mL yielding sensitivity of 88.9% and specificity of 82.2%, indicating its strong potential as an independent biomarker. Although CGRP levels were also lower in CAD patients, its diagnostic performance was comparatively limited. Overall, apelin appears to be a promising early diagnostic indicator for CAD, and further large-scale, multi-center studies are recommended to validate these findings and explore its prognostic and therapeutic relevance.
6. Data availability
The numerical data generated and analyzed during this study are available from the corresponding author upon reasonable request.
7. Acknowledgement
The authors appreciate all the workers at the Al-Sadder Teaching Hospital.
8. Conflict of interest
The authors declare that they have no conflicts of interest that could have appeared to influence the study reported in this paper.
9. Authors’ contribution
N.F.M. and M.M. Kh designed the study. and wrote the article, providing most of the substantial and intellectual contributions to the work. All the authors reviewed the manuscript.
- Nahlah F. Makki, Department of Chemistry, College of Science, Kufa University, Najaf, Iraq; Email: nahla.almutawalli@uokufa.edu.iq
- Mustafa M. Khalil, Department of Chemical Engineering, College of Engineering, Kufa University, Najaf, Iraq; Email: mustafam.almoslimawi@uokufa.edu.iq
ABSTRACT
Background & objective: Coronary artery disease (CAD) remains a major cause of global morbidity and mortality. Among the regulatory peptides involved in vascular homeostasis, apelin and calcitonin gene–related peptide (CGRP) play critical roles in vasodilation, endothelial protection, and cardiac function, highlighting their potential value as diagnostic biomarkers. This study aimed to evaluate the diagnostic utility of serum apelin and CGRP levels in patients with CAD.
Methods: A total of 90 male participants were enrolled in this case–control study, including 45 CAD patients and 45 age-matched healthy controls. Routine biochemical parameters were assessed using standard colorimetric assays, while serum apelin and CGRP levels were measured using ELISA. Anthropometric indices and atherogenic ratios were calculated using standard formulas.
Results: CAD patients demonstrated significantly higher levels of triglycerides, total cholesterol, LDL, VLDL, atherogenic indices (AIP, AC, CRI-I, CRI-II), troponin, CRP, calcium, and body fat percentage, while HDL, magnesium, apelin, and CGRP levels were markedly reduced (P < 0.001). ROC analysis showed that apelin exhibited excellent diagnostic accuracy with an AUC of 0.894 and an optimal cutoff of 2.98 ng/mL (sensitivity: 88.9%; specificity: 82.2%). CGRP showed moderate diagnostic performance, with an AUC of 0.691 and a cutoff of 30.5 pg/mL.
Conclusion: Reduced serum apelin and CGRP levels are strongly associated with CAD, with apelin demonstrating superior diagnostic value compared to CGRP. Apelin may serve as a promising independent biomarker for early CAD detection. Further large-scale studies are recommended to validate its clinical applicability.
Abbreviations: CAD-coronary artery disease; T2DM-type2 diabetes mellitus; ACS- acute coronary syndrome; HDL – high-density lipoprotein; TG - triglyceride; BMI - body mass index; SBP - systolic blood pressure; DBP - diastolic blood pressure; CRP- C-reactive protein; BF%-Body fat percentage; BMP7-Bone Morphogenetic Protein 7; TRPM7-Transient Receptor Potential Melastatin 7.
Keywords: CRI. Apelin. CRP. Troponin. WHtR.
Citation: Makki NF, Khalil MM. Apelin and CGRP as diagnostic biomarkers in coronary artery disease: a case–control study. Anaesth. pain intensive care 2026;30(1):55-61. DOI: 10.35975/apic.v30i1.3102
Received: December 19, 2025; Revised: December 26, 2025; Accepted: January 01, 2026
1. INTRODUCTION
Coronary artery disease (CAD) remains one of the leading causes of morbidity and mortality worldwide. It results from atherosclerotic narrowing of the coronary arteries and may present clinically as stable angina or acute coronary syndrome (ACS).1,2 ACS encompasses unstable angina, ST-segment elevation myocardial infarction (STEMI), and non-ST-segment elevation myocardial infarction (NSTEMI). Although the terms ACS, coronary heart disease (CHD), and CAD are often used interchangeably, they are not synonymous. ACS is a subcategory of CAD, while CHD is generally considered a consequence of CAD progression.3 Advances in noninvasive coronary imaging have significantly improved the detection of subclinical disease.4 Nevertheless, despite improvements in diagnosis and management, the molecular mechanisms underlying CAD remain inadequately understood, emphasizing the need to identify novel biomarkers and therapeutic targets.5Among the molecules of emerging interest is apelin, a small adipokine primarily secreted by adipose tissue but also expressed in the brain, heart, lungs, kidneys, and stomach.6-9 As the endogenous ligand of the APJ receptor, apelin plays essential roles in regulating blood pressure, fluid balance, cardiac contractility, angiogenesis, and energy homeostasis. Dysregulation of the apelin–APJ signaling pathway has been linked to several pathological conditions, including heart failure, obesity, and diabetes.9Another key mediator is calcitonin gene-related peptide (CGRP), a potent endogenous vasodilator widely distributed throughout the cardiovascular and nervous systems. CGRP is released predominantly from perivascular sensory nerves and acts through G-protein–coupled receptor complexes, increasing cyclic adenosine monophosphate (cAMP) and nitric oxide production to promote vascular smooth muscle relaxation. In addition to its vasodilatory effects, CGRP contributes to blood pressure regulation, suppression of vascular inflammation, and preservation of endothelial function. Emerging evidence suggests that CGRP may exert a compensatory protective role in conditions characterized by vascular dysfunction, such as hypertension, atherosclerosis, and ischemic heart disease.10-12 These attributes highlight CGRP as a potential biomarker and therapeutic target in cardiovascular disorders.Therefore, this study aimed to assess serum apelin and CGRP levels in patients with CAD compared with healthy controls, to explore their potential roles as circulating biomarkers and their relevance as possible therapeutic targets.
2. METHODOLOGY
2.1. Sampling
A 5 mL venous blood sample was obtained from each participant following a 12-hour overnight fast. One milliliter of the sample was placed into an EDTA tube for HbA1c measurement. The remaining 4 mL was transferred into a gel-separator tube, incubated at 37 °C for 10 minutes to allow clot formation, and subsequently centrifuged at 3000 rpm (~1000 × g) for 10–15 minutes to separate the serum. The resulting serum was aliquoted into sterile Eppendorf tubes and stored at −80 °C until further analysis.
2.2. Study design
A case–control study was conducted at Al-Sadder Teaching Hospital and the Endocrinology Centre. The study enrolled 45 male patients diagnosed with coronary
artery disease (CAD) and 45 age-matched healthy male controls, aged 45–65 years. Sample collection took place from November 2023 to the end of April 2024. Exclusion criteria included obesity, cancer, thyroid dysfunction, a history of liver cirrhosis, myocardial infarction within the past 1–4 weeks, recent unstable angina or acute coronary syndrome (ACS), severe heart failure, significant valvular or congenital heart disease, active infection or inflammatory disorders, and severe renal or hepatic impairment. All participants provided written informed consent before enrollment. The study protocol was approved by the Institutional Review Board (IRB) of the University of Kufa (IRB reference number: 7547 R/2023). The IRB adheres to international guidelines for the protection of human research participants and complies with the principles of the Declaration of Helsinki.
2.3. Methods
The levels of Apelin and CGRP were determined using the double-antibody sandwich ELISA technique (BioVendor, United States). Serum CRP levels were measured using a commercial ELISA kit (Demeditec, Germany). Lipid profiles, including total cholesterol (T.C.), triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C), were measured using standard enzymatic methods (Spinreact, Spain), as well as, magnesium and calcium concentrations were determined using kits from Biolabo (France). High-sensitivity troponin (hs-Tn) levels were measured using the VIDAS instrument (BioMerieux, France). Low-density lipoprotein cholesterol (LDL-C), very low-density lipoprotein cholesterol (VLDL-C) are calculated via equations.13,14 The atherogenic index of plasma (AIP) was calculated as: AIP=LOG(TG/(HDL_C )).15 The atherogenic coefficient (AC) was determined using: AC=(TC- HDL_C)/HDL_C.16 Castelli’s Risk Indexes (CRI-I and CRI-II), also referred to as cardiac risk indexes, were calculated using:
CRI_I=TC/HDL_C, CRI-II=LDL-C/HDL_C.17 Body mass index (BMI), waist-to-hip ratio (WHR), and waist-to-height ratio (WHtR) were calculated mathematically.18-20 Whereas the formula of body fat percentage (BF%) is as follows: 1.2 x BMI+0.23 x Age-10.8 x Gender-5.4, where sex = ‘1’ for men and ‘0’ for women.21
2.4. Statistical analysis
Data were entered and managed using Microsoft Excel, and statistical analyses were carried out using SPSS version 25. The Kolmogorov–Smirnov test was used to assess the normality of the variables. Quantitative data are presented as means ± standard deviations. Continuous variables were compared using the independent samples t-test. Pearson’s correlation coefficients and linear regression analyses were employed to evaluate associations between variables. The receiver operating characteristic (ROC) curve was applied to determine the cutoff values with optimal sensitivity and specificity for disease diagnosis. Statistical significance was considered at P < 0.05. The sample size was calculated using G*Power version 3.1.9.7. Based on an effect size of 0.3, a significance level of 0.05, a power of 0.8, and two groups in an analysis of covariance, a total sample size of 90 participants was recommended. Accordingly, 90 individuals were recruited, including 45 patients in the experimental group and 45 healthy controls.
3. RESULTS
The anthropometry results of the CAD and healthy control groups are presented in Table 1.
| Table1: Comparison of anthropometric measurements between coronary artery disease (CAD) patients and healthy controls. | |||
| Parameters | Control
(n = 45) |
CAD
(n = 45) |
P-value |
| Age (years) | 60.42 ± 3.48 | 60.91 ± 3.25 | 0.07 |
| Duration of Disease (years) |
- | 5.13 ± 2.04 | - |
| Weight (g) | 76.11 ± 6.44 | 82.89 ± 5.11 | < 0.001 |
| BMI (kg/m2) | 25.80 ± 2.22 | 26.37 ± 2.24 | 0.06 |
| WHR | 0.915 ± 0.025 | 0.944 ± 0.018 | < 0.001 |
| WHtR | 0.41 ± 0.038 | 0.50 ± 0.03 | < 0.05 |
| BF% | 38.70 ± 2.85 | 40.49 ± 2.63 | < 0.001 |
| SBP (mmHg) | 120.29 ± 1.19 | 156.71 ± 5.78 | < 0.001 |
| DBP (mmHg) | 80.76 ± 1.38 | 95.33 ± 4.30 | < 0.001 |
The analysis revealed no significant differences between the groups in terms of age and body mass index (BMI). However, significant increases were observed in WHR, WHtR, BF%, systolic blood pressure (SBP), and diastolic blood pressure (DBP) in the CAD group compared to controls. The results of the clinical data are displayed in Table 2, including TG, CHOL, HDL, LDL, VLDL, AIP, AC, CRI-I, CRI-II, Troponin, CRP, Ca, Mg, Apelin, and CGRP, which revealed highly significant values (p<0.05).
| Table 2: Comparison of the TG, TC, HDL, LDL, VLDL, AIP, AC, CRI-I, CRI-II, Troponin, CRP, Ca, Mg, Apelin, and CGRP in CAD patients and healthy controls. | ||||
| Parameters | Control
(n = 45) |
CAD
(n = 45) |
P-value | |
| TG (mg/dl) | 136.37 ± 8.77 | 279.62 ± 18.11 | < 0.001 | |
| TC (mg/dl) | 144.91 ± 7.27 | 230.24 ± 11.07 | < 0.001 | |
| HDL (mg/dl) | 54.36 ± 3.55 | 23.56 ± 3.83 | < 0.001 | |
| LDL (mg/dl) | 63.28 ± 8.11 | 114.76 ± 11.72 | < 0.001 | |
| VLDL (mg/dl) | 27.27 ± 1.75 | 55.92 ± 3.62 | < 0.001 | |
| AC | 1.67 ± 0.21 | 7.45 ± 1.45 | < 0.001 | |
| AIP | 0.39 ± 0.04 | 1.07 ± 0.07 | < 0.001 | |
| CRI-I | 2.67 ± 0.21 | 8.45 ± 1.45 | < 0.001 | |
| CRI-II | 1.17 ± 0.19 | 5.02 ± 1.09 | < 0.001 | |
| H.S Troponin (ng/ml) | 0.02 ± 0.01 | 2.45 ± 0.48 | < 0.001 | |
| CRP(ng/ml) | 898.80 ± 41.9 | 4409.95 ± 540.2 | < 0.001 | |
| Ca (mg/dl) | 9.50 ± 0.44 | 10.55 ± 0.07 | < 0.001 | |
| Mg (mg/dl) | 2.04 ± 0.15 | 1.39 ± 0.13 | < 0.001 | |
| Apelin (ng/ml) | 3.50 ± 0.68 | 1.89 ± 0.39 | < 0.001 | |
| CGRP (pg/ml) | 33.31 ± 5.08 | 26.96 ± 6.83 | < 0.001 | |
| P < 0.05 considered as significant | ||||
Receiver operating characteristic (ROC) curve analysis was carryout to evaluate the sensitivity and specificity of the selected biomarkers in distinguishing male CAD patients from healthy controls. The ROC coordinates and corresponding cutoff values, Area Under the Curve (AUC), optimal sensitivity and specificity are presented in Table 3.
| Table 3: Receiver operating characteristic (ROC) curves of CGRP and Apelin in the coronary artery (CAD) patients in the control groups. CI: Confidence interval. | |||
| Test Result Variable(s) | CGRP (pg/ml) | Apelin (ng/ml) | |
| AUC | 0.691 | 0.894 | |
| Cut-Off | ˂ 30.5 | ˂ 2.98 | |
| p-value | 0.002 | 0.000 | |
| Sensitivity % | 66.7 | 88.9 | |
| Specificity % | 55.6 | 82.2 | |
| Asymptotic 95% Confidence Interval (CI) |
Lower Bound | 0.579 | 0.829 |
| Upper Bound | 0.802 | 0.958 | |
4. DISCUSSION
The results presented in Table 1 show no significant differences in age or BMI between CAD patients and controls, consistent with previous findings reporting no significant BMI alterations in similar cohorts.22 However, indices of central obesity, including WHR and WHtR, were significantly higher in the CAD group, indicating that visceral adiposity is more closely associated with CAD risk than general obesity. Central fat accumulation contributes to dyslipidemia, hypertension, and atherosclerosis independently of BMI.23 In addition, individuals with CAD frequently exhibit hypertension, insulin resistance, and metabolic syndrome, all of which promote visceral fat deposition, leading to increased waist circumference and WHR. The resulting hyperinsulinemia contributes to elevated blood pressure, primarily through enhanced renal sodium retention and other pressor mechanisms.24 Body fat percentage (BF%) also demonstrated a significant elevation in CAD patients. BF% is a recognized determinant of vascular health, as higher adiposity is linked with increased cardiovascular risk, hypertension, and subclinical vascular inflammation. Importantly, elevated BF% has been shown to predict vascular dysfunction even in individuals with normal BMI, reinforcing its role as a key cardiometabolic risk indicator.25 Table 2 further demonstrates a pronounced reduction in HDL levels, along with significant elevations in total cholesterol, LDL, TG, VLDL, AC, CRI-I, CRI-II, and AIP. These findings align with established evidence identifying dyslipidemia—particularly elevated LDL as a central contributor to CAD pathogenesis. Cholesterol, being hydrophobic, circulates bound to lipoproteins such as HDL, LDL, VLDL, and chylomicrons. Under oxidative stress, LDL undergoes conversion to oxidized LDL (oxLDL), which is subsequently taken up by macrophages to form foam cells, initiating the atherosclerotic cascade.26
Markers of inflammation and myocardial injury also showed notable elevations. Both hs-CRP and hs-troponin were significantly higher in CAD patients. Inflammation plays a central role in atherogenesis, and elevated hs-CRP is known to predict up to a 70% increase in the risk of future cardiovascular events.27
Similarly, in chronic CAD, elevated hs-troponin indicates ongoing low-grade myocardial injury and is a powerful prognostic marker.28 A significant reduction in magnesium (Mg) was observed in the CAD group. Magnesium is crucial for cardiovascular homeostasis through its effects on coagulation, lipid metabolism, nitric oxide release, and synergistic action with antihypertensive agents. Mg deficiency promotes vascular calcification by dysregulating proteins such as osteopontin, Bone Morphogenetic Protein 7 (BMP7), and Transient Receptor Potential Melastatin 7 (TRPM7).29 Additionally, it contributes to chronic inflammation (elevated CRP), mitochondrial dysfunction, reduced ATP synthesis, diminished antioxidant capacity, and increased ROS generation.30 Elevated serum calcium levels, a common feature of cardiovascular disease, further reflect vascular injuryand calcification, correlating strongly with arterial plaque burden.31
As shown in Table 2 and Fig. 1, apelin and CGRP levels were significantly reduced in CAD patients compared to controls. Apelin is a potent vasodilatory and cardioprotective peptide, and decreased circulating levels may reflect impaired vascular function or a diminished compensatory response to ischemia.32 Similarly, CGRP—a neuropeptide with strong

Figure 1: Comparison of serum Apelin and CGRP between CAD men patients and men control group
vasodilatory and anti-inflammatory actions was also reduced, suggesting weakened endogenous vascular protection in CAD patients.33 The diagnostic performance of these biomarkers was further evaluated using ROC analysis. Apelin demonstrated outstanding diagnostic utility, with an AUC of 0.894 and an optimal cutoff of 2.98 ng/mL, yielding high sensitivity (88.9%) and specificity (82.2%). In contrast, CGRP showed only moderate diagnostic accuracy (AUC: 0.691) with lower sensitivity (66.7%) and specificity (55.6%) at a cutoff of 30.5 pg/mL. These findings indicate that apelin is a more robust and reliable biomarker for CAD diagnosis, whereas CGRP alone offers limited discrimination. ROC metrics reinforce that AUC values approaching 1 denote excellent diagnostic ability, whereas values below 0.5 lack diagnostic significance.34

Figure 2: ROC curve analysis of CGRP and Apelin for determining diagnostic cutoff values in CAD patients
5. CONCLUSION
This study demonstrated that patients with coronary artery disease exhibit significantly reduced serum levels of apelin, CGRP, and magnesium, alongside marked elevations in lipid profile components (except HDL), atherogenic indices, body fat percentage, and calcium levels. ROC analysis revealed that apelin provides the highest diagnostic accuracy for CAD, with a cutoff value of 2.98 ng/mL yielding sensitivity of 88.9% and specificity of 82.2%, indicating its strong potential as an independent biomarker. Although CGRP levels were also lower in CAD patients, its diagnostic performance was comparatively limited. Overall, apelin appears to be a promising early diagnostic indicator for CAD, and further large-scale, multi-center studies are recommended to validate these findings and explore its prognostic and therapeutic relevance.
6. Data availability
The numerical data generated and analyzed during this study are available from the corresponding author upon reasonable request.
7. Acknowledgement
The authors appreciate all the workers at the Al-Sadder Teaching Hospital.
8. Conflict of interest
The authors declare that they have no conflicts of interest that could have appeared to influence the study reported in this paper.
9. Authors’ contribution
N.F.M. and M.M. Kh designed the study. and wrote the article, providing most of the substantial and intellectual contributions to the work. All the authors reviewed the manuscript.
10. REFERENCES
- Gherasie, F.A., Popescu, M.R. and Bartos. Acute coronary syndrome: disparities of pathophysiology and mortality with and without peripheral artery disease.2023;13(6):944. DOI: 10.3390/jpm13060944
- Bubulytė, S. and Maneikienė, V.V. Acute coronary syndromes–ST-elevation myocardial infarction and non-ST elevation myocardial infarction–literature review. Medicinos mokslai, 2024;12(3):118-124. DOI: 10.53453/ms.2024.5.13
- De Rosa, S., Sabatino, J., Di Salvo, G., Torella, D. and Di Mario, C. Coronary artery disease in adults with congenital heart disease. International Journal of Cardiology Congenital Heart Disease, 2023;13,:100466. DOI: 10.1016/j.ijcchd.2023.100466
- Tolu‐Akinnawo, O.Z., Ezekwueme, F., Omolayo, O., Batheja, S. and Awoyemi, T. Advancements in artificial intelligence in noninvasive cardiac imaging: A comprehensive review. Clinical Cardiology.2025;48(1):70087. DOI: 10.1002/clc.70087
- Shi, X., Wang, X., Yao, W., Shi, D., Shao, X., Lu, Z., Chai, Y., Song, J., Tang, W. and Wang, X. Mechanism insights and therapeutic intervention of tumor metastasis: latest developments and perspectives. Signal transduction and targeted therapy, 2024;9(1): 192. DOI: 10.1038/s41392-024-01885-2
- Castan-Laurell, I., Dray, C. and Valet, P. The therapeutic potentials of apelin in obesity-associated diseases. Molecular and Cellular Endocrinology.2021;529:111278. DOI: 10.1016/j. mce.2021.11 12787.
- Hu, G., Wang, Z., Zhang, R., Sun, W. and Chen, X. The role of apelin/apelin receptor in energy metabolism and water homeostasis: a comprehensive narrative review. Frontiers in physiology.2021;12:632886. DOI: 10.3389/fphys.2021.632886
- Gergics, M., Pham-Dobor, G., Horváth-Szalai, Z., Kőszegi, T., Mezősi, E. and Bajnok, L. Secondary hormonal alterations in short-term severe hypothyroidism; in the focus: Apelin and copeptin. Frontiers in Endocrinology. 2022;13:981891. DOI: 10.3389/fendo.2022.981891
- Li, A., Zhao, Q., Chen, L. and Li, Z. Apelin/APJ system: an emerging therapeutic target for neurological diseases. Molecular Biology Reports.2023;50(2):1639-1653. DOI: 10.1007/s11033-022-08075-910
- Al-Hassany, L., Boucherie, D.M., Creeney, H., van Drie, R.W., Farham, F., Favaretto, S., Gollion, C., Grangeon, L., Lyons, H., Marschollek, K. and Onan, D. Future targets for migraine treatment beyond CGRP. The journal of headache and pain. 2023;24(1):76. DOI:10.1186/s10194-023-01567-411
- Eberhardt, M., Dux, M., Namer, B., Miljkovic, J., Cordasic, N., Will, C., Kichko, T.I., De La Roche, J., Fischer, M., Suárez, S.A. and Bikiel, D. H2S and NO cooperatively regulate vascular tone by activating a neuroendocrine HNO–TRPA1–CGRP signalling pathway. Nature communications. 2014;5(1):4381. DOI: 10.1038/ncomms538112
- Argunhan, F., Thapa, D., Aubdool, A.A., Carlini, E., Arkless, K., Hendrikse, E.R., de Sousa Valente, J., Kodji, X., Barrett, B., Ricciardi, C.A. and Gnudi, L. Calcitonin gene-related peptide protects against cardiovascular dysfunction independently of nitric oxide in vivo. Hypertension. 2021;77(4):1178-1190.
- DOI: 10.1161/ HYPERTENSIONAHA.120.14851
- Lütjohann, D., Klör, H.U. and Stellaard, F. Measurement of serum low density lipoprotein cholesterol and triglyceride-rich remnant cholesterol as independent predictors of atherosclerotic cardiovascular disease: possibilities and limitations. Nutrients. 2023; 15(9):2202. DOI:10.3390/nu1509220211.
- Sampson, M., Wolska, A., Meeusen, J.W., Donato, L.J., Jaffe, A.S. and Remaley, A.T. Identification of dysbetalipoproteinemia by an enhanced Sampson-NIH equation for very low-density lipoprotein-cholesterol. Frontiers in Genetics. 2022;13:935257. DOI:10.3389/fgene. 202 2.93525712.
- Li, Y., Feng, Y., Li, S., Ma, Y., Lin, J., Wan, J. and Zhao, M. The atherogenic index of plasma (AIP) is a predictor for the severity of coronary artery disease. Frontiers in Cardiovascular Medicine. 2023;10:1140215.DOI:10.3389/fcvm.2023.1140215 13.
- Agu, P.U., Egbugara, M.N., Ogboi, J.S., Ajah, L.O., Nwagha, U.I., Ugwu, E.O. and Ezugwu, E.C. Atherogenic index, cardiovascular risk ratio, and atherogenic coefficient as risk factors for cardiovascular disease in pre-eclampsia in Southeast Nigeria: A Cross-Sectional study. Nigerian Journal of Clinical Practice.2024;27(2):221-227. DOI:10.4103/njcp.njcp_633_23
- Drwiła, D., Rostoff, P., Nessler, J. and Konduracka, E. Prognostic value of non-traditional lipid parameters: Castelli Risk Index I, Castelli Risk Index II, and triglycerides to high-density lipoprotein cholesterol ratio among patients with non-ST-segment elevation myocardial infarction during 1-year follow-up. Kardiologiia. 2022;62(9):60-66. DOI: 10.18087/cardio.2022.9.n2037
- Consultation, W.H.O. Obesity: preventing and managing the global epidemic. World Health Organ Tech Rep Ser. 2000;894:1-253.
- Consultation, W.E. Waist circumference and waist-hip ratio. Report of a WHO Expert Consultation. Geneva: World Health Organization. 2008:8-11.
- Ashwell, M., Gunn, P. and Gibson, S. Waist‐to‐height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: systematic review and meta‐analysis. Obesity reviews. 2012;13(3):275-286. DOI:10.1111/j.1467-789X.2011.00952.x.
- Macek, P., Biskup, M., Terek-Derszniak, M., Stachura, M., Krol, H., Gozdz, S. and Zak, M. Optimal body fat percentage cut-off values in predicting the obesity-related cardiovascular risk factors: a cross-sectional cohort study. Diabetes, metabolic syndrome and obesity.2020:1587-1597. DOI: 10.2147/DMSO.S248038
- Makki, N.F., Mahmmad, S.B. and Ressen, T.F. Assessment of osteoprotegerin as monitor diabetic nephropathy. 2022 DOI: 10.22034/ecc.2022.321025.1283
- Lu, Y., Liu, S., Qiao, Y., Li, G., Wu, Y. and Ke, C., 2021. Waist-to-height ratio, waist circumference, body mass index, waist divided by height0. 5 and the risk of cardiometabolic multimorbidity: a national longitudinal cohort study. Nutrition, Metabolism and Cardiovascular Diseases.2022;31(9):2644-2651. DOI: 10.1016/j.numecd.2021.06.021
- Ertuglu, L.A., Elijovich, F., Laffer, C.L. and Kirabo, A. Salt-sensitivity of blood pressure and insulin resistance. Frontiers in Physiology2021;12:793924. DOI: 10.3389/fphys.2021.793924
- Ohori, K., Yano, T., Katano, S., Kouzu, H., Honma, S., Shimomura, K., Inoue, T., Takamura, Y., Nagaoka, R., Koyama, M. and Nagano, N. High percent body fat mass predicts lower risk of cardiac events in patients with heart failure: an explanation of the obesity paradox. BMC geriatrics.2021;21(1):16.. DOI: 10.1186/s12877-020-01950-9
- Gaggini, M., Gorini, F. and Vassalle, C. Lipids in atherosclerosis: pathophysiology and the role of calculated lipid indices in assessing cardiovascular risk in patients with hyperlipidemia. International journal of molecular sciences. 2022;24(1):75. DOI: 10.3390/ijms24010075 .
- Denegri, A. and Boriani, G. High sensitivity C-reactive protein (hsCRP) and its implications in cardiovascular outcomes. Current Pharmaceutical Design. 2021;27(2),263-275. DOI: 10.2174/1381612826666200717090334
- Wereski, R., Adamson, P., Shek Daud, N.S., McDermott, M., Taggart, C., Bularga, A., Kimenai, D.M., Lowry, M.T., Tuck, C., Anand, A. and Lowe, D.J. High-sensitivity cardiac troponin for risk assessment in patients with chronic coronary artery disease. Journal of the American College of Cardiology. 2023;82(6):473-485.. DOI: 10.1016/j.jacc.2023.05.046
- Montezano, A.C., Zimmerman, D., Yusuf, H., Burger, D., Chignalia, A.Z., Wadhera, V., van Leeuwen, F.N. and Touyz, R.M. Vascular smooth muscle cell differentiation to an osteogenic phenotype involves TRPM7 modulation by magnesium. Hypertension. 2010;56(3):453-462.DOI: 10.1161/HYPERTENSIONAHA.110.152058
- Liu, M. and Dudley Jr, S.C. Magnesium, oxidative stress, inflammation, and cardiovascular disease. Antioxidants. 2020;9(10):907. DOI: 10.3390/antiox9100907
- Arancibia-Hernández, Y.L., Hernández-Cruz, E.Y. and Pedraza-Chaverri, J. Magnesium (Mg2+) deficiency, not well-recognized non-infectious pandemic: origin and consequence of chronic inflammatory and oxidative stress-associated diseases. Cell Physiol Biochem. 2023;57(S1):1-23.. DOI: 10.33594/000000603
- Chapman, F.A., Maguire, J.J., Newby, D.E., Davenport, A.P. and Dhaun, N. Targeting the apelin system for the treatment of cardiovascular diseases. Cardiovascular Research. 2023;119(17):2683-2696. DOI: 10.1093/cvr/cvad171
- Argunhan, F. and Brain, S.D. The vascular-dependent and-independent actions of calcitonin gene-related peptide in cardiovascular disease. Frontiers in Physiology. 2022;13:833645.DOI: 10.3389/fphys.2022.833645
- Gneiting, T. and Walz, E.M. Receiver operating characteristic (ROC) movies, universal ROC (UROC) curves, and coefficient of predictive ability (CPA). Machine Learning. 2022; 111(8):2769-2797. DOI: 10.1007/s10994-021-06114-3