Shrouk A. H. Al.Ibraheem 1 , Ali A. H. Albakaa 2
Authors affiliations:
Background & objectives: Kidney stone formation is a prevalent and recurrent condition associated with significant morbidity. While traditional risk factors are known, the roles of subclinical renal dysfunction and inflammation in its pathogenesis are still being explored. This study aimed to evaluate serum cystatin C (Cys-C) as a marker of renal function and Interleukin-18 (IL-18) as a pro-inflammatory marker in patients with kidney stones, and to assess their correlation with conventional biochemical markers.
Methods: In this case-control study, 60 patients with a confirmed diagnosis of nephrolithiasis, and 60 age- and sex-matched healthy volunteers were enrolled. Serum levels of Cys-C and IL-18 were quantified using enzyme-linked immunosorbent assay (ELISA). Serum Ca+2, phosphorus (P), and uric acid (UA) levels were measured using a clinical chemistry analyzer. Statistical analyses were performed to compare the biomarker levels between the two groups.
Results: The patient group demonstrated significantly higher serum levels of Cys-C (0.368 ± 0.15 vs. 0.303 ± 0.08 mg/dl; P < 0.001) and IL-18 (10.04 ± 4.99 vs. 7.24 ± 2.21 ng/L; P < 0.001) compared to the healthy control group. Patients with nephrolithiasis also had significantly elevated levels of UA (7.04 ± 1.79 vs. 4.81 ± 1.36 mg/dl; P < 0.001), Ca+2 (9.97 ± 0.91 vs. 9.02 ± 0.69 mg/dl; P < 0.001), and P (4.89 ± 1.04 vs. 3.43 ± 0.72 mg/dl; P < 0.001). Within the patient group, there were no significant differences in these biomarker levels based on sex or age.
Conclusion: The significant elevation of Cys-C and IL-18 in patients with nephrolithiasis suggests that subclinical renal dysfunction and pro-inflammatory processes are key components of the disease's pathophysiology. These markers, in conjunction with traditional stone-forming minerals, may serve as a valuable panel for assessing disease activity and renal involvement in patients with kidney stones.
Abbreviations: Cys-C: cystatin C, IL-18: Interleukin-18, P: phosphorus, UA: uric acid
Keywords: Nephrolithiasis; Kidney stones; Urolithiasis; Cystatin C; Interleukin-18; Renal biomarkers
Citation: Al.Ibraheem SAH, Albakaa AAH. Case control study of renal and inflammatory biomarkers in patients with urolithiasis. Anaesth. pain intensive care 2025;29(8):897-904. DOI: 10.35975/apic.v29i8.3016
Received: July 11, 2025; Revised: August 01, 2025; Accepted: August 02, 2025
The urinary system plays a paramount role in homeostasis by controlling the water and electrolyte balance of the body, removing waste products, and releasing them as urine. Dysregulation of this system often manifesting as kidney stones is becoming more prevalent and is associated with severe health problems. Kidney stones are hard accumulations of crystals that are created from the supersaturation of minerals such as calcium, oxalate, or UA in the urine.1 Stones may range in size and anatomical location within the urinary system and lead to symptoms of intense pain, hematuria and urinary outflow obstruction, with potential to result in impaired renal function if not disintegrated.2
Nephrolithiasis is a universal disorder with increasing incidence due to changing lifestyles and dietary habits. Nephrolithiasis afflicts approximately 10–15% of the worldwide population at some point in their life, and the rates of recurrence reach 50% in 5–10 years.3 The disease is more prevalent in men than women, with age of onset most common between 30 and 60 years old. 2 Location differences are apparent, with a higher prevalence in warmer countries, which is associated with dehydration. The global burden of kidney stones has been increased due to increasing prevalence of obesity, diabetes, and metabolic syndrome.4
Kidney stones are the result of an interplay of complex genetic, environmental and biological factors. One of the major reasons is the supersaturation of the urine with stone-forming materials including Ca+2, oxalate, UA or P, which consecutively initiates the processes of crystal formation, growth and agglomeration.5 Genetic predisposition is also largely involved, as genetic variables like hypercalciuria or cystinuria can elevate the incidence of stones.6 Lifestyle risk factors for chronic kidney diseases (CKD) such as low fluid intake, high sodium and animal protein dietary intake also increase risk by shifting urine composition and pH.7
Cys-C, a low molecular weight protein synthesized by cells with nucleus, is considered a valuable marker of renal function and urinary system diseases. Cystatin C Unlike creatinine, cystatin C is relatively unaffected by muscle mass, age, and sex that make it a more robust indicator of GFR and early renal insufficiency.8 cystatin C has been associated with pathogenesis of kidney stones, which is a prevalent urinary system disease. Studies have indicated that cystatin C has the potential to inhibit the nucleation of calcium oxalate, a main component of kidney stones, through modulating inflammation pathways as well as decreasing oxidative stress.9
IL-18 is a pro-inflammatory cytokine of the IL-1 superfamily and has been implicated in the immune and inflammatory responses. It is produced as an inactive precursor, and needs cleaved by caspase-1 to be active.10,11 Regarding the disease of the urinary system, IL-18 has been identified to participate in the development of many kidney diseases such as acute kidney injury (AKI), chronic kidney disease (CKD) and nephrolithiasis. In Kidney Stone disease IL-18 are involved in inflammatory micro environment to allow crystal formation and to retain crystals.12 This study aimed to evaluate serum Cys-C levels as it’s related to kidney function and investigate the role of inflammatory marker (IL-18) in kidney stone and assess their correlation with conventional biochemical markers, particularly Ca+2, P and UA
case-control study design was used in this study. Samples were collected at the Al-Shatrah General Hospital in Thi-Qar Province from individuals diagnosed with urinary systems disorders. The study population comprised 60 patients divided into two categories: 43 male patients and 17 female patients (diagnosis with nephrolithiasis). In addition, Control Group: Healthy volunteers (n = 60) divided into two categories: 36 male individuals and 24 female individuals from the same community as the patients. Sample collection was conducted between November 2024 and April 2025, and the study protocol was approved by the IRB (289/2024–December 12, 2024).
2.1. Inclusion and exclusion criteria
Patients were eligible for inclusion in the urinary system disorders and infection of kidney stones if they had a confirmed diagnosis of urinary system disorders, based on clinical presentation, physical examination, laboratory findings, and imaging studies. Only individuals aged 27 years or older were included. Participants were excluded if they had a history of cancer patients, people with abnormalities thyroid gland, and pregnancy women. Further exclusion criteria included the use of corticosteroids or other medications known to affect Cys-C levels.
2.2. Sample collection
A total of 5 milliliters of human blood were obtained from each patient and were put in clot activator serum separating gel tube without anticoagulants and permitted to coagulate for half an hour at room temperature. Following a 15-minute centrifugation at 3000 rpm, the serum was separated and stored at -20°C until further analysis.
2.3. Biomarker quantification methods
Human Cys- C (E1104Hu) and IL-18 (E0147Hu) concentrations were quantified using ELISA kits (Biotek, Winooski, Vermont, USA) employing a sandwich methodology. Ca+2, P and UA levels were measured using a Mindray BS-230 clinical chemistry analyzer (Shenzhen Mindray Bio-Medical Electronics Co., Ltd, Shenzhen, China). The calibration protocols specified a two-point adjustment with a Mindray Human Multi-Calibrator and 9 g/L NaCl, traceable to the manufacturer-defined standards. Calibration was performed upon reagent lot changes or as mandated by the internal quality control (QC) protocols. QC procedures require the analysis of two-tiered control materials (Mindray Human Assayed Control recommended) per sample batch, following calibration updates, reagent cartridge replacement, or maintenance interventions. The system automates all procedural steps to ensure standardized processing.
2.4. The statistical analysis
The statistical analyses were conducted using SPSS (version 26) and the Mann-Whitney and Wilcoxon test for variables that were not normally distributed, and the dependent and independent t-tests (both two-tailed) for those that were. Statistical significance was defined as a p-value of less than 0.05.
The Socio-demographic characteristics of the studied groups show that a non-significant difference in age distribution (P = 0.96) between the group of healthy control and patients' group. Also, for the body mass index between the groups, no statistical significance was shown BMI (P = 0.61) as show in Table 1. The sex and Water source in the two groups show a non-significant difference (P = 0.13, P = 0.96) respectively. For the Residence between the groups no statistical significance was shown (P = 0.78). Regarding stone types in patients' group, it was high percentage in calcium oxalate stone (44%) and lowest in cysteine stones (8%) as shown in Figure 1. The Ph meter show a high percent of acidity (83%) and low percent of alkaline (17%) of the stones in patients as shown in Figure 2.
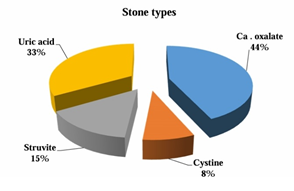
Figure 1: Types of stones and their percentage in patients' group
Figure 2: Area under curve (AUC)
A significant increase in the levels in each of Cys-C (0.368 ± 0.15 vs. 0.303 ± 0.08, P < 0.001), IL-18 (10.04 ± 4.99 vs. 7.24 ± 2.21, P < 0.001), UA (7.04 ± 1.79 vs. 4.81 ± 1.36, P < 0.001), Ca+2 (9.97 ± 0.91 vs. 9.02 ± 0.69, P < 0.001) and Phosphorus (4.89 ± 1.04 vs. 3.43 ± 0.72, P < 0.01) in patients group compared to the healthy control respectively as shown in Table 2.
As comparison between male and females in patients' group, Table 3 show a non-significant difference in Cystatin-C, Interleukin-18, Uric acid, Calcium and Phosphorus between the two groups. As comparison among age subgroup in patients' group, Table 4 show a non-significant difference in Cystatin-C, Interleukin-18, Uric acid, Calcium and Phosphorus among all groups.
Nephrolithiasis, commonly known as kidney stones, is a condition characterized by the formation of solid mineral deposits within the kidneys. These deposits can cause significant pain, urinary obstruction, and other complications. The kidneys play a crucial role in filtering waste products and maintaining fluid and electrolyte balance in the body. This study aims to investigate the biochemical and clinical parameters associated with nephrolithiasis to better understand its pathophysiology and improve diagnosis and treatment strategies.
The current results revealed a significant increase in the levels of Cys-C in patients with urolithiasis compared to control as shown in table (2). These results agree with the results of Vaidya13 who demonstrated that patients with recurrent nephrolithiasis had significantly higher serum cystatin C levels than age- and sex-matched controls. Also, it’s in agreement with research by Leem14 who confirmed that cystatin C is elevated in patients with urolithiasis-related obstruction and correlates with the severity and duration of obstruction. This correlation strengthens the argument that cystatin C is a more dynamic and responsive biomarker in acute and chronic obstructive uropathies. This result provides further evidence for the possible utility of cystatin C as a sensitive biomarker of renal dysfunction in urolithiasis. Cystatin C, a low molecular weight protein made by all nucleated cells, is filtered freely by the glomerulus and almost totally reabsorbed and catabolized in the proximal renal tubules. Due to it being constantly produced and relatively independent of age, sex, and muscle, it has become a dependable indicator for GFR, particularly in early stages of renal dysfunction.15 In patients with early-stage chronic kidney disease cystatin C levels were significantly higher in presence of normal serum creatinine, indicative of early glomerular dysfunction, which goes unnoticed by the conventional markers.16 The presence of an elevated cystatin C could reflect subclinical or early renal injury, due to obstructive uropathy, inflammation, or direct tubular injury associated with nephrolithiasis formers and passage. Nephropathy obstructed by stones can cause elevation in intratubular pressure, decrease in filtration, and, consequently, level drops in the renal function, before the serum creatinine values change.17
The level of IL-18 in this study was much higher than control group and significant higher in urolithiasis patients as presented in table (2). A study by Yao18 applied Mendelian randomization to determine the cause-effect relationship between inflammatory cytokines and urinary stones. The results revealed that the high level of IL-18 was linked with a high-risk of nephrolithiasis by promoting the formation of back stones through the increase in IL-18 levels. In another study, Lai upheld this relationship.19 Investigated IL-18 gene polymorphisms in patients with calcium oxalate renal stone; which showed strong correlation between IL-18 +105A/C gene variant and risk of urolithiasis with genetic susceptibility correlated to IL-18 activity. Other than genetic predisposition, IL-18 has been involved in renal tubular damage. The human kidney including the distal tubules and collecting ducts express the IL-18. This localization indicates that like IL-1[alpha] and IL-1[beta], IL-18 might be involved in mediating the inflammatory reaction in the kidney tubular system and may contribute to an event such as the formation of urolithiasis.20 Additionally, IL-18 has been investigated as a biomarker for renal damage. A study by Parikh21 shows increased urinary IL-18 in acute tubular necrosis, which has been linked with nephrolithiasis formation. The research suggested that IL-18 could have the role of a promising early diagnostic marker of an acute kidney injury.
These results are agreed with Ran 22 who found a significant increase in the levels of UA in people with stones compared to those without stones. It’s also in agreement with a local study in Baghdad city by Khalid & Dhahir 23 who found s significant increase in the levels of UA in both male and female patients with nephrolithiasis compared to control group According to Al-Kazwini ,24 hyperuricemia was significantly more prevalent among urolithiasis patients compared to healthy controls, suggesting a bidirectional relationship: elevated UA may promote crystal nucleation and growth, while stone-induced renal dysfunction may reduce its clearance. UA is a metabolic end-product of purine degradation, and its concentration in serum and urine is tightly regulated by renal excretion. Hyperuricemia or hyperuricosuria can contribute to the formation of UA stones through multiple mechanisms, including reduced urinary pH, low urine volume, and increased urinary UA saturation.25 When the urinary pH drops below 5.5, UA becomes less soluble and more prone to crystallization, leading to stone formation. Furthermore, and specifically, patients with metabolic syndrome or insulin resistance typically exhibit a reduction in the ability of the kidneys to excrete ammonium and maintain a stable acidic urine and thus develop UA stones.26 Such metabolic abnormalities are structurally linked to increased serous UA levels, which can support the relationship between systemic hyperuricemia and UA urolithiasis. Higher intake of both purine-rich foods, animal proteins, and fructose can lead to increased UA production and excretion resulting in a higher risk for the formation of stone.27
In a UK Biobank-based large cohort study, the authors Gao28 found that they were much more likely to develop nephrolithiasis than those with lower serum Ca+2 levels. They reported that subjects in the upper tertile of serum Ca+2 levels had a 24% higher risk of stones than those in the lower tertile, thus indicating a direct relationship between Ca+2 homeostasis and stone formation. This hypercalcemia frequently indicates other more profound endocrine or renal transport disturbances. Liu 29 recently used a mendelian randomization design and reported that genetically predicted higher serum 25-hydroxyvitamin D \[25(OH)D\] concentrations were positively associated with nephrolithiasis risk. This suggests that increased intestinal Ca+2 absorption through increased vitamin D activity could lead to higher Ca+2 in serum and urine with the precipitation of crystal along the renal tract. Together these associations between renal function and parathyroid function highlight the complex interrelationship between the kidneys and parathyroid glands, while potentially playing an important role in the development of endocrine diseases including primary hyperparathyroidism (PHPT). According to Messa30 PHPT is still a common but underrecognized etiology for hypercalcemia as well as nephrolithiasis. Their study stressed that the high-PTH receptor mediated increased renal Ca+2 reabsorption, bone resorption leading to persistently high serum Ca+2. It was concluded in this study that routine biochemical screening for serum Ca+2 and PTH in recalcitrant stone formers can detect such endocrine contributories early. In addition, molecular investigations have recently shed light on genetic and cellular determinants that may be involved in inducing such dysregulation. Mutations in the CLDN2 gene, whose protein, claudin-2, plays a role in renal Ca+2 handling, have also been associated with hypercalciuria and compensatory hypercalcemia.31 The authors speculated that abnormal Ca-sensing and -reabsorption pathways within the nephron could play a role in the development of both hypercalcemia and Ca+2 stones, especially among patients with a familial and idiopathic urolithiasis at an early age. Skeletal metabolism has also been identified as an additional source of perturbed Ca+2 homeostasis in stone formers. Rodrigues32 found in stone-forming patients that urinary Ca+2 excretion was associated with higher levels of serum sclerostin, a surrogate for bone turnover. The authors also suggested that increased bone resorption could raise serum Ca+2 and increase the filtration and excretion in the urine of Ca+2 consequently exposing the person to crystalluria and stones.
Phosphorus levels in the present study were significantly higher in urolithiasis patients than the control subjects (table 8). In a recent investigation by Zhang33 studied differential serum electrolytes in patients with and without urolithiasis. The results showed that the serum phosphorus levels of the nephrolithiasis patients were much higher than those of the healthy ones. This increase in serum phosphorus levels can lead to urinary phosphate supersaturation, which can facilitate the formation of Ca+2 phosphate stones. This study stresses the need for awareness that the serum phosphorus may be a risk factor in stone formation. A comprehensive review by Curhan34 scrutinized the association of dietary and serum phosphorus with nephrolithiasis risk. High dietary phosphorus intakes and high serum phosphorus concentrations each were independently associated with increased risk for nephrolithiasis in the study. They proposed that a high dietary intake of phosphorus could lead to hyperphosphataemia, a factor in the alteration of pH and in the precipitation process toward calculogenesis. Others, on the other hand, found no difference in levels of serum phosphorus comparing stone formers and non-stone formers. For instance, a study of Taylor35 found no significant difference in serum phosphorus between the groups. However, the authors were also able to demonstrate that the mechanism of urinary phosphorus excretion and renal phosphorus handling was more significant for stone formation than serum levels alone. Moreover, the key function of phosphorus and the relationship of this mineral with other elements, above all Ca+2, in stone formation and development are important as well. Secondary hyperparathyroidism develops from elevated serum phosphorus, which, in turn, is associated with the mobilization of Ca+2 and phosphorus. THZD, as described earlier, can lead to hypercalciuria and hyperphosphaturia, promoting stone-formation.36 Furthermore, recent studies have investigated the function of fibroblast growth factor 23 (FGF23) in phosphorus metabolism. Increased serum phosphorus concentration triggers FGF23 release that, in turn, decreases renal phosphate reabsorption and inhibits vitamin D activation. Breakdown of this regulatory pathway has been suggested to destabilize Ca+2 and phosphorus balance and promote stone formation.37
This study provides compelling evidence that patients with nephrolithiasis exhibit a distinct biochemical profile characterized by significantly elevated serum levels of Cystatin C and Interleukin-18 when compared to healthy individuals. The elevated Cys-C levels suggest the presence of subclinical or early-stage renal dysfunction, highlighting that nephrolithiasis disease may impart a greater burden on renal function than is detectable by conventional markers. Concurrently, the increased IL-18 levels support the hypothesis that a pro-inflammatory state is a critical component in the pathogenesis of nephrolithiasis, likely contributing to crystal formation and retention.
AAH: Concept, Writing, Review & Editing, Supervision, Project Administration
Authors affiliations:
- Shrouk A. H. Al.Ibraheem, Nasiriyah Technical Institute, Southern Technical University. Qrga, Sulaymaniyah, Kurdistan, Iraq; Email: shrouk.albraheem@stu.edu.iq
- Ali A. H. Albakaa, College of Health and Medical Technologies, Southern Technical University, Qrga, Sulaymaniyah, Kurdistan, Iraq; Email: alialbakaa14@stu.edu
ABSTRACT
Background & objectives: Kidney stone formation is a prevalent and recurrent condition associated with significant morbidity. While traditional risk factors are known, the roles of subclinical renal dysfunction and inflammation in its pathogenesis are still being explored. This study aimed to evaluate serum cystatin C (Cys-C) as a marker of renal function and Interleukin-18 (IL-18) as a pro-inflammatory marker in patients with kidney stones, and to assess their correlation with conventional biochemical markers.
Methods: In this case-control study, 60 patients with a confirmed diagnosis of nephrolithiasis, and 60 age- and sex-matched healthy volunteers were enrolled. Serum levels of Cys-C and IL-18 were quantified using enzyme-linked immunosorbent assay (ELISA). Serum Ca+2, phosphorus (P), and uric acid (UA) levels were measured using a clinical chemistry analyzer. Statistical analyses were performed to compare the biomarker levels between the two groups.
Results: The patient group demonstrated significantly higher serum levels of Cys-C (0.368 ± 0.15 vs. 0.303 ± 0.08 mg/dl; P < 0.001) and IL-18 (10.04 ± 4.99 vs. 7.24 ± 2.21 ng/L; P < 0.001) compared to the healthy control group. Patients with nephrolithiasis also had significantly elevated levels of UA (7.04 ± 1.79 vs. 4.81 ± 1.36 mg/dl; P < 0.001), Ca+2 (9.97 ± 0.91 vs. 9.02 ± 0.69 mg/dl; P < 0.001), and P (4.89 ± 1.04 vs. 3.43 ± 0.72 mg/dl; P < 0.001). Within the patient group, there were no significant differences in these biomarker levels based on sex or age.
Conclusion: The significant elevation of Cys-C and IL-18 in patients with nephrolithiasis suggests that subclinical renal dysfunction and pro-inflammatory processes are key components of the disease's pathophysiology. These markers, in conjunction with traditional stone-forming minerals, may serve as a valuable panel for assessing disease activity and renal involvement in patients with kidney stones.
Abbreviations: Cys-C: cystatin C, IL-18: Interleukin-18, P: phosphorus, UA: uric acid
Keywords: Nephrolithiasis; Kidney stones; Urolithiasis; Cystatin C; Interleukin-18; Renal biomarkers
Citation: Al.Ibraheem SAH, Albakaa AAH. Case control study of renal and inflammatory biomarkers in patients with urolithiasis. Anaesth. pain intensive care 2025;29(8):897-904. DOI: 10.35975/apic.v29i8.3016
Received: July 11, 2025; Revised: August 01, 2025; Accepted: August 02, 2025
1. INTRODUCTION
The urinary system plays a paramount role in homeostasis by controlling the water and electrolyte balance of the body, removing waste products, and releasing them as urine. Dysregulation of this system often manifesting as kidney stones is becoming more prevalent and is associated with severe health problems. Kidney stones are hard accumulations of crystals that are created from the supersaturation of minerals such as calcium, oxalate, or UA in the urine.1 Stones may range in size and anatomical location within the urinary system and lead to symptoms of intense pain, hematuria and urinary outflow obstruction, with potential to result in impaired renal function if not disintegrated.2
Nephrolithiasis is a universal disorder with increasing incidence due to changing lifestyles and dietary habits. Nephrolithiasis afflicts approximately 10–15% of the worldwide population at some point in their life, and the rates of recurrence reach 50% in 5–10 years.3 The disease is more prevalent in men than women, with age of onset most common between 30 and 60 years old. 2 Location differences are apparent, with a higher prevalence in warmer countries, which is associated with dehydration. The global burden of kidney stones has been increased due to increasing prevalence of obesity, diabetes, and metabolic syndrome.4
Kidney stones are the result of an interplay of complex genetic, environmental and biological factors. One of the major reasons is the supersaturation of the urine with stone-forming materials including Ca+2, oxalate, UA or P, which consecutively initiates the processes of crystal formation, growth and agglomeration.5 Genetic predisposition is also largely involved, as genetic variables like hypercalciuria or cystinuria can elevate the incidence of stones.6 Lifestyle risk factors for chronic kidney diseases (CKD) such as low fluid intake, high sodium and animal protein dietary intake also increase risk by shifting urine composition and pH.7
Cys-C, a low molecular weight protein synthesized by cells with nucleus, is considered a valuable marker of renal function and urinary system diseases. Cystatin C Unlike creatinine, cystatin C is relatively unaffected by muscle mass, age, and sex that make it a more robust indicator of GFR and early renal insufficiency.8 cystatin C has been associated with pathogenesis of kidney stones, which is a prevalent urinary system disease. Studies have indicated that cystatin C has the potential to inhibit the nucleation of calcium oxalate, a main component of kidney stones, through modulating inflammation pathways as well as decreasing oxidative stress.9
IL-18 is a pro-inflammatory cytokine of the IL-1 superfamily and has been implicated in the immune and inflammatory responses. It is produced as an inactive precursor, and needs cleaved by caspase-1 to be active.10,11 Regarding the disease of the urinary system, IL-18 has been identified to participate in the development of many kidney diseases such as acute kidney injury (AKI), chronic kidney disease (CKD) and nephrolithiasis. In Kidney Stone disease IL-18 are involved in inflammatory micro environment to allow crystal formation and to retain crystals.12 This study aimed to evaluate serum Cys-C levels as it’s related to kidney function and investigate the role of inflammatory marker (IL-18) in kidney stone and assess their correlation with conventional biochemical markers, particularly Ca+2, P and UA
2. METHODOLOGY
case-control study design was used in this study. Samples were collected at the Al-Shatrah General Hospital in Thi-Qar Province from individuals diagnosed with urinary systems disorders. The study population comprised 60 patients divided into two categories: 43 male patients and 17 female patients (diagnosis with nephrolithiasis). In addition, Control Group: Healthy volunteers (n = 60) divided into two categories: 36 male individuals and 24 female individuals from the same community as the patients. Sample collection was conducted between November 2024 and April 2025, and the study protocol was approved by the IRB (289/2024–December 12, 2024).
2.1. Inclusion and exclusion criteria
Patients were eligible for inclusion in the urinary system disorders and infection of kidney stones if they had a confirmed diagnosis of urinary system disorders, based on clinical presentation, physical examination, laboratory findings, and imaging studies. Only individuals aged 27 years or older were included. Participants were excluded if they had a history of cancer patients, people with abnormalities thyroid gland, and pregnancy women. Further exclusion criteria included the use of corticosteroids or other medications known to affect Cys-C levels.
2.2. Sample collection
A total of 5 milliliters of human blood were obtained from each patient and were put in clot activator serum separating gel tube without anticoagulants and permitted to coagulate for half an hour at room temperature. Following a 15-minute centrifugation at 3000 rpm, the serum was separated and stored at -20°C until further analysis.
2.3. Biomarker quantification methods
Human Cys- C (E1104Hu) and IL-18 (E0147Hu) concentrations were quantified using ELISA kits (Biotek, Winooski, Vermont, USA) employing a sandwich methodology. Ca+2, P and UA levels were measured using a Mindray BS-230 clinical chemistry analyzer (Shenzhen Mindray Bio-Medical Electronics Co., Ltd, Shenzhen, China). The calibration protocols specified a two-point adjustment with a Mindray Human Multi-Calibrator and 9 g/L NaCl, traceable to the manufacturer-defined standards. Calibration was performed upon reagent lot changes or as mandated by the internal quality control (QC) protocols. QC procedures require the analysis of two-tiered control materials (Mindray Human Assayed Control recommended) per sample batch, following calibration updates, reagent cartridge replacement, or maintenance interventions. The system automates all procedural steps to ensure standardized processing.
2.4. The statistical analysis
The statistical analyses were conducted using SPSS (version 26) and the Mann-Whitney and Wilcoxon test for variables that were not normally distributed, and the dependent and independent t-tests (both two-tailed) for those that were. Statistical significance was defined as a p-value of less than 0.05.
3. RESULTS
The Socio-demographic characteristics of the studied groups show that a non-significant difference in age distribution (P = 0.96) between the group of healthy control and patients' group. Also, for the body mass index between the groups, no statistical significance was shown BMI (P = 0.61) as show in Table 1. The sex and Water source in the two groups show a non-significant difference (P = 0.13, P = 0.96) respectively. For the Residence between the groups no statistical significance was shown (P = 0.78). Regarding stone types in patients' group, it was high percentage in calcium oxalate stone (44%) and lowest in cysteine stones (8%) as shown in Figure 1. The Ph meter show a high percent of acidity (83%) and low percent of alkaline (17%) of the stones in patients as shown in Figure 2.
| Table 1: Sociodemographic and anthropometric data of the study groups | ||||
| Characteristic | Patients group
(n = 60) |
Control group
(n = 60) |
P. value | |
| Age (year) | 44.63 ± 10.19 | 44.70 ± 9.36 | 0.96* | |
| BMI (kg/m2) | 24.67 ± 3.07 | 24.95 ± 2.80 | 0.61* | |
| Sex | Male | 43 (71.7) | 36 (60.0) | 0.13 ** |
| Female | 17 (28.3) | 24 (40.0) | ||
| Water source | Mineral water | 47 (78.3) | 48 (80.0) | 0.96 ** |
| Normal | 13 (21.7) | 12 (20.0) | ||
| Residence | Urban | 46 (76.7) | 48 (80.0) | 0.78 ** |
| Rural | 14 (23.3) | 12 (20.0) | ||
| Data presented as mean ± SD or n (%); *t-test, **Chi-square test; P < 0.05 is insignificant | ||||

Figure 1: Types of stones and their percentage in patients' group

Figure 2: Area under curve (AUC)
A significant increase in the levels in each of Cys-C (0.368 ± 0.15 vs. 0.303 ± 0.08, P < 0.001), IL-18 (10.04 ± 4.99 vs. 7.24 ± 2.21, P < 0.001), UA (7.04 ± 1.79 vs. 4.81 ± 1.36, P < 0.001), Ca+2 (9.97 ± 0.91 vs. 9.02 ± 0.69, P < 0.001) and Phosphorus (4.89 ± 1.04 vs. 3.43 ± 0.72, P < 0.01) in patients group compared to the healthy control respectively as shown in Table 2.
| Table 2: Cystatin-C, Interleukin-18, Uric acid, Calcium and Phosphorus levels between patients and control group | |||
| Parameters | Patient group
(n = 60) |
Control group
(n = 60) |
P- value |
| Cystatin-C (mg/dl) | 0.368 ± 0.15 | 0.303 ± 0.08 | < 0.001 |
| IL-18 (ng/L) | 10.04 ± 4.99 | 7.24 ± 2.21 | < 0.001 |
| UA (mg/dl) | 7.04 ± 1.79 | 4.81 ± 1.36 | < 0.001 |
| Ca+2 (mg/dl) | 9.97 ± 0.91 | 9.02 ± 0.69 | < 0.001 |
| P (mg/dl) | 4.89 ± 1.04 | 3.43 ± 0.72 | < 0.001 |
As comparison between male and females in patients' group, Table 3 show a non-significant difference in Cystatin-C, Interleukin-18, Uric acid, Calcium and Phosphorus between the two groups. As comparison among age subgroup in patients' group, Table 4 show a non-significant difference in Cystatin-C, Interleukin-18, Uric acid, Calcium and Phosphorus among all groups.
| Table 3: Cystatin-C, interleukin-18, uric acid, calcium and phosphorus levels in patients group according to sex | |||
| Parameters | Male group
(n = 60) |
Female group
(n = 60) |
P-value |
| Cystatin-C (mg/dl) | 0.34 ± 0.08 | 0.30 ± 0.04 | 0.22 |
| IL-18 (ng/L) | 7.05 ± 2.19 | 7.75 ± 2.24 | 0.28 |
| UA (mg/dl) | 7.20 ± 1.79 | 6.57 ± 1.79 | 0.24 |
| Ca+2 (mg/dl) | 10.03 ± 0.98 | 9.81 ± 0.72 | 0.41 |
| P (mg/dl) | 4.74 ± 1.04 | 5.31 ± 0.96 | 0.06 |
| Data are presented as mean ± SD; P < 0.05 is considered significant | |||
| Table 4: Cystatin-C, interleukin-18, uric acid, calcium and phosphorus levels in patients group according to age subgroup | ||||
| Parameters | 27-37 years | 38-48 years
|
<49 years
|
P. value |
| Cystatin-C (mg/dl) | 0.32 ± 0.08 | 0.32 ± 0.10 | 0.27 ± 0.04 | 0.06 |
| IL-18 (ng/L) | 9.86 ± 2.36 | 10.62 ± 3.47 | 9.86 ± 2.45 | 0.65 |
| UA (mg/dl) | 6.87 ± 1.64 | 7.42 ± 2.00 | 6.94 ± 1.82 | 0.64 |
| Ca+2 (mg/dl) | 10.22 ± 0.89 | 9.87 ± 0.95 | 9.82 ± 0.90 | 0.31 |
| P (mg/dl) | 4.63 ± 1.08 | 4.99 ± 1.04 | 5.05 ± 1.01 | 0.39 |
| Data are presented as mean ± SD; P < 0.05 is considered significant | ||||
4. DISCUSSION
Nephrolithiasis, commonly known as kidney stones, is a condition characterized by the formation of solid mineral deposits within the kidneys. These deposits can cause significant pain, urinary obstruction, and other complications. The kidneys play a crucial role in filtering waste products and maintaining fluid and electrolyte balance in the body. This study aims to investigate the biochemical and clinical parameters associated with nephrolithiasis to better understand its pathophysiology and improve diagnosis and treatment strategies.
The current results revealed a significant increase in the levels of Cys-C in patients with urolithiasis compared to control as shown in table (2). These results agree with the results of Vaidya13 who demonstrated that patients with recurrent nephrolithiasis had significantly higher serum cystatin C levels than age- and sex-matched controls. Also, it’s in agreement with research by Leem14 who confirmed that cystatin C is elevated in patients with urolithiasis-related obstruction and correlates with the severity and duration of obstruction. This correlation strengthens the argument that cystatin C is a more dynamic and responsive biomarker in acute and chronic obstructive uropathies. This result provides further evidence for the possible utility of cystatin C as a sensitive biomarker of renal dysfunction in urolithiasis. Cystatin C, a low molecular weight protein made by all nucleated cells, is filtered freely by the glomerulus and almost totally reabsorbed and catabolized in the proximal renal tubules. Due to it being constantly produced and relatively independent of age, sex, and muscle, it has become a dependable indicator for GFR, particularly in early stages of renal dysfunction.15 In patients with early-stage chronic kidney disease cystatin C levels were significantly higher in presence of normal serum creatinine, indicative of early glomerular dysfunction, which goes unnoticed by the conventional markers.16 The presence of an elevated cystatin C could reflect subclinical or early renal injury, due to obstructive uropathy, inflammation, or direct tubular injury associated with nephrolithiasis formers and passage. Nephropathy obstructed by stones can cause elevation in intratubular pressure, decrease in filtration, and, consequently, level drops in the renal function, before the serum creatinine values change.17
The level of IL-18 in this study was much higher than control group and significant higher in urolithiasis patients as presented in table (2). A study by Yao18 applied Mendelian randomization to determine the cause-effect relationship between inflammatory cytokines and urinary stones. The results revealed that the high level of IL-18 was linked with a high-risk of nephrolithiasis by promoting the formation of back stones through the increase in IL-18 levels. In another study, Lai upheld this relationship.19 Investigated IL-18 gene polymorphisms in patients with calcium oxalate renal stone; which showed strong correlation between IL-18 +105A/C gene variant and risk of urolithiasis with genetic susceptibility correlated to IL-18 activity. Other than genetic predisposition, IL-18 has been involved in renal tubular damage. The human kidney including the distal tubules and collecting ducts express the IL-18. This localization indicates that like IL-1[alpha] and IL-1[beta], IL-18 might be involved in mediating the inflammatory reaction in the kidney tubular system and may contribute to an event such as the formation of urolithiasis.20 Additionally, IL-18 has been investigated as a biomarker for renal damage. A study by Parikh21 shows increased urinary IL-18 in acute tubular necrosis, which has been linked with nephrolithiasis formation. The research suggested that IL-18 could have the role of a promising early diagnostic marker of an acute kidney injury.
These results are agreed with Ran 22 who found a significant increase in the levels of UA in people with stones compared to those without stones. It’s also in agreement with a local study in Baghdad city by Khalid & Dhahir 23 who found s significant increase in the levels of UA in both male and female patients with nephrolithiasis compared to control group According to Al-Kazwini ,24 hyperuricemia was significantly more prevalent among urolithiasis patients compared to healthy controls, suggesting a bidirectional relationship: elevated UA may promote crystal nucleation and growth, while stone-induced renal dysfunction may reduce its clearance. UA is a metabolic end-product of purine degradation, and its concentration in serum and urine is tightly regulated by renal excretion. Hyperuricemia or hyperuricosuria can contribute to the formation of UA stones through multiple mechanisms, including reduced urinary pH, low urine volume, and increased urinary UA saturation.25 When the urinary pH drops below 5.5, UA becomes less soluble and more prone to crystallization, leading to stone formation. Furthermore, and specifically, patients with metabolic syndrome or insulin resistance typically exhibit a reduction in the ability of the kidneys to excrete ammonium and maintain a stable acidic urine and thus develop UA stones.26 Such metabolic abnormalities are structurally linked to increased serous UA levels, which can support the relationship between systemic hyperuricemia and UA urolithiasis. Higher intake of both purine-rich foods, animal proteins, and fructose can lead to increased UA production and excretion resulting in a higher risk for the formation of stone.27
In a UK Biobank-based large cohort study, the authors Gao28 found that they were much more likely to develop nephrolithiasis than those with lower serum Ca+2 levels. They reported that subjects in the upper tertile of serum Ca+2 levels had a 24% higher risk of stones than those in the lower tertile, thus indicating a direct relationship between Ca+2 homeostasis and stone formation. This hypercalcemia frequently indicates other more profound endocrine or renal transport disturbances. Liu 29 recently used a mendelian randomization design and reported that genetically predicted higher serum 25-hydroxyvitamin D \[25(OH)D\] concentrations were positively associated with nephrolithiasis risk. This suggests that increased intestinal Ca+2 absorption through increased vitamin D activity could lead to higher Ca+2 in serum and urine with the precipitation of crystal along the renal tract. Together these associations between renal function and parathyroid function highlight the complex interrelationship between the kidneys and parathyroid glands, while potentially playing an important role in the development of endocrine diseases including primary hyperparathyroidism (PHPT). According to Messa30 PHPT is still a common but underrecognized etiology for hypercalcemia as well as nephrolithiasis. Their study stressed that the high-PTH receptor mediated increased renal Ca+2 reabsorption, bone resorption leading to persistently high serum Ca+2. It was concluded in this study that routine biochemical screening for serum Ca+2 and PTH in recalcitrant stone formers can detect such endocrine contributories early. In addition, molecular investigations have recently shed light on genetic and cellular determinants that may be involved in inducing such dysregulation. Mutations in the CLDN2 gene, whose protein, claudin-2, plays a role in renal Ca+2 handling, have also been associated with hypercalciuria and compensatory hypercalcemia.31 The authors speculated that abnormal Ca-sensing and -reabsorption pathways within the nephron could play a role in the development of both hypercalcemia and Ca+2 stones, especially among patients with a familial and idiopathic urolithiasis at an early age. Skeletal metabolism has also been identified as an additional source of perturbed Ca+2 homeostasis in stone formers. Rodrigues32 found in stone-forming patients that urinary Ca+2 excretion was associated with higher levels of serum sclerostin, a surrogate for bone turnover. The authors also suggested that increased bone resorption could raise serum Ca+2 and increase the filtration and excretion in the urine of Ca+2 consequently exposing the person to crystalluria and stones.
Phosphorus levels in the present study were significantly higher in urolithiasis patients than the control subjects (table 8). In a recent investigation by Zhang33 studied differential serum electrolytes in patients with and without urolithiasis. The results showed that the serum phosphorus levels of the nephrolithiasis patients were much higher than those of the healthy ones. This increase in serum phosphorus levels can lead to urinary phosphate supersaturation, which can facilitate the formation of Ca+2 phosphate stones. This study stresses the need for awareness that the serum phosphorus may be a risk factor in stone formation. A comprehensive review by Curhan34 scrutinized the association of dietary and serum phosphorus with nephrolithiasis risk. High dietary phosphorus intakes and high serum phosphorus concentrations each were independently associated with increased risk for nephrolithiasis in the study. They proposed that a high dietary intake of phosphorus could lead to hyperphosphataemia, a factor in the alteration of pH and in the precipitation process toward calculogenesis. Others, on the other hand, found no difference in levels of serum phosphorus comparing stone formers and non-stone formers. For instance, a study of Taylor35 found no significant difference in serum phosphorus between the groups. However, the authors were also able to demonstrate that the mechanism of urinary phosphorus excretion and renal phosphorus handling was more significant for stone formation than serum levels alone. Moreover, the key function of phosphorus and the relationship of this mineral with other elements, above all Ca+2, in stone formation and development are important as well. Secondary hyperparathyroidism develops from elevated serum phosphorus, which, in turn, is associated with the mobilization of Ca+2 and phosphorus. THZD, as described earlier, can lead to hypercalciuria and hyperphosphaturia, promoting stone-formation.36 Furthermore, recent studies have investigated the function of fibroblast growth factor 23 (FGF23) in phosphorus metabolism. Increased serum phosphorus concentration triggers FGF23 release that, in turn, decreases renal phosphate reabsorption and inhibits vitamin D activation. Breakdown of this regulatory pathway has been suggested to destabilize Ca+2 and phosphorus balance and promote stone formation.37
5. CONCLUSION
This study provides compelling evidence that patients with nephrolithiasis exhibit a distinct biochemical profile characterized by significantly elevated serum levels of Cystatin C and Interleukin-18 when compared to healthy individuals. The elevated Cys-C levels suggest the presence of subclinical or early-stage renal dysfunction, highlighting that nephrolithiasis disease may impart a greater burden on renal function than is detectable by conventional markers. Concurrently, the increased IL-18 levels support the hypothesis that a pro-inflammatory state is a critical component in the pathogenesis of nephrolithiasis, likely contributing to crystal formation and retention.
- Study limitations and future directions
- Data availability
- Ethical consideration
- Acknowledgements
- Funding Declarations
- Conflicts of interest
- Author contributions
AAH: Concept, Writing, Review & Editing, Supervision, Project Administration
13.REFERENCES
- Sorokin I, Mamoulakis C, Miyazawa K, Rodgers A, Talati J, Lotan Y. Epidemiology of stone diseaseacrosstheworld.WorldJUrol.2017;35(9):1301-1320.[PubMed] DOI: 1007/s00345-017-2008-6
- Lieske JC, Kashani K, Kellum J, Koyner J, Mehta R, Parikh CR. Use of Biomarkers to Detect and Manage Acute Kidney Injury: Has Progress Stalled?. Clin Chem. 2020;66(2):271-276. [PubMed] DOI: 1093/clinchem/hvz026
- Taguchi K, Cho SY, Ng AC, et al. The Urological Association of Asia clinical guideline for urinary stone disease. Int J Urol. 2019;26(7):688-709.[PubMed] DOI: 1111/iju.13957
- Meister MR, Zhou J, Chu H, et al. Non-invasive bladder function measures in healthy, asymptomatic female children and adolescents: A systematic review and meta-analysis. J Pediatr Urol. 2021;17(4):452-462.[PubMed] DOI: 1016/j.jpurol.2021.04.020
- Assimos D, Krambeck, A., Miller, N. L., Monga, M., Murad, M. H., Nelson, C. P., ... & Turk, T. M. Surgical management of stones. American Urological Association/Endourological Society guideline Journal of Urology. 2020;204(6):1163–9. [PubMed] DOI: 1016/j.juro.2016.05.090
- Dutta R, Mithal P, Klein I, Patel M, Gutierrez-Aceves J. Outcomes and Costs Following Mini-percutaneous Nephrolithotomy or Flexible Ureteroscopic Lithotripsy for 1-2-cm Renal Stones: Data From a Prospective, Randomized Clinical Trial. J Urol. 2023;209(6):1151-1158. [PubMed] DOI: 1097/JU.0000000000003397
- Ferraro PM, Taylor EN, Gambaro G, Curhan GC. Dietary and Lifestyle Risk Factors Associated with Incident Kidney Stones in Men and Women. J Urol. 2017;198(4):858-863.[PubMed] DOI: 1016/j.juro.2017.03.124
- Inker LA, Schmid CH, Tighiouart H, et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med. 2012;367(1):20-29. doi:10.1056/NEJMoa1114248[PubMed] DOI: 1056/NEJMoa1114248
- Tamborino F, Cicchetti R, Mascitti M, Litterio G, Orsini A, Ferretti S, Basconi M, De Palma A, Ferro M, Marchioni M, et al. Pathophysiology and Main Molecular Mechanisms of Urinary Stone Formation and Recurrence. International Journal of Molecular Sciences. 2024; 25(5):3075.[PubMed] DOI: 3390/ijms25053075
- Liang H, Xu F, Zhang T, et al. Inhibition of IL-18 reduces renal fibrosis after ischemia-reperfusion. Biomed Pharmacother. 2018;106:879-889. [PubMed] DOI: 1016/j.biopha.2018.07.031 Jalood HH, and Krarr Haider Haddawi. GSTM1 and GSTT1 null genotypes as risk factors for chronic kidney disease. Anaesthesia, Pain & Intensive Care. 2025;29[FullText]
- Hirooka Y, Nozaki Y. Interleukin-18 in Inflammatory Kidney Disease. Front Med (Lausanne).2021;8:639103[PubMed]DOI: 3389/fmed.2021.639103
- Vaidya VS, Ferguson, M. A., & Bonventre, J. V. Biomarkers of acute kidney injury. Annual Review of Pharmacology and Toxicology.2011(48):463–93.[PubMed] DOI: 1146/annurev.pharmtox.48.113006.094615
- Leem AY, Park MS, Park BH, et al. Value of Serum Cystatin C Measurement in the Diagnosis of Sepsis-Induced Kidney Injury and Prediction of Renal Function Recovery. Yonsei Med J. 2017;58(3):604-612.[PubMed] DOI: 3349/ymj.2017.58.3.604
- Singh DP, Mondal S, Sarkar D. Assessment of Plasma Cystatin C as a Marker of Acute Renal Injury in Patients Undergoing Extracorporeal Shock Wave Lithotripsy for Renal Stone Disease. Cureus. 2024;16(8):e67293.. [PubMed] DOI: 7759/cureus.67293
- Javanmardi M, Azadi NA, Amini S, Abdi M. Diagnostic value of cystatin C for diagnosis of early renal damages in type 2 diabetic mellitus patients: The first experience in Iran. J Res Med Sci. 2015;20(6):571-576. [PubMed] DOI: 4103/1735-1995.165960
- Wasung ME, Chawla, L. S., & Madero, M. . Biomarkers of renal function, which and when? . Clinica Chimica Acta. 2015;438:350–7. [PubMed]DOI: 1016/j.cca.2014.08.039
- Yao X, Wang, Y., Zhang, Y., & Li, Y. Causal associations of inflammatory cytokines and urinary stones: a two-sample Mendelian randomization study. Translational Andrology and Urology. 2023;12(1):45–54. [PubMed] DOI: 21037/tau-24-445
- Lai KC, Lin WY, Man KM, et al. Association of interleukin-18 gene polymorphisms with calcium oxalate kidney stone disease. Scand J Urol Nephrol. 2010;44(1):20-26. doi:10.3109/00365590903449332[PubMed] DOI: 3109/00365590903449332
- Gauer S, Sichler O, Obermüller N, et al. IL-18 is expressed in the intercalated cell of human kidney. Kidney Int. 2007;72(9):1081-1087. [PubMed]DOI: 1038/sj.ki.5002473 Parikh CR, Jani, A., Melnikov, V. Y., Faubel, S., & Edelstein, C. L. . Urinary interleukin-18 is a marker of human acute tubular necrosis. American Journal of Kidney Diseases. 2004;43(3):405–14. [PubMed] DOI: 10.1053/j.ajkd.2003.10.040
- Ran YML, Zhenyu MMa; Ma, Hang MMb; Li, Chaofan BMedc; Zhou, Jiangtao MMa; Li, Dongyang MMa; Guo, Ziqi BMedd; Dan, Lifeng MMa; Zhao, Zecang MMa; Zou, Zhonglin MMa; Su, Shuai PhDa; Yin, Zhikang PhDa. Associations between 25-hydroxyvitamin D/calcium/alkaline phosphatase levels and the risk of developing kidney stones: Results from NHANES (2013–2018)-based and Mendelian randomization studies. Medicine 2025;194(4). [PubMed] DOI: 1097/MD.0000000000041323
- Khalid F, Ahmed Dhahir S. Evaluation of Minerals and some Biochemical parameters in the serum of Renal stones from Iraqi patients. IJApSc. 2024;1(3):11-16.[FullText]
- Al-Kazwini AT, Al-Arif, M. S., & Handal, H. R. . Relationship between hyperuricemia and nephrolithiasis: A case-control study. Journal of Clinical Urology. 2019;12(2):130–6. [PubMed]DOI: 1053/j.ajkd.2017.01.053
- Siener R, Löhr P, Hesse A. Urinary Risk Profile, Impact of Diet, and Risk of Calcium Oxalate Urolithiasis in Idiopathic Uric Acid Stone Disease. Nutrients. 2023;15(3):572. Published 2023 Jan 21.[PubMed] DOI: 3390/nu15030572
- Maalouf NM, Cameron, M. A., Moe, O. W., & Sakhaee, K. . Metabolic basis for low urine pH in type 2 diabetes. Clinical Journal of the American Society of Nephrology. 2011;6(11):2529–34. [PubMed] DOI: 2215/CJN.08331109
- Curhan GC, Taylor EN. 24-h uric acid excretion and the risk of kidney stones. Kidney Int. 2008;73(4):489-496. [PubMed] DOI: 1038/sj.ki.5002708
- ao C, Gao M, Huang Y. Association between serum 25-hydroxyvitamin D concentrations and kidney stone: a cohort study in the UK Biobank. Int Urol Nephrol. 2024;56(11):3585-3594. [PubMed] DOI: 1007/s11255-024-04111-8
- Liu M, Wu J, Gao M, et al. Lifestyle factors, serum parameters, metabolic comorbidities, and the risk of kidney stones: a Mendelian randomization study. Front Endocrinol (Lausanne). 2023;14:1240171.[PubMed] DOI: 1007/s11255-024-04111-8
- Messa P, Castellano, G., Vettoretti, S., Alfieri, C. M., Giannese, D., Panichi, V., & Cupisti, A. . Vitamin D and calcium supplementation and urolithiasis: A controversial and multifaceted relationship. . Nutrients. 2023;15(7):1724.[PubMed]DOI: 3390/nu15071724
- Ansari Djafari A, Moradi, M., Malek Mohammadi, M., Javanmard, B., & Rismanchi, H. Correlation of calcium metabolic axis with urinary stone characteristics. Nephro-Urology Monthly.2024;15(4):e142398.[FullText]
- Rodrigues FG, Ormanji, M. S., Pietrobom, I. G., Matos, A. C. C. d., De Borst, M. H., & Heilberg, I. P. Urinary calcium is associated with serum sclerostin among stone formers. Journal of Clinical Medicine. 2023;12(15):5027. [PubMed] DOI: 3390/jcm12155027
- Zhang Z, Xu, Q., Huang, X., Liu, S., & Zhang, C. Preliminary analysis of serum electrolytes and body mass index in patients with and without urolithiasis. Journal of International Medical Research. 2020;48(6). [PubMed]DOI: 1177/0300060520925654
- Burhan GC, Willett, W. C., Rimm, E. B., & Stampfer, M. J. . Dietary phosphorus intake and the risk of kidney stones. Journal of Urology. 2022;208(1):50–6.[PMC] doi: 1146/annurev-nutr-071816-
- Taylor EN, Stampfer, M. J., & Curhan, G. C. . Plasma and urinary phosphorus levels and risk of kidney stones. American Journal of Kidney Diseases. 2023;81(4):567–74. [PMC] doi: 2215/CJN.07060714
- Moe SM. Calcium Homeostasis in Health and in Kidney Disease. Compr Physiol. 2016;6(4):1781-1800. Published 2016 Sep 15. [PubMed] DOI: 1002/cphy.c150052
- Shimada T, Hasegawa H, Yamazaki Y, et al. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res. 2004;19(3):429-435. [PubMed] DOI: 1359/JBMR.0301264