Mujeeb Ur Rehman Parrey 1, Muhammad Omer Afzal Bhatti 2, Maha M Abdul-Latif 3, Saima Rehman 4, Mohammed M. Ismail 5, Omaima A Hamid 6
Authors affiliations:
Artificial intelligence (AI) is revolutionizing retinal diagnostics by enhancing the accuracy and efficiency of disease detection in fundus photography. Manual interpretation is time-consuming and subject to interobserver variability, highlighting the need for AI-driven solutions. This study assesses the diagnostic performance of AI algorithms in detecting retinal diseases using fundus photography. A systematic search was conducted in PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus, including studies reporting AI-based retinal disease detection with sensitivity, specificity, and area under the curve (AUC) metrics. The QUADAS-2 tool was used for quality assessment, and a random-effects model was applied for meta-analysis. AI algorithms demonstrated high diagnostic accuracy, with an overall sensitivity of 89% and specificity of 92%. Performance improved with larger datasets (>500 samples), achieving a sensitivity of 92%, specificity of 95%, and an AUC of 0.96. These findings suggest that AI has strong potential for clinical implementation in retinal disease screening, offering high accuracy for early diagnosis and improved patient outcomes.
Abbreviations: AI: Artificial intelligence, AUC: area under the curve,
Keywords: Artificial Intelligence; Algorithms; Deep Learning; Meta-Analysis; Fundus Photography; Retinal Disease Detection; Diagnostic Accuracy
Citation: Parrey MUR, Bhatti MOA, Abdul-Latif MM, Rehman S, Ismail MM, Hamid OA. Meta-analysis of AI algorithm performance in detecting retinal diseases. Anaesth. pain intensive care 2025;29(4):400-406. DOI: 10.35975/apic.v29i4.2828
Received: February 17, 2025; Revised: April 25, 2025; Accepted: April 25, 2025
Artificial intelligence (AI) has emerged as a transformative force in medical diagnostics, revolutionizing clinical support systems across various specialties.1-5 In ophthalmology, AI applications have gained significant attention, particularly for detecting complex and historically challenging ocular conditions.6-9 Deep learning models have demonstrated high sensitivity and specificity in identifying retinal abnormalities by analysing large datasets, enabling them to recognize intricate disease-related features.10
By integrating AI into ophthalmic diagnostics, clinicians can expedite disease detection, minimize human error, and enhance accessibility to eye care, especially in regions with limited ophthalmologist availability.11 Retinal diseases are among the leading causes of visual impairment and blindness worldwide.12-13 Early detection and timely intervention are crucial to preventing irreversible vision loss.14 Fundus photography, a non-invasive imaging technique, plays a vital role in screening and diagnosing retinal pathologies by capturing high-resolution retinal images.15 However, manual interpretation of these images is time-consuming, subjective, and prone to interobserver variability, leading to diagnostic inconsistencies.16 The increasing prevalence of retinal diseases necessitates the development of more efficient and accurate diagnostic methods.17
Several studies have reported promising results regarding AI’s diagnostic accuracy, demonstrating high sensitivity, specificity, and area under the curve (AUC) values.18-19 However, variations in algorithm performance persist due to differences in model architecture, training datasets, and validation methodologies. Additionally, the generalizability of AI models across diverse populations remains a subject of ongoing investigation.20 A comprehensive synthesis of existing evidence is essential to assess the robustness and clinical applicability of AI in fundus photography-based diagnostics.21
This study was designed to evaluate the diagnostic performance of AI algorithms in retinal disease detection using fundus photography. By aggregating sensitivity, specificity, and AUC metrics from multiple studies, this meta-analysis provides a quantitative assessment of AI’s effectiveness. Furthermore, subgroup analyses will examine the impact of disease type, AI model, and dataset characteristics on diagnostic outcomes. The findings from the current study offer valuable insights into the strengths and limitations of AI-driven retinal disease detection, informing future research and potential clinical implementation.
The objective of this study was to evaluate the performance of AI algorithms in detecting retinal diseases using fundus photography.
A comprehensive and detailed search strategy was used to look for pertinent studies assessing the effectiveness of AI algorithms for retinal disease detection using fundus photography in several electronic databases, including PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus. "AI algorithms," "retinal disease," "fundus photography," and "diagnostic performance," among other Medical Subject Headings (MeSH) terms, were utilized in conjunction with keywords to search as shown in Table 1.
To guarantee that the most recent material was included, only English-language publications were taken into consideration, and there were no date limits. To find further research that satisfied the inclusion requirements, references from pertinent publications were manually examined. The included studies comprised one utilizing fundus photography to assess the diagnostic potential of AI algorithms in detecting retinal disorders such as diabetic retinopathy, age-related macular degeneration, and glaucoma; another presenting quantitative diagnostic parameter like AUC, sensitivity, and specificity; and a peer-reviewed study. Studies were excluded if they did not apply AI models to diagnose retinal diseases in fundus images or lacked sufficient data on diagnostic measures.
Additionally, case reports, abstracts, and studies with a sample size of fewer than thirty were disqualified. The quality of the studies included was evaluated using the QUADAS-2 approach. So, four main domains are evaluated for the possibility of bias such as: time, standard reference and selection of patient. To ensure the validity and reliability of the results, studies whose findings would be highly susceptible to bias were eliminated. Each included study's data was extracted by two independent reviewers. The data that was extracted comprised diagnostic measures like sensitivity, specificity, and AUC; dataset features like training dataset size; and study characteristics like study design, sample size, AI model utilized, and disease type.
Additionally, a random effects model was used to pool diagnostic parameters such as sensitivity, specificity, and AUC. The choice to pool diagnostics using this method was made considering the expected heterogeneity of studies, the algorithms used by AI in their research, and the disorders taken into consideration. AUCs were used to summarize the overall measurement accuracy of diagnostic detection, while sensitivity and specificity were combined for estimations of summary along with the relevant 95% CIs. The included studies' heterogeneity was evaluated using the I2 statistic. If a significant heterogeneity (I2 > 50%) was found, the variation was evaluated using subgroup analyses of the source. To find out how important aspects affect AI performance, a subgroup analysis was done. These factors included the type of disease of retina, the particular AI model used, such as deep learning models and convolutional neural networks; and the dataset's attributes, such as its size, population characteristics, and validation process.
The purpose of the analysis was to detect trends in the way AI algorithms performed and to evaluate how reliable the results were under various circumstances. A sensitivity analysis is carried out for specific studies to investigate the impact on the outcome. Studies with a high risk of bias or outliers are subjected to separate analyses to determine whether removing them significantly changes the pooled estimate. R software (version 4.0.3) and Review Manager (RevMan) version 5.4 were used for all statistical analyses. The pooled estimates of sensitivity, specificity, and AUC were displayed graphically using forest plots. Ethical approval was not required because this was a meta-analysis based on an analysis of published data.
The results of the study revealed that AI algorithms for fundus photography-based retinal disease identification perform well overall. The algorithms' 89% sensitivity means they accurately identify 89% of genuine positives, but their 92% specificity means they can correctly identify 92% of true negatives, which means there are fewer false positives. The area under the curve of 0.95 is another indication of these AI models' strong discriminative ability as shown in Table 2.
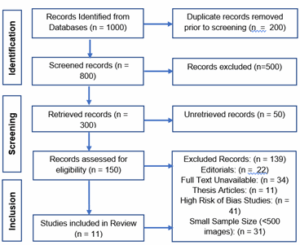
Figure 1: PRISMA flow diagram for meta-analysis of AI algorithms in retinal disease detection using fundus photography
As shown in Table 2 subgroup analyses, however, demonstrated the highest test performance in individuals with diabetic retinopathy who were found in the study. They demonstrated an excellent AUC of 0.96, a relatively high sensitivity of 90%, and a good specificity of 94%. Compared to other models, deep learning models performed better, with the best sensitivity (91%), specificity (93%), and AUC (0.97%). Convolutional neural networks and other factors caused other AI models to perform comparatively worse. AI algorithms also performed better on larger datasets; sensitivity (92%), specificity (95%), and AUC (0.96) were greater in datasets with over 500 samples than in datasets with fewer samples. These findings generally show that AI algorithms, particularly those that use deep learning approaches, perform well in identifying retinal illnesses, especially when working with larger datasets.
The results of this meta-analysis have increased the likelihood that fundus photography combined with AI algorithms will be able to diagnose retinal disorders. Excellent detection precision between true positives and true negatives was achieved with an overall sensitivity of 89% and specificity of 92%. The fact that these algorithms have a high degree of discrimination is further supported by an AUC of 0.95, which shows how much potential AI has to bridge the gap between traditional diagnosis in clinics. These findings are consistent with other research showing AI's excellent sensitivity and specificity in identifying retinal disorders, particularly age-related macular degeneration and diabetic retinopathy.33 Hence, early detection can be supported by AI's consistency in several areas for better outcomes and management of patients.
According to the subgroup study, AI algorithms perform remarkably well in diabetic retinopathy, with an AUC of 0.96, sensitivity of 90%, and specificity of 94%. This finding is consistent with a growing body of research showing that artificial intelligence (AI) may effectively diagnose diabetic retinopathy, a condition that typically manifests asymptomatically at first.34 The most remarkable aspect of this analysis is that the deep learning models had the best sensitivity, specificity, and AUC. Numerous research has demonstrated that deep learning algorithms, particularly convolutional neural networks (CNNs), are more effective than typical machine learning models at identifying complex patterns in retinal images.35
The sensitivity study also demonstrated that AI models outperformed other models when given vast datasets. Datasets with more than 500 samples produced superior AUCs of 0.96, 92% sensitivity, and 95% specificity. This result confirms the hypothesis that AI models perform better on retinal disorders across a range of populations when they have access to larger and more varied datasets, which aid in their ability to generalize. These findings imply that the AI system can only further increase diagnosis accuracy when working with huge, high-quality datasets.36 These algorithms are trained on large datasets of annotated fundus images, allowing them to learn intricate features associated with various retinal diseases.37-41
Future research should examine AI models in other patient populations where generalizability may differ significantly, particularly from non-Western settings, even though the results given are encouraging.42 For example, some retinal diseases may have greater incidence rates, or some demographic characteristics may differ greatly.
This meta-analysis has several important advantages. For starters, it uses fundus photography to give a broad picture of how well AI algorithms diagnose different retinal conditions. It provides a reliable estimate of diagnostic accuracy, including sensitivity, specificity, and area under the curve, by combining the data from multiple investigations. A viable diagnostic tool for clinical application will be provided by high pooled sensitivity and specificity, which will show off AI's enormous potential in precisely identifying retinal illness. This finding will be strengthened and additional insights into factors influencing the AI models' performance will be provided by the stratification by disease type, AI model, and dataset characteristics.
The meta-analysis employs a comprehensive methodology by systematically including studies from multiple high-quality databases such as PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus, while also using the QUADAS-2 tool for quality assessment. This ensures a rigorous evaluation of AI performance in retinal disease detection.
Additionally, the study demonstrates robust diagnostic accuracy, with AI algorithms achieving high sensitivity (89%) and specificity (92%). Performance further improves with larger datasets, reaching 92% sensitivity, 95% specificity, and an AUC of 0.96, reinforcing the reliability and clinical potential of AI in retinal disease screening.
Despite the encouraging outcomes, several limitations need to be considered. First, the quality of the research included varies, which limits the meta-analysis. Some studies may still be subject to biases because of variations in data quality, study design, and patient selection. Furthermore, research with quantitative diagnostic characteristics (sensitivity, specificity, and AUC) was the only ones permitted by the inclusion criteria; studies that might have employed alternative methodologies or inadequate data reporting were not included. Additionally, the review mostly focused on English-language articles, resulting in language bias and eliminating several research published in other languages, despite the meta-analysis involving many databases. An additional drawback is that the investigation did not look into the long-term clinical results of AI Algorithms for Retinal Disease Detection. Higher sample numbers and high-quality datasets are required for algorithmic improvement and bias reduction. AI-based algorithms for retinal disease detection must be used in long-term longitudinal studies examining clinical outcomes during extended follow-ups to evaluate actual and real-life changes in patient outcomes. Furthermore, to guarantee patient safety and privacy, the ethical guidelines need to be addressed when implementing AI models in clinical practice.
This meta-analysis underscores the impressive accuracy of AI systems, particularly deep learning models, in detecting retinal diseases with high precision. The findings reveal that AI algorithms are not only capable of identifying conditions such as glaucoma, age-related macular degeneration, and diabetic retinopathy but also demonstrate a level of reliability that holds strong potential for clinical application. By enabling rapid, automated, and consistent disease detection, AI can significantly aid ophthalmologists in diagnosing retinal disorders at earlier stages, ultimately improving patient care and prognosis.
Moreover, the subgroup analysis further highlights the superior performance of deep learning models compared to traditional AI techniques. This advantage is particularly evident when these models are trained on large, diverse datasets, which enhance their generalizability and diagnostic accuracy across different patient populations. The ability of AI to continuously learn and improve from vast amounts of retinal imaging data makes it a valuable tool in ophthalmic practice.
These findings support the integration of AI-assisted diagnostic tools into routine clinical workflows, allowing for earlier intervention and better disease management. By reducing diagnostic variability and improving accessibility to high-quality retinal screening, AI-driven approaches have the potential to revolutionize ophthalmology, particularly in regions with limited access to specialist care.
8. Conflict of Interest
None declared by the authors
9. Ethics considerations
Ethical approval not required for meta-analysis.
10. Author’s contributions
All authors contributed to conception, design, writing and reviewing of the manuscript. All authors have approved the final version to be published.
Authors affiliations:
- Mujeeb Ur Rehman Parrey, Ph.D, Department of Surgery, Faculty of Medicine; Northern Border University, Arar; Kingdom of Saudi Arabia; Email: drparrey@gmail.com] {ORCID:0000-0002-2731-5438}
- Muhammad Omer Afzal Bhatti, MCPS, Department of Surgery, Faculty of Medicine; Northern Border University, Arar; Kingdom of Saudi Arabia; Email: omer666afzal@hotmail.com
- Maha M. Abdul-Latif, MS, Department of Surgery, Faculty of Medicine; Northern Border University, Arar; Kingdom of Saudi Arabia; Email: mahamlatif79@gmail.com
- Saima Rehman, M.Phil, Department of Physiology, Baqai Medical College, Baqai Medical University, Pakistan; Email: d_saimaomar@yahoo.com
- Mohammed M. Ismail, MD, Department of Anatomy, Faculty of Medicine; Northern Border University, Arar; Kingdom of Saudi Arabia; Email: rehamsaed04@gmail.com
- Omaima A. Hamid, MD; MRCOG, Department of Obstetrics and Gynecology, Faculty of Medicine; Northern Border University, Arar; Kingdom of Saudi Arabia; Email: omima.enan@gmail.com
ABSTRACT
Artificial intelligence (AI) is revolutionizing retinal diagnostics by enhancing the accuracy and efficiency of disease detection in fundus photography. Manual interpretation is time-consuming and subject to interobserver variability, highlighting the need for AI-driven solutions. This study assesses the diagnostic performance of AI algorithms in detecting retinal diseases using fundus photography. A systematic search was conducted in PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus, including studies reporting AI-based retinal disease detection with sensitivity, specificity, and area under the curve (AUC) metrics. The QUADAS-2 tool was used for quality assessment, and a random-effects model was applied for meta-analysis. AI algorithms demonstrated high diagnostic accuracy, with an overall sensitivity of 89% and specificity of 92%. Performance improved with larger datasets (>500 samples), achieving a sensitivity of 92%, specificity of 95%, and an AUC of 0.96. These findings suggest that AI has strong potential for clinical implementation in retinal disease screening, offering high accuracy for early diagnosis and improved patient outcomes.
Abbreviations: AI: Artificial intelligence, AUC: area under the curve,
Keywords: Artificial Intelligence; Algorithms; Deep Learning; Meta-Analysis; Fundus Photography; Retinal Disease Detection; Diagnostic Accuracy
Citation: Parrey MUR, Bhatti MOA, Abdul-Latif MM, Rehman S, Ismail MM, Hamid OA. Meta-analysis of AI algorithm performance in detecting retinal diseases. Anaesth. pain intensive care 2025;29(4):400-406. DOI: 10.35975/apic.v29i4.2828
Received: February 17, 2025; Revised: April 25, 2025; Accepted: April 25, 2025
1. INTRODUCTION
Artificial intelligence (AI) has emerged as a transformative force in medical diagnostics, revolutionizing clinical support systems across various specialties.1-5 In ophthalmology, AI applications have gained significant attention, particularly for detecting complex and historically challenging ocular conditions.6-9 Deep learning models have demonstrated high sensitivity and specificity in identifying retinal abnormalities by analysing large datasets, enabling them to recognize intricate disease-related features.10
By integrating AI into ophthalmic diagnostics, clinicians can expedite disease detection, minimize human error, and enhance accessibility to eye care, especially in regions with limited ophthalmologist availability.11 Retinal diseases are among the leading causes of visual impairment and blindness worldwide.12-13 Early detection and timely intervention are crucial to preventing irreversible vision loss.14 Fundus photography, a non-invasive imaging technique, plays a vital role in screening and diagnosing retinal pathologies by capturing high-resolution retinal images.15 However, manual interpretation of these images is time-consuming, subjective, and prone to interobserver variability, leading to diagnostic inconsistencies.16 The increasing prevalence of retinal diseases necessitates the development of more efficient and accurate diagnostic methods.17
Several studies have reported promising results regarding AI’s diagnostic accuracy, demonstrating high sensitivity, specificity, and area under the curve (AUC) values.18-19 However, variations in algorithm performance persist due to differences in model architecture, training datasets, and validation methodologies. Additionally, the generalizability of AI models across diverse populations remains a subject of ongoing investigation.20 A comprehensive synthesis of existing evidence is essential to assess the robustness and clinical applicability of AI in fundus photography-based diagnostics.21
This study was designed to evaluate the diagnostic performance of AI algorithms in retinal disease detection using fundus photography. By aggregating sensitivity, specificity, and AUC metrics from multiple studies, this meta-analysis provides a quantitative assessment of AI’s effectiveness. Furthermore, subgroup analyses will examine the impact of disease type, AI model, and dataset characteristics on diagnostic outcomes. The findings from the current study offer valuable insights into the strengths and limitations of AI-driven retinal disease detection, informing future research and potential clinical implementation.
The objective of this study was to evaluate the performance of AI algorithms in detecting retinal diseases using fundus photography.
2. METHODOLOGY
A comprehensive and detailed search strategy was used to look for pertinent studies assessing the effectiveness of AI algorithms for retinal disease detection using fundus photography in several electronic databases, including PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus. "AI algorithms," "retinal disease," "fundus photography," and "diagnostic performance," among other Medical Subject Headings (MeSH) terms, were utilized in conjunction with keywords to search as shown in Table 1.
| Table 1: Summary of key study characteristics in the meta-analysis | ||||||
| Author(s) | Year | AI Model | Disease | Sample size | Diagnostic Metrics | Source of
Dataset |
| Dai et al. [22] | 2021 | Deep Learning, CNN | Diabetic Retinopathy | 200,136 images | Sensitivity, Specificity | Multiple datasets |
| Gargeya et al. [23] |
2017 | Deep Learning, CNN | Diabetic Retinopathy | 75,137 images | Sensitivity, Specificity | Multiple datasets |
| Ting et al. [24] |
2017 | Deep Learning, CNN | Diabetic Retinopathy | 494,661 images | Sensitivity, Specificity, AUC | Multiethnic Populations |
| Yi Zhen et al. [25] |
2020 | Deep Learning, CNN | Central Serous Chorioretinopathy | 2,504 fundus images | AUC | Custom dataset |
| Gulshan et al. [26] |
2016 | Deep Learning, CNN | Diabetic Retinopathy | 128,175 images | Sensitivity, Specificity, AUC | Multiple datasets |
| Kermany et al. [27] |
2018 | Deep Learning, CNN | Multiple diseases | 108,312 images | Sensitivity, Specificity | Multiple datasets |
| Li et al. [28] |
2019 | Deep Learning, CNN | Retinal Disorders | 207,228 images | Sensitivity, Specificity | Multiple datasets |
| Son et al. [29] |
2020 | Deep Learning, CNN | Multiple Abnormal Findings | 103,262 images | Sensitivity, Specificity | Multiple datasets |
| Milea et al. [30] |
2020 | Deep Learning, CNN | Papilledema | 15,846 images | Sensitivity, Specificity | Multiple datasets |
| Üzen et al. [31] | 2020 | Deep Learning, CNN | Central Serous Chorioretinopathy | 3860 images | AUC | Custom dataset |
| Brown et al. [32] |
2018 | Deep Learning, CNN | Plus Disease in ROP | 5,511 images | Sensitivity, Specificity | Multiple Data sets |
To guarantee that the most recent material was included, only English-language publications were taken into consideration, and there were no date limits. To find further research that satisfied the inclusion requirements, references from pertinent publications were manually examined. The included studies comprised one utilizing fundus photography to assess the diagnostic potential of AI algorithms in detecting retinal disorders such as diabetic retinopathy, age-related macular degeneration, and glaucoma; another presenting quantitative diagnostic parameter like AUC, sensitivity, and specificity; and a peer-reviewed study. Studies were excluded if they did not apply AI models to diagnose retinal diseases in fundus images or lacked sufficient data on diagnostic measures.
Additionally, case reports, abstracts, and studies with a sample size of fewer than thirty were disqualified. The quality of the studies included was evaluated using the QUADAS-2 approach. So, four main domains are evaluated for the possibility of bias such as: time, standard reference and selection of patient. To ensure the validity and reliability of the results, studies whose findings would be highly susceptible to bias were eliminated. Each included study's data was extracted by two independent reviewers. The data that was extracted comprised diagnostic measures like sensitivity, specificity, and AUC; dataset features like training dataset size; and study characteristics like study design, sample size, AI model utilized, and disease type.
Additionally, a random effects model was used to pool diagnostic parameters such as sensitivity, specificity, and AUC. The choice to pool diagnostics using this method was made considering the expected heterogeneity of studies, the algorithms used by AI in their research, and the disorders taken into consideration. AUCs were used to summarize the overall measurement accuracy of diagnostic detection, while sensitivity and specificity were combined for estimations of summary along with the relevant 95% CIs. The included studies' heterogeneity was evaluated using the I2 statistic. If a significant heterogeneity (I2 > 50%) was found, the variation was evaluated using subgroup analyses of the source. To find out how important aspects affect AI performance, a subgroup analysis was done. These factors included the type of disease of retina, the particular AI model used, such as deep learning models and convolutional neural networks; and the dataset's attributes, such as its size, population characteristics, and validation process.
The purpose of the analysis was to detect trends in the way AI algorithms performed and to evaluate how reliable the results were under various circumstances. A sensitivity analysis is carried out for specific studies to investigate the impact on the outcome. Studies with a high risk of bias or outliers are subjected to separate analyses to determine whether removing them significantly changes the pooled estimate. R software (version 4.0.3) and Review Manager (RevMan) version 5.4 were used for all statistical analyses. The pooled estimates of sensitivity, specificity, and AUC were displayed graphically using forest plots. Ethical approval was not required because this was a meta-analysis based on an analysis of published data.
3. RESULTS
The results of the study revealed that AI algorithms for fundus photography-based retinal disease identification perform well overall. The algorithms' 89% sensitivity means they accurately identify 89% of genuine positives, but their 92% specificity means they can correctly identify 92% of true negatives, which means there are fewer false positives. The area under the curve of 0.95 is another indication of these AI models' strong discriminative ability as shown in Table 2.
| Table 2: Meta-analysis of AI performance in retinal disease detection | ||
| Variable | Value | 95% onfidence Interval |
| Sensitivity | 89% | [85%, 93%] |
| Specificity | 92% | [88%, 96%] |
| Area Under Curve (AUC) | 0.95 | [0.92, 0.98] |

Figure 1: PRISMA flow diagram for meta-analysis of AI algorithms in retinal disease detection using fundus photography
As shown in Table 2 subgroup analyses, however, demonstrated the highest test performance in individuals with diabetic retinopathy who were found in the study. They demonstrated an excellent AUC of 0.96, a relatively high sensitivity of 90%, and a good specificity of 94%. Compared to other models, deep learning models performed better, with the best sensitivity (91%), specificity (93%), and AUC (0.97%). Convolutional neural networks and other factors caused other AI models to perform comparatively worse. AI algorithms also performed better on larger datasets; sensitivity (92%), specificity (95%), and AUC (0.96) were greater in datasets with over 500 samples than in datasets with fewer samples. These findings generally show that AI algorithms, particularly those that use deep learning approaches, perform well in identifying retinal illnesses, especially when working with larger datasets.
| Table 3: Subgroup Analysis | ||||
| Subgroups | Sensitivity (%) | Specificity (%) | AUC | |
| Type of Disease
|
Diabetic Retinopathy Age-related muscular degeneration Glaucoma Other diseases of the Retina |
90% 85% 88% 87% |
94% 90% 93% 91% |
0.96 0.94 0.95 0.93 |
| AI Model
|
Deep Learning Models Convolutional Neural Networks Other AI Models |
91% 87% 85% |
93% 91% 89% |
0.97 0.94 0.92 |
| Type of dataset
|
Small Dataset (N < 500) Large Dataset (N > 500) |
84% 92% |
89% 95% |
0.90 0.96 |
4. DISCUSSION
The results of this meta-analysis have increased the likelihood that fundus photography combined with AI algorithms will be able to diagnose retinal disorders. Excellent detection precision between true positives and true negatives was achieved with an overall sensitivity of 89% and specificity of 92%. The fact that these algorithms have a high degree of discrimination is further supported by an AUC of 0.95, which shows how much potential AI has to bridge the gap between traditional diagnosis in clinics. These findings are consistent with other research showing AI's excellent sensitivity and specificity in identifying retinal disorders, particularly age-related macular degeneration and diabetic retinopathy.33 Hence, early detection can be supported by AI's consistency in several areas for better outcomes and management of patients.
According to the subgroup study, AI algorithms perform remarkably well in diabetic retinopathy, with an AUC of 0.96, sensitivity of 90%, and specificity of 94%. This finding is consistent with a growing body of research showing that artificial intelligence (AI) may effectively diagnose diabetic retinopathy, a condition that typically manifests asymptomatically at first.34 The most remarkable aspect of this analysis is that the deep learning models had the best sensitivity, specificity, and AUC. Numerous research has demonstrated that deep learning algorithms, particularly convolutional neural networks (CNNs), are more effective than typical machine learning models at identifying complex patterns in retinal images.35
The sensitivity study also demonstrated that AI models outperformed other models when given vast datasets. Datasets with more than 500 samples produced superior AUCs of 0.96, 92% sensitivity, and 95% specificity. This result confirms the hypothesis that AI models perform better on retinal disorders across a range of populations when they have access to larger and more varied datasets, which aid in their ability to generalize. These findings imply that the AI system can only further increase diagnosis accuracy when working with huge, high-quality datasets.36 These algorithms are trained on large datasets of annotated fundus images, allowing them to learn intricate features associated with various retinal diseases.37-41
Future research should examine AI models in other patient populations where generalizability may differ significantly, particularly from non-Western settings, even though the results given are encouraging.42 For example, some retinal diseases may have greater incidence rates, or some demographic characteristics may differ greatly.
This meta-analysis has several important advantages. For starters, it uses fundus photography to give a broad picture of how well AI algorithms diagnose different retinal conditions. It provides a reliable estimate of diagnostic accuracy, including sensitivity, specificity, and area under the curve, by combining the data from multiple investigations. A viable diagnostic tool for clinical application will be provided by high pooled sensitivity and specificity, which will show off AI's enormous potential in precisely identifying retinal illness. This finding will be strengthened and additional insights into factors influencing the AI models' performance will be provided by the stratification by disease type, AI model, and dataset characteristics.
5. STRENGHTHS
The meta-analysis employs a comprehensive methodology by systematically including studies from multiple high-quality databases such as PubMed, Embase, Cochrane Library, IEEE Xplore, and Scopus, while also using the QUADAS-2 tool for quality assessment. This ensures a rigorous evaluation of AI performance in retinal disease detection.
Additionally, the study demonstrates robust diagnostic accuracy, with AI algorithms achieving high sensitivity (89%) and specificity (92%). Performance further improves with larger datasets, reaching 92% sensitivity, 95% specificity, and an AUC of 0.96, reinforcing the reliability and clinical potential of AI in retinal disease screening.
6. LIMITATIONS
Despite the encouraging outcomes, several limitations need to be considered. First, the quality of the research included varies, which limits the meta-analysis. Some studies may still be subject to biases because of variations in data quality, study design, and patient selection. Furthermore, research with quantitative diagnostic characteristics (sensitivity, specificity, and AUC) was the only ones permitted by the inclusion criteria; studies that might have employed alternative methodologies or inadequate data reporting were not included. Additionally, the review mostly focused on English-language articles, resulting in language bias and eliminating several research published in other languages, despite the meta-analysis involving many databases. An additional drawback is that the investigation did not look into the long-term clinical results of AI Algorithms for Retinal Disease Detection. Higher sample numbers and high-quality datasets are required for algorithmic improvement and bias reduction. AI-based algorithms for retinal disease detection must be used in long-term longitudinal studies examining clinical outcomes during extended follow-ups to evaluate actual and real-life changes in patient outcomes. Furthermore, to guarantee patient safety and privacy, the ethical guidelines need to be addressed when implementing AI models in clinical practice.
7. CONCLUSION
This meta-analysis underscores the impressive accuracy of AI systems, particularly deep learning models, in detecting retinal diseases with high precision. The findings reveal that AI algorithms are not only capable of identifying conditions such as glaucoma, age-related macular degeneration, and diabetic retinopathy but also demonstrate a level of reliability that holds strong potential for clinical application. By enabling rapid, automated, and consistent disease detection, AI can significantly aid ophthalmologists in diagnosing retinal disorders at earlier stages, ultimately improving patient care and prognosis.
Moreover, the subgroup analysis further highlights the superior performance of deep learning models compared to traditional AI techniques. This advantage is particularly evident when these models are trained on large, diverse datasets, which enhance their generalizability and diagnostic accuracy across different patient populations. The ability of AI to continuously learn and improve from vast amounts of retinal imaging data makes it a valuable tool in ophthalmic practice.
These findings support the integration of AI-assisted diagnostic tools into routine clinical workflows, allowing for earlier intervention and better disease management. By reducing diagnostic variability and improving accessibility to high-quality retinal screening, AI-driven approaches have the potential to revolutionize ophthalmology, particularly in regions with limited access to specialist care.
8. Conflict of Interest
None declared by the authors
9. Ethics considerations
Ethical approval not required for meta-analysis.
10. Author’s contributions
All authors contributed to conception, design, writing and reviewing of the manuscript. All authors have approved the final version to be published.
11. REFERENCES
- Al-Antari MA. Artificial intelligence for medical diagnostics - existing and future AI technology. Diagnostics (Basel). 2023;13(4):688. PubMed DOI: 3390/diagnostics13040688
- Kulkarni PA, Singh H. Artificial intelligence in clinical diagnosis: opportunities, challenges, and hype. JAMA. 2023;330(4):317-8. PubMed DOI: 1001/jama.2023.11440
- Alowais SA, Alghamdi SS, Alsuhebany N, Alqahtani T, Alshaya AI, Almohareb SN, et al. Revolutionizing healthcare: the role of artificial intelligence in clinical practice. BMC Med Educ. 2023;23(1):689. PubMed DOI: 1186/s12909-023-04698-z
- Bajwa J, Munir U, Nori A, Williams B. Artificial intelligence in healthcare: transforming the practice of medicine. Future Healthc J. 2021;8(2):e188-94. PubMed DOI: 7861/fhj.2021-0095
- Davenport T, Kalakota R. The potential for artificial intelligence in healthcare. Future Healthc J. 2019;6(2):94-8. PubMed DOI: 7861/futurehosp.6-2-94
- Honavar SG. Artificial intelligence in ophthalmology - Machines think! Indian J Ophthalmol. 2022;70(4):1075-9. PubMed DOI: 4103/ijo.IJO_644_22
- Li Z, Wang L, Wu X, Jiang J, Qiang W, Xie H, et al. Artificial intelligence in ophthalmology: the path to the real-world clinic. Cell Rep Med. 2023;4(7):101095. PubMed DOI: 1016/j.xcrm.2023.101095
- Hashemian H, Peto T, Ambrósio R Jr, Lengyel I, Kafieh R, Noori AM, et al. Application of artificial intelligence in ophthalmology: an updated comprehensive review. J Ophthalmic Vis Res. 2024;19(3):354-67. PubMed DOI: 18502/jovr.v19i3.15893
- Ahuja AS, Wagner IV, Dorairaj S, Checo L, Hulzen RT. Artificial intelligence in ophthalmology: a multidisciplinary approach. Integr Med Res. 2022;11(4):100888. PubMed DOI: 1016/j.imr.2022.100888
- Parmar UPS, Surico PL, Singh RB, Romano F, Salati C, Spadea L, et al. Artificial intelligence (AI) for early diagnosis of retinal diseases. Medicina (Kaunas). 2024;60(4):527. PubMed DOI: 3390/medicina60040527
- Khalifa M, Albadawy M. AI in diagnostic imaging: revolutionising accuracy and efficiency. Comput Methods Programs Biomed Update. 2024;5:100146. DOI: 1016/j.cmpbup.2024.100146
- GBD 2019 Blindness and Vision Impairment Collaborators, Vision Loss Expert Group of the Global Burden of Disease Study. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: the Right to Sight: an analysis for the Global Burden of Disease Study. Lancet Glob Health. 2021;9(2):e144-60. PubMed DOI: 1016/S2214-109X(20)30489-7
- Kavitha V, Heralgi MM, Gangrade AK. Commentary: Understanding irreversible blindness—the need of the hour; Reversing it - the need of the future! Indian J Ophthalmol. 2021;69(10):2636-7. PubMed DOI: 4103/ijo.IJO_1224_21
- Balyen L, Peto T. Promising artificial intelligence–machine learning–deep learning algorithms in ophthalmology. Asia Pac J Ophthalmol (Phila). 2019;8(3):264-72. PubMed DOI: 22608/APO.2018479
- Goutam B, Hashimi MF, Geem ZW, Bokde N. A comprehensive review of deep learning strategies in retinal disease diagnosis using fundus images. IEEE Access. 2022;10:57796-823. DOI: 1109/ACCESS.2022.3178372
- Medeiros FA, Jammal AA, Thompson AC. From machine to machine: an OCT-trained deep learning algorithm for objective quantification of glaucomatous damage in fundus photographs. Ophthalmology. 2019;126(4):513-21. PubMed DOI: 1016/j.ophtha.2018.12.033
- S Sorrentino FS, Gardini L, Fontana L, Musa M, Gabai A, Maniaci A, et al. Novel approaches for early detection of retinal diseases using artificial intelligence. J Pers Med. 2024;14(7):690. PubMed DOI: 3390/jpm14070690
- Nielsen KB, Lautrup ML, Andersen JKH, Savarimuthu TR, Grauslund J. Deep learning-based algorithms in screening of diabetic retinopathy: a systematic review of diagnostic performance. Ophthalmol Retina. 2019;3(4):294-304. PubMed DOI: 1016/j.oret.2018.10.014
- Shen J, Zhang CJP, Jiang B, Chen J, Song J, Liu Z, et al. Artificial intelligence versus clinicians in disease diagnosis: systematic review. JMIR Med Inform. 2019;7(3):e10010. PubMed DOI: 2196/10010
- Liang W, Tadesse GA, Ho D, et al. Advances, challenges and opportunities in creating data for trustworthy AI. Nat Mach Intell. 2022;4:669-77. DOI: 1038/s42256-022-00516-1
- Heger KA, Waldstein SM. Artificial intelligence in retinal imaging: current status and future prospects. Expert Rev Med Devices. 2024;21(1-2):73-89. PubMed DOI: 1080/17434440.2023.2294364
- Dai L, Wu L, Li H, Cai C, Wu Q, Kong H, et al. A deep learning system for detecting diabetic retinopathy across the disease spectrum. Nat Commun. 2021;12(1):3242. PubMed DOI: 1038/s41467-021-23458-5
- Gargeya R, Leng T. Automated identification of diabetic retinopathy using deep learning. Ophthalmology. 2017;124(7):962-9. PubMed DOI: 1016/j.ophtha.2017.02.008
- Ting DSW, Cheung CY, Lim G, Tan GSW, Quang ND, Gan A, et al. Development and validation of a deep learning system for diabetic retinopathy and related eye diseases using retinal images from multiethnic populations with diabetes. JAMA. 2017;318(22):2211-22. PubMed DOI: 1001/jama.2017.18152
- Zhen Y, Chen H, Zhang X, Meng X, Zhang J, Pu J. Assessment of central serous chorioretinopathy depicted on color fundus photographs using deep learning. Retina. 2020;40(8):1558-64. PubMed DOI: 1097/IAE.0000000000002621
- Gulshan V, Peng L, Coram M, Stumpe MC, Wu D, Narayanaswamy A, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-11. PubMed DOI: 1001/jama.2016.17216
- Kermany DS, Goldbaum M, Cai W, Valentim CCS, Liang H, Baxter SL, et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell. 2018;172(5):1122-31.e9. PubMed DOI: 1016/j.cell.2018.02.010
- Li F, Chen H, Liu Z, Zhang X, Wu Z. Fully automated detection of retinal disorders by image-based deep learning. Graefes Arch Clin Exp Ophthalmol. 2019;257(3):495-505. PubMed DOI: 1007/s00417-018-04224-8
- Son J, Shin JY, Kim HD, Jung KH, Park KH, Park SJ. Development and validation of deep learning models for screening multiple abnormal findings in retinal fundus images. Ophthalmology. 2020;127(1):85-94. PubMed DOI: 1016/j.ophtha.2019.05.029
- Milea D, Najjar RP, Zhubo J, Ting D, Vasseneix C, Xu X, et al. Artificial intelligence to detect papilledema from ocular fundus photographs. N Engl J Med. 2020;382(18):1687-95. PubMed DOI: 1056/NEJMoa1917130
- Üzen H, Fırat H, Özçelik STA, Yusufoğlu E, Çiçek İB, Şengür A. Central serous retinopathy classification with deep learning-based multilevel feature extraction from optical coherence tomography images. Opt Laser Technol. 2025;184:112519. DOI: 1016/j.optlastec.2025.112519
- Brown JM, Campbell JP, Beers A, Chang K, Ostmo S, Chan RVP, et al. Automated diagnosis of plus disease in retinopathy of prematurity using deep convolutional neural networks. JAMA Ophthalmol. 2018;136(7):803-10. PubMed DOI: 1001/jamaophthalmol.2018.1934
- El-Den NN, Elsharkawy M, Saleh I, Ghazal M, Khalil A, Haq MZ, et al. AI-based methods for detecting and classifying age-related macular degeneration: a comprehensive review. Artif Intell Rev. 2024;57(9):237. DOI: 1007/s10462-024-10883-3
- He J, Cao T, Xu F, Wang S, Tao H, Wu T, et al. Artificial intelligence-based screening for diabetic retinopathy at community hospital. Eye (Lond). 2020;34(3):572-6. PubMed DOI: 1038/s41433-019-0562-4
- Islam MM, Yang HC, Poly TN, Jian WS, Li YCJ. Deep learning algorithms for detection of diabetic retinopathy in retinal fundus photographs: a systematic review and meta-analysis. Comput Methods Programs Biomed. 2020;191:105320. PubMed DOI: 1016/j.cmpb.2020.105320
- Jayakumar S, Sounderajah V, Normahani P, Harling L, Markar SR, Ashrafian H, et al. Quality assessment standards in artificial intelligence diagnostic accuracy systematic reviews: a meta-research study. NPJ Digit Med. 2022;5(1):11. PubMed DOI: 1038/s41746-021-00544-y
- Schmidt-Erfurth U, Sadeghipour A, Gerendas BS, Waldstein SM, Bogunović H. Artificial intelligence in retina. Prog Retin Eye Res. 2018;67:1-29. PubMed DOI: 1016/j.preteyeres.2018.07.004
- Zhou Y, Chia MA, Wagner SK, Ayhan MS, Williamson DJ, Struyven RR, et al. A foundation model for generalizable disease detection from retinal images. Nature. 2023;622(7981):156-63. PubMed DOI: 1038/s41586-023-06555-x
- Al-Halafi AM. Applications of artificial intelligence-assisted retinal imaging in systemic diseases: a literature review. Saudi J Ophthalmol. 2023;37(3):185-92. PubMed DOI: 4103/sjopt.sjopt_153_23
- Zapata MA, Royo-Fibla D, Font O, Vela JI, Marcantonio I, Moya-Sánchez EU, et al. Artificial intelligence to identify retinal fundus images, quality validation, laterality evaluation, macular degeneration, and suspected glaucoma. Clin Ophthalmol. 2020;14:419-29. PubMed DOI: 2147/OPTH.S235751
- Cao S, Zhang R, Jiang A, Kuerban M, Wumaier A, Wu J, et al. Application effect of an artificial intelligence-based fundus screening system: evaluation in a clinical setting and population screening. Biomed Eng Online. 2023;22(1):38. PubMed DOI: 1186/s12938-023-01097-9
- Okolo CT. Optimizing human-centered AI for healthcare in the Global South. Patterns (N Y). 2022;3(2):100421. PubMed DOI: 1016/j.patter.2021.100421