Wael Sayed Algharabawy 1 , Khaled Abdou 2 , Mariam Kamal 3 , Marwa Salem 4
Authors affiliations:
Background & objective: We compared the efficacy of ibuprofen versus dexmedetomidine in combination with bupivacaine or bupivacaine alone in intraperitoneal local anesthetic (IPLA) installation for postoperative pain control after total laparoscopic hysterectomy (TLH).
Methodology: One hundred eighty female patients scheduled for total laparoscopic hysterectomy were randomly assigned to one of three groups to receive intraperitoneal injections of either 50 mL of bupivacaine 0.25% (group B), 50 mL of bupivacaine 0.25% with dexmedetomidine 1 µg/kg (Group BD), or 50 mL of bupivacaine 0.25% with ibuprofen 400 mg (Group BI). In this randomised controlled trial, we evaluated the severity of postoperative pain using a visual analogue scale (VAS). Additionally, hemodynamic changes, total postoperative opioid consumption within the first 24 hours, incidence of postoperative nausea and vomiting, patient satisfaction, sedation score, time to the first request for analgesia, onset of ambulation, and side effects were recorded.
Results: Group BI demonstrated significantly lower pain scores at 6 and 12 hours, both at rest and during movement, compared with Groups BD and B. Group BD also showed lower pain scores than Group B at 6 hours at rest and during movement (P < 0.009 and P < 0.002, respectively). The time to first analgesic request was significantly longer in Group BI than in Groups BD and B (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours, respectively). Postoperative cumulative opioid consumption was lower in Group BI compared with Groups BD and B. Sedation scores were higher in Group BD only at 2 hours postoperatively.
Conclusion: Bupivacaine-ibuprofen was superior to bupivacaine-dexmedetomidine and bupivacaine alone. This was achieved with no significant negative consequences.
Keywords: Laparoscopy; Hysterectomy; Ibuprofen; Dexmedetomidine; Bupivacaine; Pain; Postoperative.
Citation: Algharabawy WS, Abdou K, Kamal M, Salem M. Efficacy of adding dexmedetomidine versus ibuprofen as an adjuvant to intraperitoneal bupivacaine for pain control after laparoscopic gynecological procedures: a double-blind randomized comparative study. Anaesth. pain intensive care 2025;29(8):831-840. DOI: 10.35975/apic.v29i8.3004
Received: February 02, 2025; Revised: September 26, 2025; Accepted: September 28, 2025
Laparoscopic surgeries are becoming increasingly popular due to their fast-track nature, which promotes early patient recovery by reducing postoperative somatic and muscular pain components, facilitating early mobilization, rapid oral intake, shorter hospital stays, and greater patient satisfaction.Error! Reference source not found.
Hysterectomy is a common gynecological operation. Traditionally, this has been accomplished through the abdominal or vaginal routes. However, hysterectomies are increasingly being performed using minimal access techniques.Error! Reference source not found.
Postoperative abdominal and shoulder pain are common after total laparoscopic hysterectomy (TLH). Pain is due to a variety of factors, including visceral pain caused by abdominal stretching, surgical manipulations that result in peritoneal and visceral inflammation, and peritoneal irritation caused by pneumoperitoneum. Shoulder tip pain is caused by phrenic nerve irritation caused by CO2 trapped beneath the diaphragm.Error! Reference source not found.
Various analgesic techniques and drugs were used to control postoperative pain after laparoscopic surgeries, which may require systemic opioid analgesia but result in sedation and delayed return of bowel function, resulting in delayed hospital discharge. Other techniques include non-opioid analgesia, regional analgesia, regional block techniques, and intraperitoneal administration of local anesthetics (IPLA).Error! Reference source not found.
It has been observed that postoperative pain after laparoscopic surgery can be decreased by intraperitoneal instillation of local anesthetic agents either alone or in combination with opioids and α2 agonists like clonidine and dexmedetomidine. Bupivacaine is thought to block free afferent nerve endings in the peritoneum, thereby inhibiting visceral nociceptive conduction. Local anesthetics block nociception by affecting nerve membrane-associated proteins and inhibiting the release and action of prostaglandins and other agents that sensitize nociceptors and contribute to inflammation. However, systemic absorption of local anesthetic from the peritoneal cavity may also play a role in reduced nociception; this could be another mechanism of analgesia.Error! Reference source not found.
Dexmedetomidine is a selective alpha-2 (α2) adrenergic agonist with analgesic and sedative properties that can prolong the action of local anesthetics. Dexmedetomidine acts over peritoneal neural receptors and blocks nociceptive stimuli due to its high lipophilic nature.Error! Reference source not found.
Nonsteroidal anti-inflammatory drugs (NSAIDs) have been found to have both central and peripheral effects; however, local application to the site of injury may result in effective concentration in the inflamed tissues, resulting in a decreased fibroproliferative inflammatory response, which results in decreased postoperative adhesions and intestinal distension with minimal adverse effects.Error! Reference source not found.
Although NSAIDs are not a local anesthetic agent, a COX inhibitor reduces the secretion of pain mediators in the areas where surgical manipulation is applied, and it also has a local anti-nociceptive effect in addition to its analgesic effect.Error! Reference source not found.
Ibuprofen, a propionic acid derivative, is a nonsteroidal anti-inflammatory drug (NSAID) with anti-inflammatory, antipyretic, and analgesic properties similar to other agents in its class. Oral ibuprofen has long been recognized as one of the most commonly used and safest NSAIDs.Error! Reference source not found.The US FDA authorized an intravenous (IV) formulation of ibuprofen in 2009 (Caldolor®, Cumberland Pharmaceuticals, Inc., TN, USA) for the management of pain and reduction of fever in adults.Error! Reference source not found.
This study is the first to evaluate the use of intraperitoneal ibuprofen as an adjuvant to bupivacaine, in comparison with dexmedetomidine, and with bupivacaine alone, for postoperative analgesia following total laparoscopic hysterectomy (TLH), with respect to efficacy, side effects, and patient satisfaction.
Ethical approval for this study was obtained from the Research Ethics Committee (REC), Faculty of Medicine, FWA 000017585, REC-FMASU@med.asu.edu.eg on May 22, 2023 (FMASU R131/2023). The clinical trial registry (NCT06046105) coded this prospective, double-blind, randomized, controlled study on September 14, 2023. The study was conducted at Ain Shams University Hospitals from October 2023 to June 2024. Written informed consent to participate was obtained from all of the participants in the study. The study enrolled 180 patients who met the inclusion criteria and were scheduled for total laparoscopic hysterectomy (TLH) in the Gynaecology and Obstetrics theatre at Ain Shams University Hospitals. Inclusion criteria included age between 18 and 80 years, female sex, BMI between 18.5 and 35 kg/m2, and ASA physical status I-II. Exclusion criteria included age < 18 or more than 80 years, BMI > 40 or <18.5 kg/m2, ASA III-IV, allergy to one of the agents used, local infection at the port site, coagulopathy, severe cardiac (NYHA III) or pulmonary dysfunction (known COPD, previous thoracic surgeries, or recent pulmonary infection), severe hepatic impairment (Child C) (INR >2, albumin <2.5), severe renal dysfunction (creatinine clearance <30), conversion to open surgery, emergency operations, neurologic, psychiatric or mental illness, or refusal to participate in the study.
Using a computer-generated random number table, the study subjects were randomly assigned to three equal groups. One investigator assessed the patients' eligibility and obtained written informed consent from them during the pre-anesthetic interview the day before surgery. The primary investigator was responsible for opening opaque envelopes with sequential numbers that contained the allocation of groups. The study group assignments were blinded to both the participants and the outcome assessors. Group B (bupivacaine only) (n = 60) was given an intraperitoneal injection of 50 mL of diluted Bupivacaine 0.25 percent (125 mg) in 100 mL of normal saline. Group BD (bupivacaine-dexmedetomidine) (n = 60), assigned for intraperitoneal administration of 50 mL bupivacaine 0.25 percent (125 mg) with dexmedetomidine 1 µg/kg diluted in 100 mL normal saline. Group BI (Bupivacaine-Ibuprofen) (n = 60), assigned for intraperitoneal administration of 50 mL Bupivacaine 0.25% (125 mg) with ibuprofen 400 mg diluted in 100 mL normal saline. Drugs mentioned previously were injected intraperitoneally through trocars at the surgical end.
In this study, a simple spraying cannula attached to a 20-mL syringe was used for intraperitoneal instillation. This method is readily available, time-efficient, and eliminates the need for expensive or complex devices to aerosolize or nebulize the local anesthetic.8 There is no previous evidence of using ibuprofen intraperitoneally in combination with bupivacaine in humans; however, it has been administered intraperitoneally in mice to determine equivalent doses of ibuprofen delivered through drinking water or diet.9 Therefore, a mixture of bupivacaine and ibuprofen was prepared under aseptic conditions using 0.9% NaCl as a diluent, while avoiding alkalinization and carefully inspecting for any precipitation. The solution was used immediately. This approach aligns with standard pharmaceutical compounding practices when formal stability data are unavailable.
All patients underwent a standard preoperative evaluation. Upon arrival in the preoperative holding area, a thorough history was obtained, followed by a complete physical examination, laboratory investigations, and other necessary assessments. During the preoperative visit, all patients were instructed on the use of the visual analogue scale (VAS) for pain assessment at rest and during movement, with scores ranging from 0 (no pain) to 10 (worst imaginable pain).
A crystalloid solution IV drip was started after an 18-gauge IV cannula was inserted. All patients were given IV antibiotics and Granisetron 3 mg. Standard monitoring was used in the operating room, including ECG, non-invasive blood pressure, and pulse oximetry. The following baseline parameters were also recorded: mean blood pressure (MBP), heart rate (HR), and arterial oxygen saturation (SpO2). Following pre-oxygenation, IV 2 mg/kg propofol, atracurium 0.5 mg/kg, and 2 µg/kg fentanyl were used to induce general anesthesia. Following endotracheal intubation, anesthesia was maintained with 1-1.5 percent isoflurane in a FiO2 0.5 oxygen, air mixture to keep the hemodynamics within 20% of basal values. IV boluses of fentanyl (up to 3 µg/kg) were administered, and muscle relaxation was maintained with IV 0.1 mg/kg atracurium boluses every 30 minutes.
To maintain normocarbia, mechanical ventilation parameters were adjusted to keep the end-tidal CO2 (EtCO2) between 30-35 mmHg. Hypotension was defined as having a mean blood pressure (MBP) of less than 60 mmHg, and bradycardia as having a heart rate (HR) of less than 50 beats per minute. IV boluses of 0.1 mg/kg ephedrine were used to treat hypotension, and IV 0.01 mg/kg atropine was used to treat bradycardia. The maximum insufflation pressure of CO2 was 15 mmHg.
The surgeon administered intraperitoneal injections via trocars at the end of the procedure, after which discontinuation of isoflurane was done, and the neuromuscular blockage was antagonised by using neostigmine 0.05 mg/kg and atropine 0.01 mg/kg after assessment of nerve stimulator following train of four, and was administered to reverse atracurium effect. Patients were transferred to the post-anesthetic care unit (PACU) after the surgical procedure was completed and the extubation criteria were met. Standard monitoring was used in the PACU, and readings were documented, and the level of pain was measured. After achieving an Aldrete score = 9, the patient was assessed and discharged. The patient was transferred to the ward for ongoing monitoring and management throughout the study.
To maintain blinding, a separate anesthesiologist who was not involved in the study prepared the study drugs in injectable form. Consequently, both the patients and the anesthesiologist administering the medication were blinded to the group allocation. In addition, the physician responsible for data collection was also unaware of the administered drugs. All patients were given a post-operative pain protocol during the first 24 hours post-surgery (1 gm paracetamol IV every 8 hr). Meperidine (Pethidine) 0.5 mg/kg, which was diluted in 5mL, was titrated intravenously as a rescuing analgesic if VAS was greater than three or when the patient required additional analgesia at any time. Complications such as nausea and vomiting postoperatively, as well as other drug-related complications (e.g., toxicity) were recorded till 24 hours post-surgery. Postoperative abdominal pain intensity was measured using a 10-cm linear visual analogue scale at 2, 4, 6, 12, and 24 h intervals (VAS). The time of the first analgesic request (rescue analgesia) of pethidine, total postoperative opioid consumption, side effects such as nausea, vomiting, sweating, dizziness, tinnitus, muscular twitches, and circumoral numbness, onset of ambulation, and modified RAMSAY sedation score were all recorded at equal time intervals. Patients who were converted to open surgery or in whom drains were placed were excluded from the study.
The primary outcome of this study was the measurement of pain severity on a Numerical Rating Scale (NRS) ranging from 0 to 10. A 10-cm scale was used to assess pain intensity, with 0 representing no pain and 10 representing the most severe pain, measured at 2, 4, 6, 12, and 24 hours postoperatively at rest and during
movement. Secondary outcomes included cumulative postoperative opioid use in the first 24 hours postoperatively, the initial analgesic request, postoperative nausea and vomiting, the incidence of complications (e.g., LA toxicity), patient satisfaction after the first postoperative day (1 = excellent, 2 = good, 3 = fair, and 4 = poor), patients' hemodynamics, and the onset of ambulation.
2.1. Statistical analysis
A sample size of 58 patients per group was calculated to detect an effect size of 0.8 for VAS scores between the study groups, with a two-sided α error of 0.01 and a statistical power of 0.95. Statistical Package for the Social Sciences (SPSS) version 27 was used to analyze the data. Quantitative data were represented as mean, standard deviation (SD), or median (IQR) when indicated. Percentage and frequency were used to express qualitative data. The Chi-square (X2) test of significance was employed to compare proportions between two qualitative measures. The post-hoc test was used for pairwise comparison of subgroups when the ANOVA test was positive. One-way analysis of variance (ANOVA) was used to evaluate differences within the means of various subgroups of a variable (multiple testing). The Kruskal–Wallis test was used to compare multiple subgroups for non-parametric data. When a significant difference was detected, pairwise comparisons were performed. The confidence interval was set at 95%, and the margin of error was set at 5%. Accordingly, P-values less than 0.05 were considered statistically significant.
A total of two hundred patients were enrolled in the trial; 11 patients refused to participate, therefore a total of 189 patients were randomly assigned to one of the three groups (B-BD-BI). Three patients from each group did not participate in the study due to either the insertion of drains or conversion into open surgery. The study adheres to CONSORT guidelines. Each group included 60 patients who were analyzed in this study. Age, body mass index (BMI), duration of surgery, and ASA physical status (I–II) were comparable among the three groups, with no significant differences. Heart rate and mean arterial pressure were also comparable across all groups and remained within normal ranges (Table 1). Total postoperative opioid consumption was lower in Group BI (69.17 ± 32.0 mg) than in Groups BD (88.33 ± 39.5 mg) and B (93.33 ± 40.6 mg), and this difference was statistically significant (P = 0.001) (Table 1). The time to the first request for analgesia was significantly longer in the BI group compared with the BD and B groups (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours, respectively; P < 0.001). Although the duration was longer in the BD group than in the B group, the difference was not statistically significant (Table 1).
There was no significant difference between the three groups in terms of ambulation onset (146.25 ± 19.7, 147.13 ± 17.4, 144.08 ± 20.5 minutes), respectively. Using a four-point rating system (1 = excellent, 2 = good, 3 = fair, and 4 = poor), patient satisfaction was equivalent across the three groups, and there was no significant difference (P < 0.548) (Table 1). The incidence of postoperative nausea and vomiting was comparable in all three groups till the end of the study and was statistically insignificant. Sedation was evaluated using the Modified Ramsay sedation score. Sedation was higher in the BD group only at 2 hours postoperative measurement and was statistically significant (P ˂ 0.018) compared to the other two groups. However, the sedation score was never greater than three, indicating that all patients were arousable and did not require any intervention (Table 2). Postoperative VAS at rest after 6 hours was considerably lower in Group BI compared to Groups B and BD. It was also significantly lower in Group BD compared to Group B. At 12 hours, the VAS score at rest in the BI group was significantly lower than in the other two groups, with P-values of 0.001 and 0.031, respectively. However, there was no significant difference between groups B and BD. During movement, the VAS score at 6 hours was significantly lower in the BI group compared with the other two groups, and it was also lower in the BD group compared with the B group. The VAS score in the BI group remained significantly lower than in the other two groups (B and BD), which were comparable (Table 2).
Postoperative pain management following laparoscopic gynecological surgeries is a significant concern. Postoperative discomfort arises from distention of the abdominal wall and peritoneum, irritation of the diaphragm and peritoneum due to residual gas, and visceral pain resulting from surgical manipulation.Error! Reference source not found. Several methods have been employed to reduce postoperative discomfort after laparoscopic procedures, including intraperitoneal closed suction drains, intraperitoneal injection of local anesthetics (IPLA), transversus abdominis nerve block, epidural analgesia, and other regional analgesia and block techniques. The intraperitoneal route of administration of local anesthetics (LA) is simple, safe, and promotes ambulatory anesthesia.Error! Reference source not found.
Adjuvants added to IPLA may increase effectiveness, lengthen duration, improve outcome, and give greater analgesia.Error! Reference source not found.Many medications are utilized as adjuvants to IPLA, such as dexmedetomidine, opioids like tramadolError! Reference source not found.or fentanylError! Reference source not found. or NSAIDS like tenoxicamError! Reference source not found.or ketorolac.Error! Reference source not found.
Dexmedetomidine, an α 2-adrenergic agonist, has been introduced into clinical anesthesia as a sedative, sympatholytic, hemodynamic-stabilizing, and anesthetic-sparing agent. Its effects are centrally mediated via α2-adrenoceptors, which are found in the spinal cord's dorsal horn and locus coeruleus.Error! Reference source not found. The side effects of dexmedetomidine include bradycardia without respiratory depression, nausea, hypertension, and hypotension.Error! Reference source not found.
Ibuprofen is one of the most often used analgesic-antipyretic-anti-inflammatory medications. Like many traditional NSAIDs, ibuprofen inhibits both the constitutive cyclo-oxygenase-1 (COX-1), which is responsible for the synthesis of prostanoids (PGs, and thromboxane-A2) that control a range of physiological functions (vascular, blood flow, gastric and renal functions), and the inducible COX-2, whose synthesis is increased, leading to augmented production of PGE2 in inflammation and pain. Error! Reference source not found.
The intravenous formula of ibuprofen is a racemic combination of [-] R- and [+] S-isomers, with the [+] S-isomer responsible for the drug's efficacy and the [-] R-isomer acting as an inactive reservoir that is actively converted to the active [+] S-isomer. Intravenous ibuprofen has a very varied volume of distribution based on the patient's age and health. It is almost 99% plasma proteins bound at 20 µg/mL. Error! Reference source not found.
Significant pharmacological characteristics of racemic ibuprofen, which has a 50:50 mixture of the two isomers, are derived from the stereochemical configuration of R (-)- and S (+)-ibuprofen. When compared to the active S (+) enantiomer, R (-)-ibuprofen is a weaker inhibitor of COX-1 and COX-2, which has an impact on prostaglandin synthesis and thromboxane A2.24 R (-)-ibuprofen, on the other hand, is an effective inhibitor of leukotrienes and other products produced by activated polymorphonuclear leukocytes. R (-)-ibuprofen also suppresses the formation of anandamide, an endogenous cannabinoid that causes analgesia in the central nervous system. Moreover, R (-)-ibuprofen reduces mucosal irritation from COX-1 in the stomach by competing with S (+)-ibuprofen for the binding of this enzyme. However, upon absorption from the upper GI tract, 40–60% of R (-)-ibuprofen is changed to the S (+) isomer, resulting in a "ballast" of the latter's reserves in the body. Error! Reference source not found.
Like some other NSAIDs, ibuprofen acts as an analgesic through both PG-dependent Error! Reference source not found. and PG-independent Error! Reference source not found. pathways. Prostaglandin E2 (PGE2), prostaglandin I2 (PGI2), and prostaglandin D2 (PGD2) interact with their corresponding EP, IP, or DP receptors on nerves to activate certain channels or enzymes that control the neuronal transmission of pain signals. These interactions are the basis for prostaglandin-dependent mechanisms. Error! Reference source not found. The N-type CaV 2.2 channels, glutamate, N-methyl-D-aspartate (NMDA) receptors, neurokinin (NK1) receptors, gamma-aminobutyric acid (GABAA and GABAB) receptors, glycine receptors, and adenosine P2X3 receptors are non-PG central (dorsal horn) targets involved in pain modulation. Error! Reference source not found. Ibuprofen reduces neuronal hyperexcitability by blocking alterations in the NaV 1.7 and 1.8 channels. Error! Reference source not found.
This study was the first to evaluate the use of intraperitoneal ibuprofen as an adjuvant to bupivacaine, in comparison with dexmedetomidine, and with bupivacaine alone, for postoperative analgesia following total laparoscopic hysterectomy (TLH), with respect to efficacy, side effects, and patient satisfaction. We found that administering Ibuprofen-bupivacaine (Group BI) intraperitoneally reduced postoperative pain scores significantly, decreased postoperative cumulative opioid consumption, and extended the time to first request analgesia when compared to administering bupivacaine-dexmedetomidine (Group BD) or Bupivacaine alone (Group B).
The postoperative VAS was statistically significant (P < 0.001 and 0.031) after 6 and 12 hours at rest, and (P < 0.001 and 0.008) at 6 and 12 hours on movement, respectively (Table 2), favoring better pain management in the Ibuprofen-bupivacaine (BI group). After 24 hours, the bupivacaine-ibuprofen group consumed fewer opioids (69.17 ± 32.0 mg) than the bupivacaine-dexmedetomidine group (88.33 ± 39.5 mg) or bupivacaine alone (93.33 ± 40.6 mg), with a statistically significant difference (P = 0.001). This might be attributed to ibuprofen's extended half-life. Error! Reference source not found. Thus, emphasizing the idea that combined therapy may produce much better analgesia. Furthermore, there was a statistically significant (P < 0.001) difference in the duration of time until the first request for analgesia (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours) between the BI group and the BD and B alone groups. This might be due to the anti-inflammatory and analgesic qualities of ibuprofen; also, local use of NSAIDs may result in effective and fast concentration levels in inflamed tissues with less systemic adverse effects. Error! Reference source not found. A lower VAS score and lower total opioid use in group BI resulted in fewer opioid-related adverse effects, such as dizziness, drowsiness, and delayed bowel movement. Gupta et al. Error! Reference source not found. found that administering 1 µq/kg dexmedetomidine with 125 mg bupivacaine intraperitoneally during laparoscopic gynecological surgeries reduced VAS, postoperative analgesic requirements, and increased time to first request postoperative morphine. The findings of this study support it.
Numerous studies indicate that NSAIDs and bupivacaine together provide sufficient analgesia and opioid-sparing effectiveness during intraperitoneal instillation for various surgical procedures. El Hakim et al. revealed the excellent intraperitoneal instillation efficacy of lidocaine and tenoxicam that lowered discomfort with movement over 24 hours following laparoscopic cholecystectomy compared to the control group. Patients in the tenoxicam intraperitoneal group required less nalbuphine than the control group, demonstrating that intraperitoneal combination therapy produces superior analgesia. Error! Reference source not found. The bupivacaine-dexmedetomidine (BD) group had significantly higher sedation scores than the other two groups in the first two hours (P = 0.018). This could be attributed to its α2 adrenergic agonist properties, which cause sympatholysis, anxiolysis, and sedation while maintaining respiratory functions. The sedation scores were similar between bupivacaine-ibuprofen (Group BI) and bupivacaine alone (Group B).
The results were consistent with other results done by Shukla et al. 15 who examined the effects of intraperitoneal bupivacaine alone (B), in combination with either dexmedetomidine (BD) or tramadol (BT), during laparoscopic cholecystectomy. Compared to bupivacaine 0.25% alone, intraperitoneal instillation of dexmedetomidine 1 μ/kg in conjunction with bupivacaine 0.25% significantly reduces post-operative pain and the need for analgesics during the post-operative period; this combination may even prevail over bupivacaine combined with tramadol. Bakhamees et al. Error! Reference source not found. also investigated the intraoperative dexmedetomidine infusion effect compared to placebo on the total amount of intraoperative fentanyl and propofol required to maintain anesthesia, the recovery profile, the pain score, and the total amount of morphine used via patient-controlled analgesia. They found that the total amounts of propofol and fentanyl required to maintain anesthesia were reduced, intraoperative and postoperative hemodynamic stability was improved, postoperative pain was lower, and the total morphine consumption was decreased.
Moreover, recovery was better compared to the placebo group. The incidence of postoperative nausea and vomiting did not differ significantly among the three groups (P = 0.624), consistent with earlier studies evaluating postoperative nausea and vomiting following intraperitoneal administration of bupivacaine–dexmedetomidine32 or bupivacaine–tenoxicam.17 No signs of local anesthetic systemic toxicity were observed during the study.
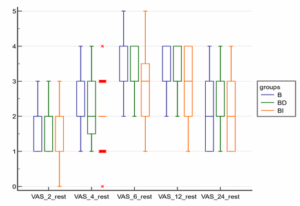
Figure 1: Box and whisker graph between the groups as regards VAS at rest
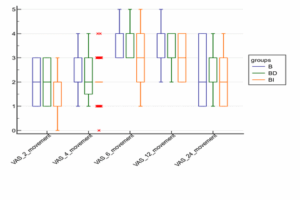
Figure 2: Box and whisker graph between the groups regarding VAS at movement
The study's limitations include the fact that the visual analogue score is not an objective indicator, but it is clear and reliable. Additionally, the study was conducted in a single center, comparing the three groups of pain medications, including bupivacaine alone, bupivacaine with dexmedetomidine, and ibuprofen. To the best of our knowledge, our study is the first to evaluate the effects of combining ibuprofen with bupivacaine for intraperitoneal instillation to control postoperative pain and enhance recovery criteria. Thus, more analytical studies involving a larger number of patients, as well as the establishment of large multi-center databases, are required to evaluate efficacy and modify the effective dosages required without experiencing adverse effects.
In conclusion, bupivacaine and ibuprofen intraperitoneal instillation provide better analgesia than bupivacaine and dexmedetomidine (BD Group) or bupivacaine alone (B). This was performed with no apparent side effects. This study supports the growing use of adding ibuprofen to intraperitoneal local anesthetics (IPLA) for pain management postoperatively in laparoscopic gynecological operations.
Authors affiliations:
- Wael Sayed Algharabawy, MD, Assistant Professor of Anesthesia, Intensive Care, & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: gharabawy76@med.asu.edu.eg; {ORCID:0000-0002-8651-4533}
- Khaled Abdou, MD, Lecturer of Anesthesia, Intensive Care, & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: Khaledabdou@med.asu.edu.eg; {ORCID:0000-0001-9777-080X}
- Mariam Kamal, MD, Lecturer of Anesthesia, Intensive Care, & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: drmariam@med.asu.edu.eg; {ORCID:0009-0001-1336-0094}
- Marwa Salem, MD, Lecturer of Anesthesia, Intensive Care, & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; marwamostafa@med.asu.edu.eg; {ORCID:0000-0002-1223-5670}
ABSTRACT
Background & objective: We compared the efficacy of ibuprofen versus dexmedetomidine in combination with bupivacaine or bupivacaine alone in intraperitoneal local anesthetic (IPLA) installation for postoperative pain control after total laparoscopic hysterectomy (TLH).
Methodology: One hundred eighty female patients scheduled for total laparoscopic hysterectomy were randomly assigned to one of three groups to receive intraperitoneal injections of either 50 mL of bupivacaine 0.25% (group B), 50 mL of bupivacaine 0.25% with dexmedetomidine 1 µg/kg (Group BD), or 50 mL of bupivacaine 0.25% with ibuprofen 400 mg (Group BI). In this randomised controlled trial, we evaluated the severity of postoperative pain using a visual analogue scale (VAS). Additionally, hemodynamic changes, total postoperative opioid consumption within the first 24 hours, incidence of postoperative nausea and vomiting, patient satisfaction, sedation score, time to the first request for analgesia, onset of ambulation, and side effects were recorded.
Results: Group BI demonstrated significantly lower pain scores at 6 and 12 hours, both at rest and during movement, compared with Groups BD and B. Group BD also showed lower pain scores than Group B at 6 hours at rest and during movement (P < 0.009 and P < 0.002, respectively). The time to first analgesic request was significantly longer in Group BI than in Groups BD and B (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours, respectively). Postoperative cumulative opioid consumption was lower in Group BI compared with Groups BD and B. Sedation scores were higher in Group BD only at 2 hours postoperatively.
Conclusion: Bupivacaine-ibuprofen was superior to bupivacaine-dexmedetomidine and bupivacaine alone. This was achieved with no significant negative consequences.
Keywords: Laparoscopy; Hysterectomy; Ibuprofen; Dexmedetomidine; Bupivacaine; Pain; Postoperative.
Citation: Algharabawy WS, Abdou K, Kamal M, Salem M. Efficacy of adding dexmedetomidine versus ibuprofen as an adjuvant to intraperitoneal bupivacaine for pain control after laparoscopic gynecological procedures: a double-blind randomized comparative study. Anaesth. pain intensive care 2025;29(8):831-840. DOI: 10.35975/apic.v29i8.3004
Received: February 02, 2025; Revised: September 26, 2025; Accepted: September 28, 2025
1. INTRODUCTION
Laparoscopic surgeries are becoming increasingly popular due to their fast-track nature, which promotes early patient recovery by reducing postoperative somatic and muscular pain components, facilitating early mobilization, rapid oral intake, shorter hospital stays, and greater patient satisfaction.Error! Reference source not found.
Hysterectomy is a common gynecological operation. Traditionally, this has been accomplished through the abdominal or vaginal routes. However, hysterectomies are increasingly being performed using minimal access techniques.Error! Reference source not found.
Postoperative abdominal and shoulder pain are common after total laparoscopic hysterectomy (TLH). Pain is due to a variety of factors, including visceral pain caused by abdominal stretching, surgical manipulations that result in peritoneal and visceral inflammation, and peritoneal irritation caused by pneumoperitoneum. Shoulder tip pain is caused by phrenic nerve irritation caused by CO2 trapped beneath the diaphragm.Error! Reference source not found.
Various analgesic techniques and drugs were used to control postoperative pain after laparoscopic surgeries, which may require systemic opioid analgesia but result in sedation and delayed return of bowel function, resulting in delayed hospital discharge. Other techniques include non-opioid analgesia, regional analgesia, regional block techniques, and intraperitoneal administration of local anesthetics (IPLA).Error! Reference source not found.
It has been observed that postoperative pain after laparoscopic surgery can be decreased by intraperitoneal instillation of local anesthetic agents either alone or in combination with opioids and α2 agonists like clonidine and dexmedetomidine. Bupivacaine is thought to block free afferent nerve endings in the peritoneum, thereby inhibiting visceral nociceptive conduction. Local anesthetics block nociception by affecting nerve membrane-associated proteins and inhibiting the release and action of prostaglandins and other agents that sensitize nociceptors and contribute to inflammation. However, systemic absorption of local anesthetic from the peritoneal cavity may also play a role in reduced nociception; this could be another mechanism of analgesia.Error! Reference source not found.
Dexmedetomidine is a selective alpha-2 (α2) adrenergic agonist with analgesic and sedative properties that can prolong the action of local anesthetics. Dexmedetomidine acts over peritoneal neural receptors and blocks nociceptive stimuli due to its high lipophilic nature.Error! Reference source not found.
Nonsteroidal anti-inflammatory drugs (NSAIDs) have been found to have both central and peripheral effects; however, local application to the site of injury may result in effective concentration in the inflamed tissues, resulting in a decreased fibroproliferative inflammatory response, which results in decreased postoperative adhesions and intestinal distension with minimal adverse effects.Error! Reference source not found.
Although NSAIDs are not a local anesthetic agent, a COX inhibitor reduces the secretion of pain mediators in the areas where surgical manipulation is applied, and it also has a local anti-nociceptive effect in addition to its analgesic effect.Error! Reference source not found.
Ibuprofen, a propionic acid derivative, is a nonsteroidal anti-inflammatory drug (NSAID) with anti-inflammatory, antipyretic, and analgesic properties similar to other agents in its class. Oral ibuprofen has long been recognized as one of the most commonly used and safest NSAIDs.Error! Reference source not found.The US FDA authorized an intravenous (IV) formulation of ibuprofen in 2009 (Caldolor®, Cumberland Pharmaceuticals, Inc., TN, USA) for the management of pain and reduction of fever in adults.Error! Reference source not found.
This study is the first to evaluate the use of intraperitoneal ibuprofen as an adjuvant to bupivacaine, in comparison with dexmedetomidine, and with bupivacaine alone, for postoperative analgesia following total laparoscopic hysterectomy (TLH), with respect to efficacy, side effects, and patient satisfaction.
2. METHODOLOGY
Ethical approval for this study was obtained from the Research Ethics Committee (REC), Faculty of Medicine, FWA 000017585, REC-FMASU@med.asu.edu.eg on May 22, 2023 (FMASU R131/2023). The clinical trial registry (NCT06046105) coded this prospective, double-blind, randomized, controlled study on September 14, 2023. The study was conducted at Ain Shams University Hospitals from October 2023 to June 2024. Written informed consent to participate was obtained from all of the participants in the study. The study enrolled 180 patients who met the inclusion criteria and were scheduled for total laparoscopic hysterectomy (TLH) in the Gynaecology and Obstetrics theatre at Ain Shams University Hospitals. Inclusion criteria included age between 18 and 80 years, female sex, BMI between 18.5 and 35 kg/m2, and ASA physical status I-II. Exclusion criteria included age < 18 or more than 80 years, BMI > 40 or <18.5 kg/m2, ASA III-IV, allergy to one of the agents used, local infection at the port site, coagulopathy, severe cardiac (NYHA III) or pulmonary dysfunction (known COPD, previous thoracic surgeries, or recent pulmonary infection), severe hepatic impairment (Child C) (INR >2, albumin <2.5), severe renal dysfunction (creatinine clearance <30), conversion to open surgery, emergency operations, neurologic, psychiatric or mental illness, or refusal to participate in the study.
Using a computer-generated random number table, the study subjects were randomly assigned to three equal groups. One investigator assessed the patients' eligibility and obtained written informed consent from them during the pre-anesthetic interview the day before surgery. The primary investigator was responsible for opening opaque envelopes with sequential numbers that contained the allocation of groups. The study group assignments were blinded to both the participants and the outcome assessors. Group B (bupivacaine only) (n = 60) was given an intraperitoneal injection of 50 mL of diluted Bupivacaine 0.25 percent (125 mg) in 100 mL of normal saline. Group BD (bupivacaine-dexmedetomidine) (n = 60), assigned for intraperitoneal administration of 50 mL bupivacaine 0.25 percent (125 mg) with dexmedetomidine 1 µg/kg diluted in 100 mL normal saline. Group BI (Bupivacaine-Ibuprofen) (n = 60), assigned for intraperitoneal administration of 50 mL Bupivacaine 0.25% (125 mg) with ibuprofen 400 mg diluted in 100 mL normal saline. Drugs mentioned previously were injected intraperitoneally through trocars at the surgical end.
In this study, a simple spraying cannula attached to a 20-mL syringe was used for intraperitoneal instillation. This method is readily available, time-efficient, and eliminates the need for expensive or complex devices to aerosolize or nebulize the local anesthetic.8 There is no previous evidence of using ibuprofen intraperitoneally in combination with bupivacaine in humans; however, it has been administered intraperitoneally in mice to determine equivalent doses of ibuprofen delivered through drinking water or diet.9 Therefore, a mixture of bupivacaine and ibuprofen was prepared under aseptic conditions using 0.9% NaCl as a diluent, while avoiding alkalinization and carefully inspecting for any precipitation. The solution was used immediately. This approach aligns with standard pharmaceutical compounding practices when formal stability data are unavailable.
All patients underwent a standard preoperative evaluation. Upon arrival in the preoperative holding area, a thorough history was obtained, followed by a complete physical examination, laboratory investigations, and other necessary assessments. During the preoperative visit, all patients were instructed on the use of the visual analogue scale (VAS) for pain assessment at rest and during movement, with scores ranging from 0 (no pain) to 10 (worst imaginable pain).
A crystalloid solution IV drip was started after an 18-gauge IV cannula was inserted. All patients were given IV antibiotics and Granisetron 3 mg. Standard monitoring was used in the operating room, including ECG, non-invasive blood pressure, and pulse oximetry. The following baseline parameters were also recorded: mean blood pressure (MBP), heart rate (HR), and arterial oxygen saturation (SpO2). Following pre-oxygenation, IV 2 mg/kg propofol, atracurium 0.5 mg/kg, and 2 µg/kg fentanyl were used to induce general anesthesia. Following endotracheal intubation, anesthesia was maintained with 1-1.5 percent isoflurane in a FiO2 0.5 oxygen, air mixture to keep the hemodynamics within 20% of basal values. IV boluses of fentanyl (up to 3 µg/kg) were administered, and muscle relaxation was maintained with IV 0.1 mg/kg atracurium boluses every 30 minutes.
To maintain normocarbia, mechanical ventilation parameters were adjusted to keep the end-tidal CO2 (EtCO2) between 30-35 mmHg. Hypotension was defined as having a mean blood pressure (MBP) of less than 60 mmHg, and bradycardia as having a heart rate (HR) of less than 50 beats per minute. IV boluses of 0.1 mg/kg ephedrine were used to treat hypotension, and IV 0.01 mg/kg atropine was used to treat bradycardia. The maximum insufflation pressure of CO2 was 15 mmHg.
The surgeon administered intraperitoneal injections via trocars at the end of the procedure, after which discontinuation of isoflurane was done, and the neuromuscular blockage was antagonised by using neostigmine 0.05 mg/kg and atropine 0.01 mg/kg after assessment of nerve stimulator following train of four, and was administered to reverse atracurium effect. Patients were transferred to the post-anesthetic care unit (PACU) after the surgical procedure was completed and the extubation criteria were met. Standard monitoring was used in the PACU, and readings were documented, and the level of pain was measured. After achieving an Aldrete score = 9, the patient was assessed and discharged. The patient was transferred to the ward for ongoing monitoring and management throughout the study.
To maintain blinding, a separate anesthesiologist who was not involved in the study prepared the study drugs in injectable form. Consequently, both the patients and the anesthesiologist administering the medication were blinded to the group allocation. In addition, the physician responsible for data collection was also unaware of the administered drugs. All patients were given a post-operative pain protocol during the first 24 hours post-surgery (1 gm paracetamol IV every 8 hr). Meperidine (Pethidine) 0.5 mg/kg, which was diluted in 5mL, was titrated intravenously as a rescuing analgesic if VAS was greater than three or when the patient required additional analgesia at any time. Complications such as nausea and vomiting postoperatively, as well as other drug-related complications (e.g., toxicity) were recorded till 24 hours post-surgery. Postoperative abdominal pain intensity was measured using a 10-cm linear visual analogue scale at 2, 4, 6, 12, and 24 h intervals (VAS). The time of the first analgesic request (rescue analgesia) of pethidine, total postoperative opioid consumption, side effects such as nausea, vomiting, sweating, dizziness, tinnitus, muscular twitches, and circumoral numbness, onset of ambulation, and modified RAMSAY sedation score were all recorded at equal time intervals. Patients who were converted to open surgery or in whom drains were placed were excluded from the study.
The primary outcome of this study was the measurement of pain severity on a Numerical Rating Scale (NRS) ranging from 0 to 10. A 10-cm scale was used to assess pain intensity, with 0 representing no pain and 10 representing the most severe pain, measured at 2, 4, 6, 12, and 24 hours postoperatively at rest and during
movement. Secondary outcomes included cumulative postoperative opioid use in the first 24 hours postoperatively, the initial analgesic request, postoperative nausea and vomiting, the incidence of complications (e.g., LA toxicity), patient satisfaction after the first postoperative day (1 = excellent, 2 = good, 3 = fair, and 4 = poor), patients' hemodynamics, and the onset of ambulation.
2.1. Statistical analysis
A sample size of 58 patients per group was calculated to detect an effect size of 0.8 for VAS scores between the study groups, with a two-sided α error of 0.01 and a statistical power of 0.95. Statistical Package for the Social Sciences (SPSS) version 27 was used to analyze the data. Quantitative data were represented as mean, standard deviation (SD), or median (IQR) when indicated. Percentage and frequency were used to express qualitative data. The Chi-square (X2) test of significance was employed to compare proportions between two qualitative measures. The post-hoc test was used for pairwise comparison of subgroups when the ANOVA test was positive. One-way analysis of variance (ANOVA) was used to evaluate differences within the means of various subgroups of a variable (multiple testing). The Kruskal–Wallis test was used to compare multiple subgroups for non-parametric data. When a significant difference was detected, pairwise comparisons were performed. The confidence interval was set at 95%, and the margin of error was set at 5%. Accordingly, P-values less than 0.05 were considered statistically significant.
3. RESULTS
A total of two hundred patients were enrolled in the trial; 11 patients refused to participate, therefore a total of 189 patients were randomly assigned to one of the three groups (B-BD-BI). Three patients from each group did not participate in the study due to either the insertion of drains or conversion into open surgery. The study adheres to CONSORT guidelines. Each group included 60 patients who were analyzed in this study. Age, body mass index (BMI), duration of surgery, and ASA physical status (I–II) were comparable among the three groups, with no significant differences. Heart rate and mean arterial pressure were also comparable across all groups and remained within normal ranges (Table 1). Total postoperative opioid consumption was lower in Group BI (69.17 ± 32.0 mg) than in Groups BD (88.33 ± 39.5 mg) and B (93.33 ± 40.6 mg), and this difference was statistically significant (P = 0.001) (Table 1). The time to the first request for analgesia was significantly longer in the BI group compared with the BD and B groups (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours, respectively; P < 0.001). Although the duration was longer in the BD group than in the B group, the difference was not statistically significant (Table 1).
| Table 1: demographic data, hemodynamic parameters, and rescue analgesia | |||||||
| Demographic data | Group B
(n = 60) |
Group BD
(n = 60) |
Group BI
(n = 60) |
Test | P-value | ||
| Age (years) | 52.30 ± 6.9 | 50.62 ± 6.9 | 52.20 ± 6.6 | 1.17f | 0.314 | ||
| BMI (kg) | 33.98 ± 3.6 | 34.37 ± 3.3 | 33.53 ± 3.2 | 0.93 f | 0.398 | ||
| Surgical time (min) | 142.15 ± 26.8 | 133.37 ± 27.1 | 134.20 ± 24.2 | 2.07 f | 0.129 | ||
| ASA | I | 31 (51.7) | 30 (50) | 28 (46.7) | 0.31X2 | 0.856 | |
| II | 29 (48.3) | 23 (50) | 32 (53.3) | ||||
| Mean Arterial Pressure (mmHg) | 2 hr | 87.27 ± 8.2 | 84.35 ± 9.4 | 85.90 ± 9.3 | 1.58 f | 0.209 | |
| 4 hr | 90.03 ± 8.4 | 88.42 ± 7.1 | 87.60 ± 9.5 | 1.30 f | 0.275 | ||
| 6 hr | 87.82 ± 10.0 | 87.67 ± 9.2 | 86.73 ± 9.0 | 0.24 f | 0.791 | ||
| 12 hr | 83.47 ± 10.1 | 82.32 ± 10.3 | 83.50 ± 9.7 | 0.27 f | 0.764 | ||
| 24 hr | 85.35 ± 10.2 | 85.75 ± 8.7 | 85.63 ± 10.3 | 0.03 f | 0.974 | ||
| Heart rate (beat/min) | 2 hr | 86.03 ± 7.9 | 85.77 ± 8.5 | 85.47 ± 7.5 | 0.08 f | 0.927 | |
| 4 hr | 80.93 ± 14.5 | 83.10 ± 14.0 | 79.32 ± 15.3 | 1.01 f | 0.366 | ||
| 6 hr | 82.75 ± 8.7 | 85.18 ± 9.5 | 82.72 ± 10.4 | 1.30 f | 0.274 | ||
| 12 hr | 86.67 ± 9.8 | 88.48 ± 9.4 | 86.32 ± 9.9 | 0.86 f | 0.424 | ||
| 24 hr | 84.72 ± 10.4 | 84.33 ± 9.2 | 83.60 ± 9.0 | 0.21 f | 0.809 | ||
| Postop opioid consumption (mg) | 93.33 ± 40.6 a | 88.33 ± 39.5 a | 69.17 ± 32.0 b | 6.92 f | 0.001* | ||
| 1st rescue analgesia (hr) | 6.73 ± 2.6 a | 6.80 ± 2.9 a | 8.50 ± 2.3 b | 9.04 f | <0.001* | ||
| Onset of ambulation (min) | 146.25 ± 19.7 | 147.13 ± 17.4 | 144.08 ± 20.5 | 0.40 f | 0.672 | ||
| Satisfaction | 2 | 28 (46.7) | 31 (51.7) | 34 (56.7) | 1.2 x2 | 0.548 | |
| 3 | 32 (53.3) | 29 (48.3) | 26 (43.3) | ||||
| PONV | 20 (33.3) | 23 (38.3) | 18 (30) | 0.94 f | 0.624 | ||
| Data is expressed as mean ± SD or proportion, f = ANOVA test, X2 = chi square test, different letters indicate significant difference in post hoc (Tukey) test | |||||||
There was no significant difference between the three groups in terms of ambulation onset (146.25 ± 19.7, 147.13 ± 17.4, 144.08 ± 20.5 minutes), respectively. Using a four-point rating system (1 = excellent, 2 = good, 3 = fair, and 4 = poor), patient satisfaction was equivalent across the three groups, and there was no significant difference (P < 0.548) (Table 1). The incidence of postoperative nausea and vomiting was comparable in all three groups till the end of the study and was statistically insignificant. Sedation was evaluated using the Modified Ramsay sedation score. Sedation was higher in the BD group only at 2 hours postoperative measurement and was statistically significant (P ˂ 0.018) compared to the other two groups. However, the sedation score was never greater than three, indicating that all patients were arousable and did not require any intervention (Table 2). Postoperative VAS at rest after 6 hours was considerably lower in Group BI compared to Groups B and BD. It was also significantly lower in Group BD compared to Group B. At 12 hours, the VAS score at rest in the BI group was significantly lower than in the other two groups, with P-values of 0.001 and 0.031, respectively. However, there was no significant difference between groups B and BD. During movement, the VAS score at 6 hours was significantly lower in the BI group compared with the other two groups, and it was also lower in the BD group compared with the B group. The VAS score in the BI group remained significantly lower than in the other two groups (B and BD), which were comparable (Table 2).
| Table 2: Comparison between the three groups as regards VAS and RSS. | ||||||||||
| Parameter | Group B (n = 60) | Group BD (n = 60) | Group BI (n = 60) | |||||||
| range | Median | IQR | Range | Median | IQR | Range | Median | IQR | P | |
| VAS rest | ||||||||||
| 2 | 1-3 | 2 | 1-2 | 1-3 | 2 | 1-2 | 0-3 | 2 | 1-2 | 0.496 |
| 4 | 1-4 | 2 | 2-3 | 1-4 | 2 | 1.5-3 | 0-4 | 2 | 2-2 | 0.268 |
| 6 | 2-5 | 4 a | 3-4 | 2-4 | 3 b | 3-4 | 1-5 | 3 c | 2-3.5 | < 0.001* |
| 12 | 2-4 | 3 a | 3-4 | 2-4 | 3 a | 3-4 | 1-4 | 3 b | 2-4 | 0.031* |
| 24 | 1-4 | 2 | 1-3 | 1-4 | 2 | 2-3 | 1-4 | 2 | 1-3 | 0.392 |
| VAS movement | ||||||||||
| 2 | 1-3 | 2 | 1-3 | 1-3 | 2 | 1-3 | 0-3 | 2 | 1-2 | 0.233 |
| 4 | 1-4 | 2 | 2-3 | 1-4 | 2 | 1.5-3 | 0-4 | 2 | 2-2 | 0.269 |
| 6 | 3-5 | 4 a | 3-4 | 3-5 | 3 b | 3-4 | 1-5 | 3 c | 2-4 | < 0.001* |
| 12 | 2-5 | 4 a | 3-4 | 2-4 | 3 a | 3-4 | 2-4 | 3 b | 2-4 | 0.008* |
| 24 | 1-4 | 2 | 1-4 | 1-4 | 3 | 2-3 | 1-4 | 2 | 1-3 | 0.618 |
| RSS | ||||||||||
| 2 | 1-2 | 1 a | 1-1 | 1-2 | 1 b | 1-2 | 1-2 | 1 a | 1-1 | 0.018* |
| 4 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1 |
| 6 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1 |
| 12 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1 |
| 24 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1-1 | 1 | 1-1 | 1 |
| Data expressed as median and IQR, P-value done by Kruskal Wallis test, different letters indicate significant difference in pairwise comparison between groups. | ||||||||||
4. DISCUSSION
Postoperative pain management following laparoscopic gynecological surgeries is a significant concern. Postoperative discomfort arises from distention of the abdominal wall and peritoneum, irritation of the diaphragm and peritoneum due to residual gas, and visceral pain resulting from surgical manipulation.Error! Reference source not found. Several methods have been employed to reduce postoperative discomfort after laparoscopic procedures, including intraperitoneal closed suction drains, intraperitoneal injection of local anesthetics (IPLA), transversus abdominis nerve block, epidural analgesia, and other regional analgesia and block techniques. The intraperitoneal route of administration of local anesthetics (LA) is simple, safe, and promotes ambulatory anesthesia.Error! Reference source not found.
Adjuvants added to IPLA may increase effectiveness, lengthen duration, improve outcome, and give greater analgesia.Error! Reference source not found.Many medications are utilized as adjuvants to IPLA, such as dexmedetomidine, opioids like tramadolError! Reference source not found.or fentanylError! Reference source not found. or NSAIDS like tenoxicamError! Reference source not found.or ketorolac.Error! Reference source not found.
Dexmedetomidine, an α 2-adrenergic agonist, has been introduced into clinical anesthesia as a sedative, sympatholytic, hemodynamic-stabilizing, and anesthetic-sparing agent. Its effects are centrally mediated via α2-adrenoceptors, which are found in the spinal cord's dorsal horn and locus coeruleus.Error! Reference source not found. The side effects of dexmedetomidine include bradycardia without respiratory depression, nausea, hypertension, and hypotension.Error! Reference source not found.
Ibuprofen is one of the most often used analgesic-antipyretic-anti-inflammatory medications. Like many traditional NSAIDs, ibuprofen inhibits both the constitutive cyclo-oxygenase-1 (COX-1), which is responsible for the synthesis of prostanoids (PGs, and thromboxane-A2) that control a range of physiological functions (vascular, blood flow, gastric and renal functions), and the inducible COX-2, whose synthesis is increased, leading to augmented production of PGE2 in inflammation and pain. Error! Reference source not found.
The intravenous formula of ibuprofen is a racemic combination of [-] R- and [+] S-isomers, with the [+] S-isomer responsible for the drug's efficacy and the [-] R-isomer acting as an inactive reservoir that is actively converted to the active [+] S-isomer. Intravenous ibuprofen has a very varied volume of distribution based on the patient's age and health. It is almost 99% plasma proteins bound at 20 µg/mL. Error! Reference source not found.
Significant pharmacological characteristics of racemic ibuprofen, which has a 50:50 mixture of the two isomers, are derived from the stereochemical configuration of R (-)- and S (+)-ibuprofen. When compared to the active S (+) enantiomer, R (-)-ibuprofen is a weaker inhibitor of COX-1 and COX-2, which has an impact on prostaglandin synthesis and thromboxane A2.24 R (-)-ibuprofen, on the other hand, is an effective inhibitor of leukotrienes and other products produced by activated polymorphonuclear leukocytes. R (-)-ibuprofen also suppresses the formation of anandamide, an endogenous cannabinoid that causes analgesia in the central nervous system. Moreover, R (-)-ibuprofen reduces mucosal irritation from COX-1 in the stomach by competing with S (+)-ibuprofen for the binding of this enzyme. However, upon absorption from the upper GI tract, 40–60% of R (-)-ibuprofen is changed to the S (+) isomer, resulting in a "ballast" of the latter's reserves in the body. Error! Reference source not found.
Like some other NSAIDs, ibuprofen acts as an analgesic through both PG-dependent Error! Reference source not found. and PG-independent Error! Reference source not found. pathways. Prostaglandin E2 (PGE2), prostaglandin I2 (PGI2), and prostaglandin D2 (PGD2) interact with their corresponding EP, IP, or DP receptors on nerves to activate certain channels or enzymes that control the neuronal transmission of pain signals. These interactions are the basis for prostaglandin-dependent mechanisms. Error! Reference source not found. The N-type CaV 2.2 channels, glutamate, N-methyl-D-aspartate (NMDA) receptors, neurokinin (NK1) receptors, gamma-aminobutyric acid (GABAA and GABAB) receptors, glycine receptors, and adenosine P2X3 receptors are non-PG central (dorsal horn) targets involved in pain modulation. Error! Reference source not found. Ibuprofen reduces neuronal hyperexcitability by blocking alterations in the NaV 1.7 and 1.8 channels. Error! Reference source not found.
This study was the first to evaluate the use of intraperitoneal ibuprofen as an adjuvant to bupivacaine, in comparison with dexmedetomidine, and with bupivacaine alone, for postoperative analgesia following total laparoscopic hysterectomy (TLH), with respect to efficacy, side effects, and patient satisfaction. We found that administering Ibuprofen-bupivacaine (Group BI) intraperitoneally reduced postoperative pain scores significantly, decreased postoperative cumulative opioid consumption, and extended the time to first request analgesia when compared to administering bupivacaine-dexmedetomidine (Group BD) or Bupivacaine alone (Group B).
The postoperative VAS was statistically significant (P < 0.001 and 0.031) after 6 and 12 hours at rest, and (P < 0.001 and 0.008) at 6 and 12 hours on movement, respectively (Table 2), favoring better pain management in the Ibuprofen-bupivacaine (BI group). After 24 hours, the bupivacaine-ibuprofen group consumed fewer opioids (69.17 ± 32.0 mg) than the bupivacaine-dexmedetomidine group (88.33 ± 39.5 mg) or bupivacaine alone (93.33 ± 40.6 mg), with a statistically significant difference (P = 0.001). This might be attributed to ibuprofen's extended half-life. Error! Reference source not found. Thus, emphasizing the idea that combined therapy may produce much better analgesia. Furthermore, there was a statistically significant (P < 0.001) difference in the duration of time until the first request for analgesia (8.50 ± 2.3, 6.80 ± 2.9, and 6.73 ± 2.6 hours) between the BI group and the BD and B alone groups. This might be due to the anti-inflammatory and analgesic qualities of ibuprofen; also, local use of NSAIDs may result in effective and fast concentration levels in inflamed tissues with less systemic adverse effects. Error! Reference source not found. A lower VAS score and lower total opioid use in group BI resulted in fewer opioid-related adverse effects, such as dizziness, drowsiness, and delayed bowel movement. Gupta et al. Error! Reference source not found. found that administering 1 µq/kg dexmedetomidine with 125 mg bupivacaine intraperitoneally during laparoscopic gynecological surgeries reduced VAS, postoperative analgesic requirements, and increased time to first request postoperative morphine. The findings of this study support it.
Numerous studies indicate that NSAIDs and bupivacaine together provide sufficient analgesia and opioid-sparing effectiveness during intraperitoneal instillation for various surgical procedures. El Hakim et al. revealed the excellent intraperitoneal instillation efficacy of lidocaine and tenoxicam that lowered discomfort with movement over 24 hours following laparoscopic cholecystectomy compared to the control group. Patients in the tenoxicam intraperitoneal group required less nalbuphine than the control group, demonstrating that intraperitoneal combination therapy produces superior analgesia. Error! Reference source not found. The bupivacaine-dexmedetomidine (BD) group had significantly higher sedation scores than the other two groups in the first two hours (P = 0.018). This could be attributed to its α2 adrenergic agonist properties, which cause sympatholysis, anxiolysis, and sedation while maintaining respiratory functions. The sedation scores were similar between bupivacaine-ibuprofen (Group BI) and bupivacaine alone (Group B).
The results were consistent with other results done by Shukla et al. 15 who examined the effects of intraperitoneal bupivacaine alone (B), in combination with either dexmedetomidine (BD) or tramadol (BT), during laparoscopic cholecystectomy. Compared to bupivacaine 0.25% alone, intraperitoneal instillation of dexmedetomidine 1 μ/kg in conjunction with bupivacaine 0.25% significantly reduces post-operative pain and the need for analgesics during the post-operative period; this combination may even prevail over bupivacaine combined with tramadol. Bakhamees et al. Error! Reference source not found. also investigated the intraoperative dexmedetomidine infusion effect compared to placebo on the total amount of intraoperative fentanyl and propofol required to maintain anesthesia, the recovery profile, the pain score, and the total amount of morphine used via patient-controlled analgesia. They found that the total amounts of propofol and fentanyl required to maintain anesthesia were reduced, intraoperative and postoperative hemodynamic stability was improved, postoperative pain was lower, and the total morphine consumption was decreased.
Moreover, recovery was better compared to the placebo group. The incidence of postoperative nausea and vomiting did not differ significantly among the three groups (P = 0.624), consistent with earlier studies evaluating postoperative nausea and vomiting following intraperitoneal administration of bupivacaine–dexmedetomidine32 or bupivacaine–tenoxicam.17 No signs of local anesthetic systemic toxicity were observed during the study.

Figure 1: Box and whisker graph between the groups as regards VAS at rest

Figure 2: Box and whisker graph between the groups regarding VAS at movement
5. LIMITATIONS
The study's limitations include the fact that the visual analogue score is not an objective indicator, but it is clear and reliable. Additionally, the study was conducted in a single center, comparing the three groups of pain medications, including bupivacaine alone, bupivacaine with dexmedetomidine, and ibuprofen. To the best of our knowledge, our study is the first to evaluate the effects of combining ibuprofen with bupivacaine for intraperitoneal instillation to control postoperative pain and enhance recovery criteria. Thus, more analytical studies involving a larger number of patients, as well as the establishment of large multi-center databases, are required to evaluate efficacy and modify the effective dosages required without experiencing adverse effects.
6. CONCLUSION
In conclusion, bupivacaine and ibuprofen intraperitoneal instillation provide better analgesia than bupivacaine and dexmedetomidine (BD Group) or bupivacaine alone (B). This was performed with no apparent side effects. This study supports the growing use of adding ibuprofen to intraperitoneal local anesthetics (IPLA) for pain management postoperatively in laparoscopic gynecological operations.
- Availability of Data
- Acknowledgements
- Ethics approval, and consent to participate
- Competing interests
- Authors’ contributions
12. REFERENCES
- Michaloliakou C, Chung F, Sharma S. Preoperative multimodal analgesia facilitates recovery after ambulatory laparoscopic cholecystectomy. Anesth Analg. 1996 Jan;82(1):44-51. DOI: 1097/00000539-199601000-00009. PMID: 8712425.
- Walsh CA, Walsh SR, Tang TY, Slack M. Total abdominal hysterectomy versus total laparoscopic hysterectomy for benign disease: a meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2009 May;144(1):3-7. DOI: 1016/j.ejogrb.2009.01.003. Epub 2009 Mar 25. PMID: 19324491.
- Alexander JI. Pain after laparoscopy. Br J Anaesth. 1997 Sep;79(3):369-78. DOI: 1093/bja/79.3.369. PMID: 9389858.
- Goldstein A, Grimault P, Henique A, Keller M, Fortin A, Darai E. Preventing postoperative pain by local anesthetic instillation after laparoscopic gynecologic surgery: a placebo-controlled comparison of bupivacaine and ropivacaine. Anesth Analg. 2000 Aug;91(2):403-7. DOI: 1097/00000539-200008000-00032. PMID: 10910857.
- Arain SR, Ruehlow RM, Uhrich TD, Ebert TJ. The efficacy of dexmedetomidine versus morphine for postoperative analgesia after major inpatient surgery. Anesth Analg. 2004 Jan;98(1):153-158. DOI: 1213/01.ANE.0000093225.39866.75. PMID: 14693611.
- Rosenblum M, Weller RS, Conard PL, Falvey EA, Gross JB. Ibuprofen provides longer lasting analgesia than fentanyl after laparoscopic surgery. Anesth Analg. 1991 Sep;73(3):255-9. DOI: 1213/00000539-199109000-00004. PMID: 1831017.
- Bookstaver PB, Miller AD, Rudisill CN, Norris LB. Intravenous ibuprofen: the first injectable product for the treatment of pain and fever. J Pain Res. 2010 May 25; 3:67-79. DOI: 2147/jpr. s6993. PMID: 21197311; PMCID: PMC3004645.
- Dubey N, Bellamy F, Bhat S., MacFacter W, & Rossaak, J. (2024). The impact of timing, type, and method of instillation of intraperitoneal local anaesthetic in laparoscopic abdominal surgery: a systematic review and network meta-analysis. British Journal of Anaesthesia, 132(3), 562–574. https://doi.org/10.1016/J.BJA.2023.11.046
- Salama RAM, El Gayar NH, Georgy SS, Hamza M. Equivalent intraperitoneal doses of ibuprofen supplemented in drinking water or in diet: a behavioral and biochemical assay using antinociceptive and thromboxane inhibitory dose-response curves in mice. Peerj. 2016;4: e2239. DOI: 7717/peerj.2239. PMID: 27547547; PMCID: PMC4958011.
- Golubović S, Golubović V, Cindrić-Stancin M, Tokmadzić VS. Intraperitoneal analgesia for laparoscopic cholecystectomy: bupivacaine versus bupivacaine with tramadol. Coll Antropol. 2009 Mar;33(1):299-302. PMID: 19408641.
- Hartley JE, Monson JR. The role of laparoscopy in the multimodality treatment of colorectal cancer. Surg Clin North Am. 2002 Oct;82(5):1019-33. DOI: 1016/s0039-6109(02)00039-7. PMID: 12507207.
- Benincá Albuquerque, A., Carlesso de Freitas, C., Corrêa Bitencourt, I., Alves Pereira Chiuchetta, I., Baú, P. C., & Vontobel Padoin, A. (2025). Postoperative analgesia with instillation of intraperitoneal ropivacaine in patients undergoing laparoscopic cholecystectomies. Journal of Gastrointestinal Surgery, 29(6), 102034. https://doi.org/10.1016/J.GASSUR.2025.102034
- Rahman, A., Ummer, S., S, K., & K, C. (2025). Comparison of post operative analgesia following intraperitoneal instillation of ropivacaine with or without dexamethasone for patients undergoing laparoscopic surgeries. Perioperative Care and Operating Room Management, 38, 100475. https://doi.org/10.1016/J.PCORM.2025.100475
- Marks JL, Ata B, Tulandi T. Systematic review and metaanalysis of intraperitoneal instillation of local anesthetics for reduction of pain after gynecologic laparoscopy. J Minim Invasive Gynecol. 2012 Sep-Oct;19(5):545-53. DOI: 1016/j.jmig.2012.04.002. Epub 2012 Jul 3. PMID: 22763313.
- Shukla U, Prabhakar T, Malhotra K, Srivastava D, Malhotra K. Intraperitoneal bupivacaine alone or with dexmedetomidine or tramadol for post-operative analgesia following laparoscopic cholecystectomy: A comparative evaluation. Indian J Anaesth. 2015 Apr;59(4):234-9. DOI: 4103/0019-5049.155001. PMID: 25937650; PMCID: PMC4408652.
- Praveena BL, Bharathi B, Sahana VR. Intraperitoneal Ropivacaine with Dexmedetomidine or Fentanyl for Postoperative Analgesia Following Laparoscopic Cholecystectomy: A Comparative Randomized Trial. Anesth Essays Res. 2019 Jan-Mar;13(1):169-173. DOI: 4103/aer.AER_191_18. PMID: 31031500; PMCID: PMC6444959.
- Elhakim M, Amine H, Kamel S, Saad F. Effects of intraperitoneal lidocaine combined with intravenous or intraperitoneal tenoxicam on pain relief and bowel recovery after laparoscopic cholecystectomy. Acta Anaesthesiol Scand. 2000 Sep;44(8):929-33. DOI: 1034/j.1399-6576.2000.440806. x. PMID: 10981568.
- Murdoch J, Ramsey G, Day AG, McMullen M, Orr E, Phelan R, Jalink D. Intraperitoneal ketorolac for post-cholecystectomy pain: a double-blind randomized-controlled trial. Can J Anaesth. 2016 Jun;63(6):701-8. English. DOI: 1007/s12630-016-0611-4. Epub 2016 Feb 10. PMID: 26864193.
- Kamibayashi T, Maze M, Weiskopf RB, Weiskopf RB, Todd MM. Clinical uses of α2-adrenergic agonists. Anesthesiology. 2000 Nov 1;93(5):1345–9. DOI:1097/00000542-200011000-00030
- Chad M. Brummett, Elizabeth K. Hong, Allison M. Janda, Francesco S. Amodeo, Ralph Lydic; Perineural Dexmedetomidine Added to Ropivacaine for Sciatic Nerve Block in Rats Prolongs the Duration of Analgesia by Blocking the Hyperpolarization-activated Cation Current. Anesthesiology 2011; 115:836–843 DOI: https://doi.org/10.1097/ALN.0b013e318221fcc9
- Aho M, Erkola O, Kallio A, Scheinin H, Korttila K. Comparison of dexmedetomidine and midazolam sedation and antagonism of dexmedetomidine with atipamezole. J Clin Anesth. 1993 May-Jun;5(3):194-203. DOI: 1016/0952-8180(93)90014-6. PMID: 8100428.
- Cryer B, Feldman M. Cyclooxygenase-1 and cyclooxygenase-2 selectivity of widely used nonsteroidal anti-inflammatory drugs. Am J Med. 1998 May;104(5):413-21. DOI: 1016/s0002-9343(98)00091-6. PMID: 9626023.
- Davies NM. Clinical pharmacokinetics of ibuprofen. The first 30 years. Clin Pharmacokinet. 1998 Feb;34(2):101-54. DOI: 10.2165/00003088-199834020-00002. PMID: 9515184.
- Rainsford KD. Ibuprofen: pharmacology, efficacy and safety. Inflammopharmacology. 2009 Dec;17(6):275-342. DOI: 10.1007/s10787-009-0016-x. Epub 2009 Nov 21. PMID: 19949916.
- Vanegas H, Schaible H. Prostaglandins and cycloxygenases in the spinal cord. Prog Neurobiol. 2001;64(4):327-363. DOI:1016/S0301-0082(00)00063-0
- Rainsford, K.D. (2012). Mechanisms of Inflammation and Sites of Action of NSAIDs. In: Ibuprofen: Pharmacology, Therapeutics and Side Effects. Springer, Basel. https://doi.org/10.1007/978-3-0348-0496-7_3
- Gould HJ 3rd, England JD, Soignier RD, Nolan P, Minor LD, Liu ZP, et al. Ibuprofen blocks changes in Na v 1.7 and 1.8 sodium channels associated with complete Freund's adjuvant-induced inflammation in rat. J Pain. 2004;5(5):270-280. DOI: 1016/j.jpain.2004.04.005
- Mattheolabakis G, Mackenzie GG, Huang L, Ouyang N, Cheng KW, Rigas B. Topically applied phospho-sulindac hydrogel is efficacious and safe in the treatment of experimental arthritis in rats. Pharm Res. 2013;30(6):1471. DOI:1007/s11095-012-0953-8
- McCleane G. Topical application of analgesics: a clinical option in day case anaesthesia? Curr Opin Anaesthesiol. 2010;23(6):704-707. DOI:1097/ACO.0b013e328338469b
- Rømsing J, Mysager S, Vilmann P. Postoperative analgesia is not different after local vs systemic administration of meloxicam in patients undergoing inguinal hernia repair. Can J Anaesth. 2001; 48:978-984. DOI:1007/BF03016587
- Mulshine JL, Atkinson JC, Greer RO, Papadimitrakopoulou VA, Van Waes C, Rudy S, et al. Randomized, double-blind, placebo-controlled phase IIb trial of the cyclooxygenase inhibitor ketorolac as an oral rinse in oropharyngeal leukoplakia. Clin Cancer Res. 2004;10(5):1565-1573. DOI: 1158/1078-0432.ccr-1020-3
- Gupta B, Verma V, Chaudhary UK, Sidhu R, Chandel A. Effect of intraperitoneal instillation of dexmedetomidine or fentanyl as adjuvants to bupivacaine on fast tracking discharge criteria in patients undergoing ambulatory laparoscopic cholecystectomy: A randomized double-blind control trial. Ain-Shams J Anesthesiol. 2021;13(1):1-7. DOI:1186/s42077-021-00175-1
- Bakhamees HS, El-Halafawy YM, El-Kerdawy HM, Gouda NM, Altemyatt S. Effects of dexmedetomidine in morbidly obese patients undergoing laparoscopic (1)gastric bypass. Middle East J Anaesthesiol. 2007;19(3):537-551.