Hien Van Vo 1 , Quynh Van Nguyen 2 , Thang Xuan Vy 3 , Anh Ngoc Le 4
Authors affiliation:
Background: This study compared the effects of Target-Controlled Infusion (TCI) versus Manually-Controlled Infusion (MCI) of propofol on hemodynamic stability and recovery profiles in this vulnerable cohort, aiming to establish the safer administration method.
Methodology: This was a single-center, non-randomized, prospective, observational study, involving 106 elderly patients (≥ 65 years) (TCI n = 53, MCI n = 53). TCI used the Schnider model for propofol titration, while MCI involved slow bolus injection. The primary endpoints were the incidence of post-induction hypotension (MAP <65 mmHg or ≥30% reduction from baseline) and the need for vasopressor (phenylephrine) intervention. Secondary outcomes included intra-induction hemodynamic trends and key recovery milestones (spontaneous respiration, verbal command, LMA removal, and PACU discharge).
Results: Despite similar propofol induction doses, the TCI group showed a significantly longer induction time (140.49 s vs. 52.81 s, 0.001). TCI provided superior hemodynamic stability, resulting in a substantially lower incidence of post-induction hypotension (22.6% vs. 47.2%, p = 0.008) and a reduced need for phenylephrine (9.4% vs. 34%, p = 0.002). Hemodynamic analysis showed that TCI mitigated both the depth of hypotension post-induction and the degree of reactive hypertension post-LMA insertion. The TCI group achieved all key recovery milestones significantly faster, including time to spontaneous respiration (8.15 min vs. 9.60 min, P < 0.001) and time to PACU discharge (29.21 min vs. 35.43 min, P < 0.001).
Conclusion: In high-risk elderly patients undergoing sacral flap surgery, propofol TCI may improve hemodynamic stability during induction and facilitate recovery compared to MCI. The controlled, gradual titration of TCI is highly effective in minimizing cardiovascular stress and could be considered a preferred technique for propofol administration in frail geriatric surgical populations.
Abbreviation: TCI: Target-Controlled Infusion; MCI: Manually Controlled Infusion; LMA: Laryngeal Mask Airway; MAP: Mean Arterial Blood Pressure; PACU: Post Anaesthesia Care Unit
Keywords: Propofol; Target-Controlled Infusion; TCI; Manual Controlled Infusion; MCI; Elderly Patients; Hypotension; Recovery Profile; Sacral Pressure Ulcer Flap Surgery
Citation: Vo HV, Nguyen QV, Vy AN. Target-controlled versus manual-controlled infusion of propofol: comparative hemodynamic stability during induction and recovery profiles in elderly patients undergoing sacral pressure ulcer flap surgery-a non-randomized prospective observational study. Anaesth. pain intensive care 2025;30(1):47-54. DOI: 10.35975/apic.v30i1.3101
Received: October 15, 2025; Revised: November 06, 2024; Accepted: November 06, 2025
Anesthesia induction in elderly patients presents significant challenges due to age-related physiological changes, comorbidities, and reduced cardiovascular reserve. Such patients are especially prone to hypotension, myocardial ischemia, cerebral hypoperfusion, and other complications when undergoing major surgery.1 Flap surgery for sacral pressure ulcers represents a particularly hemodynamically challenging context. These procedures are typically prolonged and associated with substantial blood loss, large wound areas, and fluid shifts. Moreover, patients are often frail, nutritionally depleted, chronically immobilized, and affected by systemic inflammation, all of which further compromise cardiovascular stability and increase anesthetic risk.
Propofol remains a commonly used agent for induction of general anesthesia because of its rapid onset and favorable recovery profile. However, its hemodynamic effect such as vasodilation, decreased systemic vascular resistance, and myocardial depression can be profound in the elderly.2 The induction dose of propofol required for loss of consciousness decreases with age, approximately by 0.1 mg.kg-1 for every 10-year increase, necessitating precise dosing strategies to avoid complications such as profound hypotension.3 The method of administration of propofol contributes a critical role in hemodynamic response: Manual Controlled Infusion (MCI) tend to cause rapid rises in plasma concentration, producing vasodilation, reduction in systemic vascular resistance, decreased preload, and myocardial depression. In contrast, Target-Controlled Infusion (TCI) employs pharmacokinetic/pharmacodynamic (PK/PD) models (e.g., Schneider, Marsh) to titrate propofol gradually toward predefined effect-site concentration, which may provide a more stable induction profile. A recent randomized controlled trial demonstrated that TCI induction significantly reduced the incidence of post-induction hypotension compared to manual induction (13% vs. 34%)4 consistent with other studies showing lower hypotension rates with TCI in various populations.5
However, evidence concerning its superiority in dedicated elderly cohorts remains conflicting; for instance, at least one comparative study in elderly patients found no significant differences in hemodynamic stability or the need for vasoactive drugs between TCI and MCI.6 Moreover, prior comparative studies have often emphasized intra-induction hemodynamics while overlooking recovery quality - an increasingly important metric in geriatric anesthesia.7
To address these gaps, we conducted a prospective observational study comparing TCI and MCI propofol induction in elderly patients undergoing flap surgery for sacral pressure ulcers. The primary objective was to evaluate their hemodynamic impact during induction, including blood pressure trends, heart rate responses, and the need for corrective interventions such as vasopressors or treatment of bradycardia. In addition, the study assessed key recovery milestones (time to spontaneous respiration, eye opening, verbal response, and extubation) to characterize the overall induction–recovery profile associated with each method.
By evaluating both the induction and emergence phases, this observational design aimed to offer preliminary insights into the hemodynamic safety and recovery characteristics of TCI versus MCI in a vulnerable geriatric surgical population.
This single-center, non-randomized, prospective, observational study was conducted at the Le Huu Trac National Burns Hospital (Vietnam Military Medical University), the central specialized burn care hospital in Hanoi, Vietnam, between August 2024 and May 2025.
The study protocol underwent institutional review and received official approval from the Medical Research Committee of the Le Huu Trac National Burns Hospital, Vietnam Military Medical University (Official Dispatch No. 329/BVB- KHTH dated March 28, 2024). The research strictly adhered to the ethical principles outlined in the Declaration of Helsinki published by the World Medical Association (WMA). Informed written consent was obtained from all participating patients and their families prior to study participation. Patient anonymity and data confidentiality were maintained and stored securely throughout the duration of the study.
Patients aged ≥ 65 years; ASA (American Society of Anesthesiologists) physical status I-III; scheduled for flap transfer surgery for sacral pressure ulcer repair under general anesthesia with LMA. Known allergy to propofol or adjunct medications; severe baseline cognitive impairment; inability to consent; history of malignant hyperthermia; major cardiovascular instability preoperatively; need for alternative airway.
All participants were evaluated to ensure they fulfilled the study’s selection requirements, given information about the anesthesia technique and the medications used in the research, and asked to sign a consent form to take part in the study. Anesthetists were instructed to assign patients to either the TCI or MCI group according to their routine clinical practice, primarily based on the availability of the infusion system and the patient’s cardiovascular tolerance to induction. Importantly, allocation was not influenced by hemodynamic status, comorbidities, or anesthetist preference toward a specific technique.
Baseline characteristics, including hypertension, cardiac disease, diabetes, and ASA physical status, were statistically compared between groups and showed no significant differences (Table 2), thereby minimizing potential selection bias.
A peripheral intravenous line was used to infuse the patients with a 0.9% sodium chloride solution at a rate of 5 mL.min-1. A monitor (Nihon Kohden PVM 2701- Japan) was set up to track standard parameters including non-invasive arterial blood pressure (ABP), heart rate, electrocardiogram (DII), end-tidal CO₂ pressure (etCO₂), oxygen saturation (SpO2), and Bispectral Index (BIS). A slow intravenous injection of 1 mg midazolam was administered to each patient as a pre-anesthetic medication.
All patients received fentanyl 2 µg / kg IV before induction.
TCI group: Propofol was infused by PERFUSOR® SPACE TCI pump (B. Braun, Germany) using the Schneider model with an initial effect-site concentration (Ce) of 1.5 µg/mL; incrementally increased by 0.5 µg/mL until loss of consciousness (LOC), defined by BIS < 60.
MCI group: Propofol was given manually by slow IV bolus, dose 1.0–1.5 mg/kg, administered in 20 mg increments until LOC (BIS < 60).
No neuromuscular blocking agents were given to the patients. An appropriate size Pro-Seal LMA was inserted for perioperative airway management. A/C mode mechanical ventilation (Carestation™ 620 anesthesia machine, GE Healthcare, USA) was used with a tidal volume of 6–8 mL/kg, respiratory rate of 12-16 breaths per minute, and an inspiratory-to-expiratory (I:E) ratio of 1:2.
Anaesthesia was maintained by propofol with either TCI pump (Ce = 2-3.5 µg/mL (TCI group) or a continuous intravenous infusion of propofol at the dose of 8- 10 mg/kg/h (MCI group). The maintenance dose of propofol was adjusted based on mean arterial pressure and the BIS index within the range of 40–60. Additional fentanyl (1-2 µg/kg) was given as need.
Immediately following the final suture, a propofol infusion was also stopped in both groups. "Zero ending time" (E0) was the moment when propofol was stopped; the total amount of propofol taken up to this point was measured. Upon completion of the surgery, the LMA was removed, and intravenous paracetamol (10 mg/kg) was administered for postoperative analgesia.
2.1. Data Collection & Outcomes
Data collected prospectively included:
Occurrence of hypotension: defined as MAP < 65 mmHg or ≥ 30% reduction from baseline at any of the timepoints during induction through 10 minutes post-induction. When hypotension occurred, phenylephrine 100 μg intravenous bolus was administered and repeated as required to restore MAP within 20% of baseline (total dose up to 200 µg).
For recovery characteristic, time was measured from the propofol stoppage time (E0), and the two groups were compared (Table 1).
2.1. Statistical analysis
Continuous variables assessed for normality (Shapiro-Wilk). Normally distributed data expressed as mean ± SD; non-normal as median (IQR). Between-group comparisons with independent t-test or Mann-Whitney U test. Categorical data: Chi-square or Fisher’s exact test. All analyses were performed using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). A two-tailed P < 0.05 was considered statistically significant.
3. RESULTS
During the period of the study, a total of 125 elderly patients scheduled for flap surgery for sacral pressure ulcers were initially considered for inclusion. After applying inclusion and exclusion criteria, 106 patients fulfilled eligibility and were enrolled in the study. The remaining patients were excluded for various reasons (e.g. not meeting criteria, withdrawal of consent, intraoperative cancellation). Of the 106 enrolled, 53 patients were allocated to the TCI group and 53 patients to the MCI group (see Table 1). The values of the variables obtained in the study were statistically analyzed using the Shapiro-Wilk test to ensure normal distribution.
There were no differences between the two groups’ demographics in terms of age, sex, ASA status, history of hypertension, or other comorbidities (Table 2)
Although there was no difference in the amount of propofol for induction (TCI: 88.68 mg; MCI: 92.26 mg; p = 0.11) or propofol induction dose (TCI: 1.72; MCI: 1.78 mg.kg-1, P = 0.16) between the groups, the TCI group’s induction time (the amount of time from the beginning of propofol infusion/injection to a BIS < 60) was noticeably longer than the manual group (140.49 vs. 52.81, respectively; P < 0.001). The total propofol consumption during maintenance was recorded and analyzed; no significant difference was observed between the two groups (P = 0.73).
For LMA insertion, the time to successful insertion was 14.66 ± 2.96 seconds in the TCI group and 15.15 ± 2.79 seconds in the MCI group; this difference was not statistically significant (P = 0.40). First attempt success rates for LMA insertion were 100 % in both groups (53/53).
Table 2 also findings demonstrated that the TCI group’s percentage of hypotensive patients and the rate of phenylephrine required to treat hypotension were lower than those of the MCI group (22.6% vs. 47.2% (P = 0.008) and 9.4% vs. 34% (P = 0.002), respectively).
Hemodynamic changes during anesthesia induction and LMA insertion in both study groups are presented in Table 3 and Figure 1. At the baseline (T0), there were no statistically significant differences in hemodynamic parameters between the two groups (P > 0.05). Following induction of anesthesia (T1), both groups exhibited a decrease in ABP; however, the reduction was significantly greater in the MCI group, with a higher incidence of hypotension compared with the TCI group (P < 0.001). During LMA insertion (T2), stimulation caused a transient increase in blood pressure, which was significantly higher in the MCI group than in the TCI group. Thereafter, hemodynamic parameters in both groups remained within normal limits, although the TCI group demonstrated more stable trends over time.
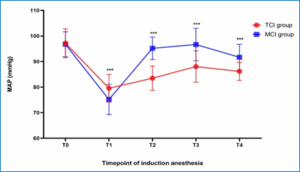
Figure 1: Changes in MAP during induction of anesthesia in the TCI and MCI groups at five predefined timepoints (T0–T4).Values are expressed as mean ± SD.
In terms of recovery profile, Table 4 and Figure 2 shows that the time for the patient to breathe again, the time for the patient to respond to commands, the time for LMA removal, and the time for transfer patients from the recovery room (as measured by the modified Aldrete scale) were statistically significantly shorter in the propofol-TCI group than in the MCI-propofol group.
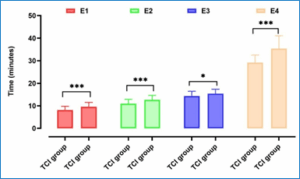
Figure 2: Recovery profile (E1–E4) comparing the TCI and MCI groups (Values are expressed as mean ± SD; statistically significant differences between groups were determined using independent t-tests (p-values obtained from the independent t-test; P < 0.05 considered significant); * P < 0.05; *** P < 0.001)
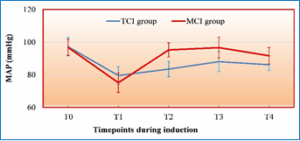
Figure 3: Mean arterial pressure changes during induction and LMA insertion
This prospective observational study describes differences between propofol TCI and MCI differ in terms of induction control, hemodynamic stability, and recovery characteristics when applied for anesthesia in elderly patients undergoing flap surgery for sacral pressure ulcers.
Our primary result indicates a potential cardiovascular phase in a high-risk geriatric cohort. The TCI group demonstrated a significantly lower incidence of hypotension (22.6% vs. 47.2%, P = 0.008) and a substantially reduced requirement for vasopressor intervention (phenylephrine: 9.4% vs. 34%, p = 0.002). In line with our study’s findings, recent evidence suggests propofol TCI provides superior hemodynamic stability during induction. Yildirim et al. demonstrated that TCI induction with propofol provided better hemodynamic stability than manual induction, reflecting a lower incidence of post-induction hypotension under computer-guided dosing.4 Although TCI often involves a higher total propofol dose, this does not appear to compromise anesthesia quality. Leslie et al. review noted that TCI was associated with higher propofol consumption but required “fewer interventions” by the anesthetist, with no significant differences in anesthetic depth or adverse events between TCI and manual infusions. These findings collectively indicate that TCI allows more precise titration (and thus smoother hemodynamic profiles) without adversely affecting patient outcomes 8. This success can be attributed to the controlled and precise infusion pattern provided by the TCI technique. Although the average propofol induction dose used in our study
(1.75 mg.kg-1) exceeded commonly recommended geriatric induction doses reported in prior studies,7 the TCI system effectively minimized risk by prolonging the induction time (TCI: 140.49 seconds vs. MCI: 52.81 seconds, P < 0.001). This controlled titration, dictated by the TCI model, avoids the rapid, dose-dependent peaks in plasma concentration that characterize manual bolus injection, thereby giving the elderly patient's compromised circulatory system sufficient time to compensate. Previous analyses have demonstrated that higher propofol doses are independently associated with greater risk of severe pre-incision hypotension, especially in the oldest patients. The universally accepted principle that the required induction dose decreases by approximately 0.1 mg.kg-1 for every 10-year increase in age further underscores the necessity of this precise titration.3
Furthermore, our findings contribute to addressing the inconsistency in prior geriatric studies. Although earlier research did not consistently demonstrate a hemodynamic advantage of TCI, our results in this particularly frail subgroup indicate that TCI may offer improved stability when cardiovascular reserve is critically impaired.6 These observations should be interpreted with caution but highlight the need for further controlled trials to verify whether TCI truly confers a hemodynamic benefit in such high-risk elderly patients.
Detailed analysis of the blood pressure trajectory further supports the precision of TCI. Figure 1 shows that the MCI group had more noticeable hemodynamic swings, as evidenced by a deeper drop in MAP post-induction (T1) and a much sharper increase right after LMA insertion (T2) (MAP 95.19 mmHg in MCI group versus 83.47 mmHg in TCI group; P < 0.001). Avoiding these large drops and subsequent reactive hypertension is vital in older patients to prevent end-organ damage.1 The utility of TCI in providing stable hemodynamics has been repeatedly shown across various settings, including sedation for complex procedures like transjugular intrahepatic portosystemic shunt (TIPS) 5 and during general anesthesia induction.9
The effectiveness of our TCI technique relied on the Schnider pharmacokinetic model. Comparative systematic reviews indicate that while models like Eleveld may offer high predictive accuracy, the Schnider model is known to exhibit better bias control in elderly patients compared to Marsh or Eleveld, making it a suitable choice for our geriatric cohort. Furthermore, utilizing a stepwise TCI technique (which TCI facilitates by gradual concentration adjustment) provides a more stable anesthesia induction and effective attenuation of the cardiovascular response in elderly patients compared to non-stepwise bolus techniques.10
A secondary finding of our study—addressing the established clinical gap by evaluating recovery characteristics—was that the TCI group appeared to reach all key recovery timepoints faster than the MCI group (Figure 2). This trend toward accelerated recovery has also been observed in multiple surgical contexts, which supports but does not confirm the hypothesis that TCI may not inherently delay recovery despite higher total doses reported in electroconvulsive therapy studies.11 Studies comparing TCI and MCI in general surgery, endoscopic retrograde cholangio-pancreatography procedures 12, and gynecological endoscopy have also found TCI leads to significantly shorter recovery times (e.g., TCI 7.48 min vs. MCI 10.3 min; TCI 11.60 min vs. MCI 15.40 min).13 Conversely, some reports on short-duration laparoscopic surgery have found recovery profiles to be comparable, suggesting the duration of the case or the surgical context may influence this benefit. 14 The computer-controlled pump in TCI automatically adjusts the infusion rate based on the programmed pharmacokinetic model and patient data (age, weight, height, sex). This prevents large fluctuations in drug concentration that can occur with manual adjustments, leading to a more stable level of anesthesia. By maintaining a more precise and consistent concentration, TCI can often lead to lower total drug consumption compared to manual infusion where excess drug might be administered to account for variability in patient response. A lower total dose results in a shorter time for the drug to be cleared from the body and for the patient to recover. In geriatric care, the objective is to speed recovery and minimize functional decline, as prolonged recovery is associated with an increased risk of postoperative complications, including cognitive dysfunction.1, 7, 15-17 Our findings are consistent with faster achievement of key recovery endpoints in the TCI group, thereby reducing the duration of exposure to residual anesthetics. This observation aligns with the overall goal of improving short-term postoperative outcomes in vulnerable elderly patients.
Our study, being observational and single-center, has limitations. The non-randomized allocation, though reflecting real-world clinical practice by anesthetists “blind to the study”, introduces the possibility of unmeasured confounding factors influencing group assignment or outcomes.
Although no a priori sample size calculation was performed, a post-hoc power analysis for the primary endpoint (incidence of post-induction hypotension) demonstrated a power of 0.91 at α = 0.05. This indicates that the study was adequately powered to detect the observed difference between groups, thereby supporting the reliability of the statistical findings despite the non-randomized design.
The use of the Schnider model for TCI may also limit the generalizability of quantitative results to other pharmacokinetic models (e.g., Marsh or Eleveld). Finally, the cohort’s homogeneity (elderly, sacral ulcer surgery, LMA anesthesia) limits the generalizability of these findings beyond similar high-risk, fragile surgical populations.
In conclusion, our prospective observational study suggests that TCI propofol may offer improved hemodynamic stability and a faster recovery profile compared to MCI in highly vulnerable elderly patients undergoing flap surgery for sacral pressure ulcers. TCI significantly reduced the incidence of hypotension and the need for vasopressor intervention while accelerating recovery milestones.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
The authors declare no conflict of interest, and no external or industry funding was involved.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
HVV: Participated in patient management, conceptualized the study, designed the methodology, collected and analyzed the data, and drafted the manuscript.
QVN: Participated in patient management, contributed to study design, collected and analyzed the data, and drafted the manuscript.
TXV, ANL: Collected and analyzed the data and contributed to drafting the manuscript.
Authors affiliation:
- Hien Van Vo, Department of Anesthesia, Le Huu Trac National Burn Hospital / Department of Anesthesia, Vietnam Military Medical University, Hanoi, Vietnam; Email: vanhien@vmmu.edu.vn
- Quynh Van Nguyen, Department of Anesthesia, Le Huu Trac National Burn Hospital, Hanoi, Vietnam; Email: nguyenvanquynh304@gmail.com
- Thang Xuan Vy, Department of Anesthesia, Le Huu Trac National Burn Hospital, Hanoi, Vietnam; Email: thangvx.anes@gmail.com
- Anh Ngoc Le, Department of Anesthesia, Le Huu Trac National Burn Hospital, Hanoi, Vietnam; Email: quangpro911@gmail.com
ABSTRACT
Background: This study compared the effects of Target-Controlled Infusion (TCI) versus Manually-Controlled Infusion (MCI) of propofol on hemodynamic stability and recovery profiles in this vulnerable cohort, aiming to establish the safer administration method.
Methodology: This was a single-center, non-randomized, prospective, observational study, involving 106 elderly patients (≥ 65 years) (TCI n = 53, MCI n = 53). TCI used the Schnider model for propofol titration, while MCI involved slow bolus injection. The primary endpoints were the incidence of post-induction hypotension (MAP <65 mmHg or ≥30% reduction from baseline) and the need for vasopressor (phenylephrine) intervention. Secondary outcomes included intra-induction hemodynamic trends and key recovery milestones (spontaneous respiration, verbal command, LMA removal, and PACU discharge).
Results: Despite similar propofol induction doses, the TCI group showed a significantly longer induction time (140.49 s vs. 52.81 s, 0.001). TCI provided superior hemodynamic stability, resulting in a substantially lower incidence of post-induction hypotension (22.6% vs. 47.2%, p = 0.008) and a reduced need for phenylephrine (9.4% vs. 34%, p = 0.002). Hemodynamic analysis showed that TCI mitigated both the depth of hypotension post-induction and the degree of reactive hypertension post-LMA insertion. The TCI group achieved all key recovery milestones significantly faster, including time to spontaneous respiration (8.15 min vs. 9.60 min, P < 0.001) and time to PACU discharge (29.21 min vs. 35.43 min, P < 0.001).
Conclusion: In high-risk elderly patients undergoing sacral flap surgery, propofol TCI may improve hemodynamic stability during induction and facilitate recovery compared to MCI. The controlled, gradual titration of TCI is highly effective in minimizing cardiovascular stress and could be considered a preferred technique for propofol administration in frail geriatric surgical populations.
Abbreviation: TCI: Target-Controlled Infusion; MCI: Manually Controlled Infusion; LMA: Laryngeal Mask Airway; MAP: Mean Arterial Blood Pressure; PACU: Post Anaesthesia Care Unit
Keywords: Propofol; Target-Controlled Infusion; TCI; Manual Controlled Infusion; MCI; Elderly Patients; Hypotension; Recovery Profile; Sacral Pressure Ulcer Flap Surgery
Citation: Vo HV, Nguyen QV, Vy AN. Target-controlled versus manual-controlled infusion of propofol: comparative hemodynamic stability during induction and recovery profiles in elderly patients undergoing sacral pressure ulcer flap surgery-a non-randomized prospective observational study. Anaesth. pain intensive care 2025;30(1):47-54. DOI: 10.35975/apic.v30i1.3101
Received: October 15, 2025; Revised: November 06, 2024; Accepted: November 06, 2025
1. INTRODUCTION
Anesthesia induction in elderly patients presents significant challenges due to age-related physiological changes, comorbidities, and reduced cardiovascular reserve. Such patients are especially prone to hypotension, myocardial ischemia, cerebral hypoperfusion, and other complications when undergoing major surgery.1 Flap surgery for sacral pressure ulcers represents a particularly hemodynamically challenging context. These procedures are typically prolonged and associated with substantial blood loss, large wound areas, and fluid shifts. Moreover, patients are often frail, nutritionally depleted, chronically immobilized, and affected by systemic inflammation, all of which further compromise cardiovascular stability and increase anesthetic risk.
Propofol remains a commonly used agent for induction of general anesthesia because of its rapid onset and favorable recovery profile. However, its hemodynamic effect such as vasodilation, decreased systemic vascular resistance, and myocardial depression can be profound in the elderly.2 The induction dose of propofol required for loss of consciousness decreases with age, approximately by 0.1 mg.kg-1 for every 10-year increase, necessitating precise dosing strategies to avoid complications such as profound hypotension.3 The method of administration of propofol contributes a critical role in hemodynamic response: Manual Controlled Infusion (MCI) tend to cause rapid rises in plasma concentration, producing vasodilation, reduction in systemic vascular resistance, decreased preload, and myocardial depression. In contrast, Target-Controlled Infusion (TCI) employs pharmacokinetic/pharmacodynamic (PK/PD) models (e.g., Schneider, Marsh) to titrate propofol gradually toward predefined effect-site concentration, which may provide a more stable induction profile. A recent randomized controlled trial demonstrated that TCI induction significantly reduced the incidence of post-induction hypotension compared to manual induction (13% vs. 34%)4 consistent with other studies showing lower hypotension rates with TCI in various populations.5
However, evidence concerning its superiority in dedicated elderly cohorts remains conflicting; for instance, at least one comparative study in elderly patients found no significant differences in hemodynamic stability or the need for vasoactive drugs between TCI and MCI.6 Moreover, prior comparative studies have often emphasized intra-induction hemodynamics while overlooking recovery quality - an increasingly important metric in geriatric anesthesia.7
To address these gaps, we conducted a prospective observational study comparing TCI and MCI propofol induction in elderly patients undergoing flap surgery for sacral pressure ulcers. The primary objective was to evaluate their hemodynamic impact during induction, including blood pressure trends, heart rate responses, and the need for corrective interventions such as vasopressors or treatment of bradycardia. In addition, the study assessed key recovery milestones (time to spontaneous respiration, eye opening, verbal response, and extubation) to characterize the overall induction–recovery profile associated with each method.
By evaluating both the induction and emergence phases, this observational design aimed to offer preliminary insights into the hemodynamic safety and recovery characteristics of TCI versus MCI in a vulnerable geriatric surgical population.
2. METHODOLOGY
This single-center, non-randomized, prospective, observational study was conducted at the Le Huu Trac National Burns Hospital (Vietnam Military Medical University), the central specialized burn care hospital in Hanoi, Vietnam, between August 2024 and May 2025.
The study protocol underwent institutional review and received official approval from the Medical Research Committee of the Le Huu Trac National Burns Hospital, Vietnam Military Medical University (Official Dispatch No. 329/BVB- KHTH dated March 28, 2024). The research strictly adhered to the ethical principles outlined in the Declaration of Helsinki published by the World Medical Association (WMA). Informed written consent was obtained from all participating patients and their families prior to study participation. Patient anonymity and data confidentiality were maintained and stored securely throughout the duration of the study.
Patients aged ≥ 65 years; ASA (American Society of Anesthesiologists) physical status I-III; scheduled for flap transfer surgery for sacral pressure ulcer repair under general anesthesia with LMA. Known allergy to propofol or adjunct medications; severe baseline cognitive impairment; inability to consent; history of malignant hyperthermia; major cardiovascular instability preoperatively; need for alternative airway.
All participants were evaluated to ensure they fulfilled the study’s selection requirements, given information about the anesthesia technique and the medications used in the research, and asked to sign a consent form to take part in the study. Anesthetists were instructed to assign patients to either the TCI or MCI group according to their routine clinical practice, primarily based on the availability of the infusion system and the patient’s cardiovascular tolerance to induction. Importantly, allocation was not influenced by hemodynamic status, comorbidities, or anesthetist preference toward a specific technique.
Baseline characteristics, including hypertension, cardiac disease, diabetes, and ASA physical status, were statistically compared between groups and showed no significant differences (Table 2), thereby minimizing potential selection bias.
A peripheral intravenous line was used to infuse the patients with a 0.9% sodium chloride solution at a rate of 5 mL.min-1. A monitor (Nihon Kohden PVM 2701- Japan) was set up to track standard parameters including non-invasive arterial blood pressure (ABP), heart rate, electrocardiogram (DII), end-tidal CO₂ pressure (etCO₂), oxygen saturation (SpO2), and Bispectral Index (BIS). A slow intravenous injection of 1 mg midazolam was administered to each patient as a pre-anesthetic medication.
All patients received fentanyl 2 µg / kg IV before induction.
TCI group: Propofol was infused by PERFUSOR® SPACE TCI pump (B. Braun, Germany) using the Schneider model with an initial effect-site concentration (Ce) of 1.5 µg/mL; incrementally increased by 0.5 µg/mL until loss of consciousness (LOC), defined by BIS < 60.
MCI group: Propofol was given manually by slow IV bolus, dose 1.0–1.5 mg/kg, administered in 20 mg increments until LOC (BIS < 60).
No neuromuscular blocking agents were given to the patients. An appropriate size Pro-Seal LMA was inserted for perioperative airway management. A/C mode mechanical ventilation (Carestation™ 620 anesthesia machine, GE Healthcare, USA) was used with a tidal volume of 6–8 mL/kg, respiratory rate of 12-16 breaths per minute, and an inspiratory-to-expiratory (I:E) ratio of 1:2.
Anaesthesia was maintained by propofol with either TCI pump (Ce = 2-3.5 µg/mL (TCI group) or a continuous intravenous infusion of propofol at the dose of 8- 10 mg/kg/h (MCI group). The maintenance dose of propofol was adjusted based on mean arterial pressure and the BIS index within the range of 40–60. Additional fentanyl (1-2 µg/kg) was given as need.
Immediately following the final suture, a propofol infusion was also stopped in both groups. "Zero ending time" (E0) was the moment when propofol was stopped; the total amount of propofol taken up to this point was measured. Upon completion of the surgery, the LMA was removed, and intravenous paracetamol (10 mg/kg) was administered for postoperative analgesia.
2.1. Data Collection & Outcomes
Data collected prospectively included:
- Baseline demographics and comorbidities (age, sex, weight, height, BMI, ASA status, hypertension history)
- Hemodynamic parameters: SBP, DBP, MAP, HR at the timepoints (Table 1)
- The percentage requirement for vasopressors (phenylephrine)
Occurrence of hypotension: defined as MAP < 65 mmHg or ≥ 30% reduction from baseline at any of the timepoints during induction through 10 minutes post-induction. When hypotension occurred, phenylephrine 100 μg intravenous bolus was administered and repeated as required to restore MAP within 20% of baseline (total dose up to 200 µg).
For recovery characteristic, time was measured from the propofol stoppage time (E0), and the two groups were compared (Table 1).
| Table 1: Timepoints for evaluation of hemodynamic and recovery parameters | |
| T0 | Prior to induction |
| T1 | Post-induction, pre-LMA insertion |
| T2 | Immediately after LMA insertion |
| T3 | 5 minutes after LMA insertion |
| T4 | 10 minutes after LMA |
| E0 | The propofol infusion is stopped |
| E1 | Spontaneous respiration resumes (assessed by manually feeling the reservoir bag on spontaneous mode with the valve fully open) |
| E2 | Verbal command response (assessed by asking the patient to open their eyes, protrusion of tongue on opening the mouth, and looking for the follow‑up of the command) |
| E3 | LMA removal |
| E4 | Move out of the Post Anaesthesia Care Unit (PACU) (The Modified Aldrete score was assigned to each group before shifting the patient out of PACU). |
2.1. Statistical analysis
Continuous variables assessed for normality (Shapiro-Wilk). Normally distributed data expressed as mean ± SD; non-normal as median (IQR). Between-group comparisons with independent t-test or Mann-Whitney U test. Categorical data: Chi-square or Fisher’s exact test. All analyses were performed using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). A two-tailed P < 0.05 was considered statistically significant.
3. RESULTS
During the period of the study, a total of 125 elderly patients scheduled for flap surgery for sacral pressure ulcers were initially considered for inclusion. After applying inclusion and exclusion criteria, 106 patients fulfilled eligibility and were enrolled in the study. The remaining patients were excluded for various reasons (e.g. not meeting criteria, withdrawal of consent, intraoperative cancellation). Of the 106 enrolled, 53 patients were allocated to the TCI group and 53 patients to the MCI group (see Table 1). The values of the variables obtained in the study were statistically analyzed using the Shapiro-Wilk test to ensure normal distribution.
There were no differences between the two groups’ demographics in terms of age, sex, ASA status, history of hypertension, or other comorbidities (Table 2)
| Table 2: Demographic parameters and study data between TCI and MCI induction groups | |||
| Patients’ Characteristics | Propofol –TCI group (n = 53) | Propofol- MCI
group (n = 53) |
P- value |
| Age (years) | 75.21 ± 6.68 | 74.21 ± 7.06 | 0.46* |
| Sex (Male/Female) | 30/23 | 35/18 | 0.32# |
| Height (cm) | 159.58 ± 7.84 | 157.42 ± 7.04 | 0.14* |
| Weight (kg) | 51.47± 7.23 | 52.15 ± 5.77 | 0.59* |
| BMI | 20.25 ± 3.44 | 21.17 ± 2.99 | 0.20* |
| History of hypertension (n,%) | 32 (60.4) | 36 (67.9) | 0.42# |
| ASA status (I/II/III) | 0/28/25 | 0/29/24 | 0.85# |
| Amount of propofol for induction (mg) | 88.68 ± 12.25 | 92.26 ± 10.49 | 0.11* |
| Propofol dose for induction (mg.kg-1) | 1.72 ± 0.15 | 1.78 ± 0.22 | 0.16* |
| Induction time (seconds) | 140.49 ± 33.06 | 52.81 ± 7.08 | <0.001* |
| Time for successful LMA insertion (seconds) | 14.66 ± 2.96 | 15.15 ± 2.79 | 0.40* |
| First attempt success at LMA insertion (n, %) | 53 (100) | 53 (100) | - |
| Hypotension after induction (n, %) | 12/53 (22.6%) | 25/53 (47.2%) | 0.008** |
| Hypotension requiring treatment (n, %) | 5/53 (9.4%) | 18/53 (34%) | 0.002** |
| Bradycardia requiring treatment (n, %) | 1/53 (1.9%) | 6/53 (11.3%) | 0.113## |
| Total propofol consumption (mg) | 471.43 ± 36.38 | 474.04 ± 40.91 | 0.73* |
| Total fentanyl consumption (µg) | 226.60 ± 21.74 | 228.83 ± 18.98 | 0.58* |
| BMI, Body Mass Index; BIS, Bispectral index; P-value obtained from the independent T-test Independent T- test(*); Chi- square test(#);Pearson- Chi square test(**);and Fisher’s exact test(##); P < 0.05 considered significant. | |||
For LMA insertion, the time to successful insertion was 14.66 ± 2.96 seconds in the TCI group and 15.15 ± 2.79 seconds in the MCI group; this difference was not statistically significant (P = 0.40). First attempt success rates for LMA insertion were 100 % in both groups (53/53).
Table 2 also findings demonstrated that the TCI group’s percentage of hypotensive patients and the rate of phenylephrine required to treat hypotension were lower than those of the MCI group (22.6% vs. 47.2% (P = 0.008) and 9.4% vs. 34% (P = 0.002), respectively).
Hemodynamic changes during anesthesia induction and LMA insertion in both study groups are presented in Table 3 and Figure 1. At the baseline (T0), there were no statistically significant differences in hemodynamic parameters between the two groups (P > 0.05). Following induction of anesthesia (T1), both groups exhibited a decrease in ABP; however, the reduction was significantly greater in the MCI group, with a higher incidence of hypotension compared with the TCI group (P < 0.001). During LMA insertion (T2), stimulation caused a transient increase in blood pressure, which was significantly higher in the MCI group than in the TCI group. Thereafter, hemodynamic parameters in both groups remained within normal limits, although the TCI group demonstrated more stable trends over time.
| Table 3: Comparison of hemodynamic changes pre- and post- anesthesia induction and LMA insertion hemodynamic parameters | |||
| Time point | TCI Group | MCI group | P-value |
| SAP (mmHg) | |||
| T0 | 140.64 ± 10.31 | 138.17 ± 10.95 | 0.23* |
| T1 | 101.94 ± 10.17 | 97.60 ± 9.39 | 0.025* |
| T2 | 99.60 ± 6.73 | 119.81 ± 9.96 | <0.001* |
| T3 | 114.32 ± 12.51 | 122.06 ±15.64 | 0.06* |
| T4 | 111.40 ± 13.81 | 117.47 ± 9.16 | <0.001* |
| MAP (mmHg) | |||
| T0 | 97.18 ± 5.67 | 96.74 ± 4.86 | 0.66* |
| T1 | 79.52 ± 5.42 | 75.13 ± 5.92 | <0.001* |
| T2 | 83.47 ± 4.74 | 95.19 ± 4.38 | <0.001* |
| T3 | 88.03 ± 6.14 | 96.66 ± 6.33 | <0.001* |
| T4 | 86.11 ± 3.50 | 91.66 ± 5.07 | <0.001* |
| DAP (mmHg) | |||
| T0 | 75.45 ± 5.98 | 76.02 ± 6.11 | 0.63* |
| T1 | 68.30 ± 6.70 | 63.89 ± 5.89 | <0.001* |
| T2 | 75.40 ± 6.13 | 82.89 ± 4.40 | <0.001* |
| T3 | 74.89 ± 6.13 | 83.96 ± 5.05 | <0.001* |
| T4 | 69.66 ± 2.68 | 73.08 ± 3.52 | <0.001* |
| Heart rate (beats.min-1) | |||
| T0 | 70.83 ± 6.09 | 70.64 ± 6.78 | 0.88* |
| T1 | 65.87 ± 4.58 | 60.47 ± 5.95 | <0.001* |
| T2 | 70.42 ± 3.02 | 73.00 ± 7.10 | 0.17* |
| T3 | 72.62 ± 4.60 | 72.91 ± 4.16 | 0.74* |
| T4 | 67.72 ± 4.65 | 68.11 ± 4.47 | 0.65* |
| SAP, systolic arterial pressure; DAP, diastolic arterial pressure; MAP, mean arterial pressure.
(*):P < 0.05 considered significant; independent T-test. |
|||

Figure 1: Changes in MAP during induction of anesthesia in the TCI and MCI groups at five predefined timepoints (T0–T4).Values are expressed as mean ± SD.
In terms of recovery profile, Table 4 and Figure 2 shows that the time for the patient to breathe again, the time for the patient to respond to commands, the time for LMA removal, and the time for transfer patients from the recovery room (as measured by the modified Aldrete scale) were statistically significantly shorter in the propofol-TCI group than in the MCI-propofol group.
| Table 4: Recovery profile of the patients in both groups | |||
| Patients’ Characteristics | TCI group
(n = 53) |
MCI group
(n = 53) |
p- value |
| Time to spontaneous breathing (min) | 8.15 ± 1.63 | 9.60 ± 1.89 | < 0.001* |
| Time to obey verbal command (min) | 10.96 ± 1.93 | 12.64 ± 1.99 | < 0.001* |
| Time to removal of LMA (min) | 14.32 ± 2.13 | 15.38 ± 1.99 | < 0.05* |
| Time for PACU discharge (min) | 29.21 ± 3.31 | 35.43 ± 5.66 | < 0.001* |
| (*):P-value obtained from the Independent T-test. | |||

Figure 2: Recovery profile (E1–E4) comparing the TCI and MCI groups (Values are expressed as mean ± SD; statistically significant differences between groups were determined using independent t-tests (p-values obtained from the independent t-test; P < 0.05 considered significant); * P < 0.05; *** P < 0.001)

Figure 3: Mean arterial pressure changes during induction and LMA insertion
4. DISCUSSION
This prospective observational study describes differences between propofol TCI and MCI differ in terms of induction control, hemodynamic stability, and recovery characteristics when applied for anesthesia in elderly patients undergoing flap surgery for sacral pressure ulcers.
Our primary result indicates a potential cardiovascular phase in a high-risk geriatric cohort. The TCI group demonstrated a significantly lower incidence of hypotension (22.6% vs. 47.2%, P = 0.008) and a substantially reduced requirement for vasopressor intervention (phenylephrine: 9.4% vs. 34%, p = 0.002). In line with our study’s findings, recent evidence suggests propofol TCI provides superior hemodynamic stability during induction. Yildirim et al. demonstrated that TCI induction with propofol provided better hemodynamic stability than manual induction, reflecting a lower incidence of post-induction hypotension under computer-guided dosing.4 Although TCI often involves a higher total propofol dose, this does not appear to compromise anesthesia quality. Leslie et al. review noted that TCI was associated with higher propofol consumption but required “fewer interventions” by the anesthetist, with no significant differences in anesthetic depth or adverse events between TCI and manual infusions. These findings collectively indicate that TCI allows more precise titration (and thus smoother hemodynamic profiles) without adversely affecting patient outcomes 8. This success can be attributed to the controlled and precise infusion pattern provided by the TCI technique. Although the average propofol induction dose used in our study
(1.75 mg.kg-1) exceeded commonly recommended geriatric induction doses reported in prior studies,7 the TCI system effectively minimized risk by prolonging the induction time (TCI: 140.49 seconds vs. MCI: 52.81 seconds, P < 0.001). This controlled titration, dictated by the TCI model, avoids the rapid, dose-dependent peaks in plasma concentration that characterize manual bolus injection, thereby giving the elderly patient's compromised circulatory system sufficient time to compensate. Previous analyses have demonstrated that higher propofol doses are independently associated with greater risk of severe pre-incision hypotension, especially in the oldest patients. The universally accepted principle that the required induction dose decreases by approximately 0.1 mg.kg-1 for every 10-year increase in age further underscores the necessity of this precise titration.3
Furthermore, our findings contribute to addressing the inconsistency in prior geriatric studies. Although earlier research did not consistently demonstrate a hemodynamic advantage of TCI, our results in this particularly frail subgroup indicate that TCI may offer improved stability when cardiovascular reserve is critically impaired.6 These observations should be interpreted with caution but highlight the need for further controlled trials to verify whether TCI truly confers a hemodynamic benefit in such high-risk elderly patients.
Detailed analysis of the blood pressure trajectory further supports the precision of TCI. Figure 1 shows that the MCI group had more noticeable hemodynamic swings, as evidenced by a deeper drop in MAP post-induction (T1) and a much sharper increase right after LMA insertion (T2) (MAP 95.19 mmHg in MCI group versus 83.47 mmHg in TCI group; P < 0.001). Avoiding these large drops and subsequent reactive hypertension is vital in older patients to prevent end-organ damage.1 The utility of TCI in providing stable hemodynamics has been repeatedly shown across various settings, including sedation for complex procedures like transjugular intrahepatic portosystemic shunt (TIPS) 5 and during general anesthesia induction.9
The effectiveness of our TCI technique relied on the Schnider pharmacokinetic model. Comparative systematic reviews indicate that while models like Eleveld may offer high predictive accuracy, the Schnider model is known to exhibit better bias control in elderly patients compared to Marsh or Eleveld, making it a suitable choice for our geriatric cohort. Furthermore, utilizing a stepwise TCI technique (which TCI facilitates by gradual concentration adjustment) provides a more stable anesthesia induction and effective attenuation of the cardiovascular response in elderly patients compared to non-stepwise bolus techniques.10
A secondary finding of our study—addressing the established clinical gap by evaluating recovery characteristics—was that the TCI group appeared to reach all key recovery timepoints faster than the MCI group (Figure 2). This trend toward accelerated recovery has also been observed in multiple surgical contexts, which supports but does not confirm the hypothesis that TCI may not inherently delay recovery despite higher total doses reported in electroconvulsive therapy studies.11 Studies comparing TCI and MCI in general surgery, endoscopic retrograde cholangio-pancreatography procedures 12, and gynecological endoscopy have also found TCI leads to significantly shorter recovery times (e.g., TCI 7.48 min vs. MCI 10.3 min; TCI 11.60 min vs. MCI 15.40 min).13 Conversely, some reports on short-duration laparoscopic surgery have found recovery profiles to be comparable, suggesting the duration of the case or the surgical context may influence this benefit. 14 The computer-controlled pump in TCI automatically adjusts the infusion rate based on the programmed pharmacokinetic model and patient data (age, weight, height, sex). This prevents large fluctuations in drug concentration that can occur with manual adjustments, leading to a more stable level of anesthesia. By maintaining a more precise and consistent concentration, TCI can often lead to lower total drug consumption compared to manual infusion where excess drug might be administered to account for variability in patient response. A lower total dose results in a shorter time for the drug to be cleared from the body and for the patient to recover. In geriatric care, the objective is to speed recovery and minimize functional decline, as prolonged recovery is associated with an increased risk of postoperative complications, including cognitive dysfunction.1, 7, 15-17 Our findings are consistent with faster achievement of key recovery endpoints in the TCI group, thereby reducing the duration of exposure to residual anesthetics. This observation aligns with the overall goal of improving short-term postoperative outcomes in vulnerable elderly patients.
Our study, being observational and single-center, has limitations. The non-randomized allocation, though reflecting real-world clinical practice by anesthetists “blind to the study”, introduces the possibility of unmeasured confounding factors influencing group assignment or outcomes.
Although no a priori sample size calculation was performed, a post-hoc power analysis for the primary endpoint (incidence of post-induction hypotension) demonstrated a power of 0.91 at α = 0.05. This indicates that the study was adequately powered to detect the observed difference between groups, thereby supporting the reliability of the statistical findings despite the non-randomized design.
The use of the Schnider model for TCI may also limit the generalizability of quantitative results to other pharmacokinetic models (e.g., Marsh or Eleveld). Finally, the cohort’s homogeneity (elderly, sacral ulcer surgery, LMA anesthesia) limits the generalizability of these findings beyond similar high-risk, fragile surgical populations.
5. CONCLUSION
In conclusion, our prospective observational study suggests that TCI propofol may offer improved hemodynamic stability and a faster recovery profile compared to MCI in highly vulnerable elderly patients undergoing flap surgery for sacral pressure ulcers. TCI significantly reduced the incidence of hypotension and the need for vasopressor intervention while accelerating recovery milestones.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
The authors declare no conflict of interest, and no external or industry funding was involved.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
HVV: Participated in patient management, conceptualized the study, designed the methodology, collected and analyzed the data, and drafted the manuscript.
QVN: Participated in patient management, contributed to study design, collected and analyzed the data, and drafted the manuscript.
TXV, ANL: Collected and analyzed the data and contributed to drafting the manuscript.
10. REFERENCES
- Kanonidou Z, Karystianou G. Anesthesia for the elderly. Hippokratia. 2007;11(4):175-7.
- Schonberger RB, Dai F, Michel G, Vaughn MT, Burg MM, Mathis M, et al. Association of propofol induction dose and severe pre-incision hypotension among surgical patients over age 65. J Clin Anesth. 2022;80:110846. DOI:10.1016/j.jclinane.2022.110846.
- Nishimura K, Hirata K, Noriaki F, Watabe A, Morimoto Y. The Relationship Between Age and the Propofol Dose for Anesthesia Induction: A Single-Center Retrospective Study Utilizing Neural Network Model Simulation. Appl Sci. 2025;15:6052. DOI:10.3390/app15116052.
- Yildirim SA, Dogan L, Sarikaya ZT, Ulugol H, Gucyetmez B, Toraman F. Hypotension after Anesthesia Induction: Target-Controlled Infusion Versus Manual Anesthesia Induction of Propofol. J Clin Med. 2023;12(16):5280. DOI:10.3390/jcm12165280.
- Elshafie MA, Salem MA, Ghoniem AM, Abdelrahman AI. Target controlled vs. manually controlled infusion of propofol for sedation during transjugular intrahepatic portosystemic shunt procedure: a randomized controlled trial. Anaesth Pain Intensive Care. 2025;29(3):403-10. DOI:10.35975/apic.v29i3.2754.
- Li M, Xu CY, Wang XD, Zhang LP, Guo XY. A comparison of target controlled versus manually controlled infusion of propofol in elderly patients. Zhonghua Yi Xue Za Zhi. 2011;91(9):600-3.
- Phillips AT, Deiner S, Mo Lin H, Andreopoulos E, Silverstein J, Levin MA. Propofol Use in the Elderly Population: Prevalence of Overdose and Association With 30-Day Mortality. Clin Ther. 2015;37(12):2676-85. DOI:10.1016/j.clinthera.2015.10.005.
- Leslie K, Clavisi O, Hargrove J. Target-controlled infusion versus manually-controlled infusion of propofol for general anaesthesia or sedation in adults. Cochrane Database Syst Rev. 2016;16(3):CD006059. DOI:10.1002/14651858.CD006059.pub2.
- Vale AGG, Govêia CS, Guimarães GMN, Terra LR, Ladeira LCA, Essado GA. Comparison of arterial hypotension incidence during general anesthesia induction - target-controlled infusion vs. bolus injection of propofol: a randomized clinical trial. Braz J Anesthesiol. 2024;74(4):844503. DOI:10.1016/j.bjane.2024.844503.
- Linassi F, Zanatta P, Spano L, Burelli P, Farnia A, Carron M. Schnider and Eleveld Models for Propofol Target-Controlled Infusion Anesthesia: A Clinical Comparison. Life (Basel). 2023;13(10):2065. DOI:10.3390/life13102065.
- Hsieh ML, Lu YT, Lin CC, Lee CP. Comparison of the target-controlled infusion and the manual infusion of propofol anesthesia during electroconvulsive therapy: an open-label randomized controlled trial. BMC Psychiatry. 2021;21(1):71. DOI:10.1186/s12888-021-03069-6.
- Sahu A, Tripathy DK, Gupta B, Talawar P, Gupta R. Recovery time in target controlled infusion versus manual infusion of propofol in total intravenous anaesthesia for endoscopic retrograde cholangio-pancreatography procedure using laryngeal mask airway-gastro: A randomised comparative study. Indian J Anaesth. 2023;67(Suppl 2):S120-S5. DOI:10.4103/ija.ija_484_22.
- Subramaniyan , K., & Kuppusamy, A. Comparison of Target-Controlled Infusion vs. Manual Infusion of Propofol on Postoperative Recovery in Gynecological Endoscopic Procedures: An Open-Label Randomized Controlled Trial. South Eastern European Journal of Public Health. 2025, XXVI(S2): 4104–4109. DOI: 10.70135/seejph.vi.5984
- Kateliya R, Madhukant, Dubey M, Chandra S, Sahay N. Comparison of recovery profiles in target-controlled infusions (TCI) versus manually controlled infusions for total intravenous anesthesia (TIVA) in laparoscopic surgeries. A randomized controlled trial. J Anaesthesiol Clin Pharmacol. 2023;39(2):258-63. DOI:10.4103/joacp.joacp_396_21.
- Igarashi T, Nagata O, Kuroyanagi A, Iwakiri H, Ozaki M. Reliability of propofol target-controlled infusion in elderly patients. Masui. 2009;58(11):1407-12.
- Jia L, Hou J, Zheng H, Sun L, Fan Y, Wang X, et al. Study of the rational dose of propofol in elderly patients under bispectral index monitoring during total intravenous anesthesia: A PRISMA-compliant systematic review. Medicine (Baltimore). 2020;99(5):e19043. DOI:10.1097/MD.0000000000019043.
- Liu SH, Wei W, Ding GN, Ke JD, Hong FX, Tian M. Relationship between depth of anesthesia and effect-site concentration of propofol during induction with the target-controlled infusion technique in elderly patients. Chin Med J (Engl). 2009;122(8):935-40.