Angham Najah Al-Khafaji 1 , Zahraa Yosif Motaweq 2
Authors affiliations:
Background & Objective: Lupus Nephritis (LN) is considered a serious manifestation of SLE (SLE). which is diagnosed through Immunological biomarker. Interleukin-35 (IL-35), a member of the interleukin-12 family, has been identified as immunosuppressive and anti-inflammatory cytokine. In the past ten years, the role of IL-35 in inflammatory and autoimmune diseases has been studied extensively. We aimed to detect LN using immunological markers and to evaluate serum levels of iL-35 in LN patients with and without bacteria.
Methodology: Serum levels of iL-35 were measured in 120 adults, including 60 patients with LN, 30 patients with kidney failure and 30 from healthy volunteers using ELISA.
Results: Females were found to be highly infected with LN compared with males. LN was more prevalent at the age of 15-45 years and more highly infected with the disease compared with other ages. Anti-dsDNA and Anti-C1q antibodies are good Immunological markers to consider to detect LN patients. Levels of serum IL-35 were significantly higher (P < 0.05) in the LN group compared with those with kidney failure (KF Group) and healthy groups. IL-35 levels were significantly higher in LN patients - (mean 90.36 ± 20.02 pg/mL), than Kidney failure group (77.15 ± 25.7 pg/mL) and the Healthy group (70.76 ± 17.96 pg/mL) (P ≤ 0.001). The concentration of IL-35 was high in males infected and non-infected with bacteria as compared to females. The differences in the results of these two subgroups were statistically not significant at a mean probability of P ≤ 0.05.
Conclusion: It was concluded from the results of this study that the increase in interleukin-35 in patients with lupus nephritis compared with renal failure and healthy groups is considered a predictive sign for detecting the disease, in addition to the role of bacteria in raising interleukin 35 in the subgroups lupus nephritis and renal failure compared to the healthy groups.
Abbreviations: APC: antigen presenting cells, IL-35: Interlukein-35, SLE; SLE, LN: Lupus nephritis, ELISA: Enzyme Linked Immuno-Sorbent Assay, dsDNA: double stranded DNA, OD: optical density, ROC: Receiver operator characteristic
Keywords: Anti-dsDNA; Anti C1q; ELIZA; Interlukein-35; Lupus Nephritis; SLE
Citation: Al-Khafaji AN, Motaweq ZY. Evaluation and management of interlukein-35 (IL-35) in prognosis of patient with lupus nephritis. Anaesth. pain intensive care 2025;29(4):308-17. DOI: 10.35975/apic.v29i4.xxxx
Received: March 07, 2025; Revised: April 27, 2025; Accepted: April 27, 2025
Lupus nephritis (LN) is a common complication of SLE (SLE). Mild sub-nephrotic proteinuria to diffuse progressive glomerulonephritis leading to chronic kidney disease are the results. LN usually manifests first in the course of SLE. Proteinuria, hematuria, edema of the lower extremities, increased creatinine levels and hypertension are the signs of LN. In order to diagnose LN and rule out alternative causes, a histopathological biopsy is essential. The six LN classes are revealed by the histopathology biopsy used in this investigation.1 T-cells play a major role in the pathogenesis of SLE, by promoting inflammation through the release of pro-inflammatory cytokines, inducing B-cells to produce auto-antibodies, and maintaining the disease through an accumulation of auto-reactive memory T-cells.
T-follicular helper-cells are essential for germinal center induction, proliferation, isotype-switching, and somatic hyper-mutation, as well as for direct infiltration and cytotoxicity to renal parenchymal tissues. However, the ratios of specific T-cell subsets and their function are aberrant in LN patients. The two main subpopulations of T-lymphocytes are CD4+ helper T-cells and CD8+ cytotoxic T-cells.2 Numerous mononuclear cells, CD4+ T-cells and CD8+ T-cells have been seen in the tubulointerstitial lesions in patients with LN.3
The most reliable method for identifying, categorizing and predicting LN is renal biopsy. However, it cannot be extensively used due to a number of disadvantages, such as it being an invasive procedure, bleeding risk, and potential of sample error.4 Furthermore, serial biopsies are not possible due to the invasive nature and the possibility of complications linked with the procedure.5
Anti-double stranded DNA (dsDNA) antibodies play an important role in the diagnosis, classification and management of SLE.6 High titers of anti-dsDNA antibodies are associated with the presence of SLE and are frequently used to follow the progress of LN.7
Interleukin-35 is a unique inhibitory cytokine that is part of the IL-12 cytokine family containing IL-12 subunit a chain (P35) subunits and Epstein-Barr virus-induced gene 3 (EBI3). It was discovered that, in contrast to other members of the IL-12 family, which are known to be mainly secreted by activated antigen presenting cells (APC), it is secreted by a variety of cell types and in a wide range of tissues, including monocytes, smooth muscle cells, endothelial cells, dendritic cells (DCs), and regulatory B-cells.
Numerous autoimmune diseases, inflammatory diseases, bacterial and viral infectious diseases, and tumors have also been shown to be significantly regulated by IL-35.8 Surprisingly, its biological functions differ from those of its siblings primarily in that it improves Treg-cell differentiation and immune suppression by transforming native T-cells into an IL-35 producing induced Treg-cell population known as iTr35,9 which directly inhibits effector T-cell proliferation in an APC-free culture in vitro.10
The present study aimed to estimate the levels of IL-35 in the serum of patients with and without bacterial infections. In addition, we sought to examine any possible variations between males and females at these levels. This research will help better understanding of how IL-35 functions in autoimmune nephritis and how gender affects these levels.
A case control study design was used for this study. We collected 120 samples; e.g., 60 from LN patients, 30 from kidney failure patients (KF Group) and 30 healthy adults, with age range 11-70 years. Clinical samples were collected from Al-Sader Medical City in Al-Najaf Province from December 2023 to May 2024. The research was preapproved by the Institutional Higher Scientific and Ethical Committee. All participants gave their written informed permission.
2.1. Specimen Collection
Venous blood samples 15 mL were drawn from all LN patients and healthy groups. Blood of all samples, 4 mL was transmitted into vacuum gel tubes and left at room temperature for at least 30 min for clotting, then centrifuged at 4000 rpm for 10 min. The serum 0.5 mL was poured into Eppendrff tubes for estimating the levels of anti-dsDNA, anti-C1q, and IL-35, and frozen at -20°C until used.
2.2. Estimation of serum biomarker levels
Enzyme Linked Immuno-sorbent Assay (ELISA) kit (Mindray; Germany) uses Sandwich-ELISA as the technique. The Micro ELIZA strip plate provided in this kit has been pre-coated with an antibody specific to (anti-dsDNA, anti-C1q, IL-35). Added, standards or samples to the suitable Micro ELIZA strip plate wells, combines to the specific antibody. Then, added a Horseradish Peroxidase (HRP)-conjugated antibody specific for biomarker to each Micro ELIZA strip plate well and incubated. Free components are washed method. Added the substrate solution (TMB) to all well. Only those wells that contain biomarker and HRP-conjugated double stranded antibody will appear blue in color and then turn yellow after the addition of the stop solution. The optical density (OD) is measured with spectrophotometer (Mindray; Germany) at a wavelength of 450 nm. The value of OD is relative estimate to the concentration of biomarker. One can calculate the concentration of biomarker in the samples by comparing the OD of the samples to the standard curve, as demonstrated in Figures 1 (a, b and c).
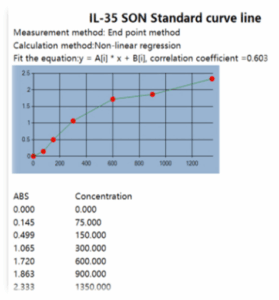
Figure 1 (a): The standard curve of biomarker IL-35
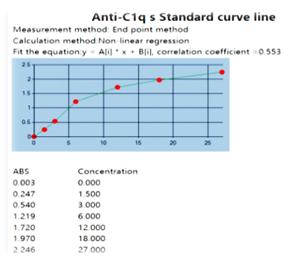
Figure 1 (b): The standard curve of biomarker anti-C1q
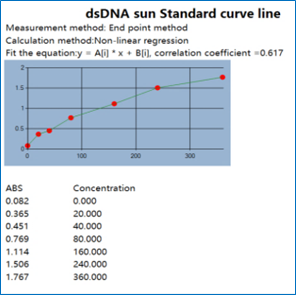
Figure 1 (c): The standard curve of biomarker
anti-dsDNA.
2.3. Statistical Analysis
This is a case control study. Data were tested using statistical software IBM SPSS. The data were accessible as mean ± standard deviation. The Kolmogorov-Smirnov test for Normal distribution uses the normal distribution. Comparison of groups using Chi-square have been done in order to investigate the association of the categorical and numerical variables, respectively, ANOVA, ROC curve analysis, Pearson’s correlation coefficient (r) test was used to evaluate the degree of relationship between two sets of variables. P < 0.05 was considered significant.
A total of 120 samples were collected and divided into two groups, the patients group and the control group.
The patient group included 60 patients suffering from LN, diagnosed by a specialist physician, and with laboratory findings. While the control group was divided into two group (KF Group and Healthy adults).
The majority of LN patients were females; 45 (75%) vs. 15 (25%) males. The age of patients in LN group ranged between 11-70 years (Figure 2).
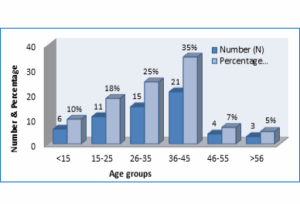
Figure 2: Distribution of LN patients according to age group
3.1. Immunological Marker to Detect LN
3.1.1. Anti-dsDNA antibody
The results of present study have illustrated a significant difference in the serum level of anti-dsDNA Ab in patients infected with LN, compare with control groups. The anti-dsDNA Ab level was elevated in the serum of LN patients; e.g., 10.47 ± 8.85 pg/mL versus 9.25 ± 4.3 pg/mL in KF Group and 4.37 ± 1.64 pg/mL in Healthy group (P = 0.001) as show in Figure 3.
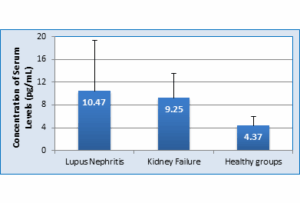
Figure 3: Anti-dsDNA antibody levels in serum of patients with LN compared with Control Groups (P = 0.05 - Significant)
3.1.2. Anti-C1q Antibody
Figure 4 shows the serum levels of Anti-C1q antibody in LN group compared with control group. The results demonstrate that there was a significant decrease (P ≤ 0.002) in patients mean serum levels; e.g., 0.90 ± 0.27 pg/mL in LN group versus 0.94 ± 0.21 pg/mL in KF Group and 1.19 ± 0.5 pg/mL in Healthy patient group.
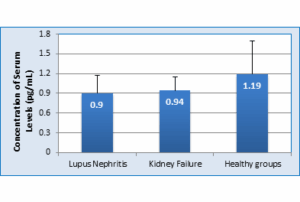
Figure 4: Anti-C1q antibody levels in serum of patients infect LN compared with Control Groups (P = 0.05 Significant)
3.1.3. Evaluation of serum IL-35 levels
The results showed that mean serum levels of IL-35 were significantly higher in LN group (85.40 ± 9.7 pg/mL),
compared to KF Group (67.60 ± 12.1 pg/mL) and Healthy group (62.80 ± 12.2 pg/mL). The differences were significant (P = 0.0001) as show in Figure 5.
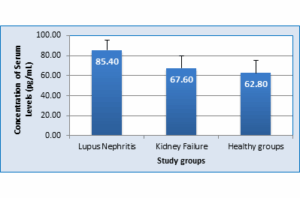
Figure 5: Interleukin-35 levels of serum in LN patients compared with control groups (P = 0.05 Significant)
Being higher in LN cases, the IL-35 levels were analyzed using receiver operator characteristic curve (ROC) analysis in order to find cutoff value for IL-35 that can predict positive diagnosis of LN. The cutoff value of IL-35 was > 85.69. However, it had confidence interval (Cl) 0.86-0.98 % since the area under curve (AUC) was 0.923 despite that ROC test was highly significant (P= 0.0001). The sensitivity and specificity of IL-35 cutoff value were 77.5% and 96%, respectively as shown in Figure 6 and Table 1.
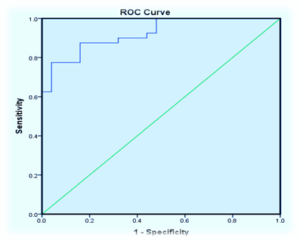
Figure 6: ROC curve analysis to identify the IL-35 to find the cutoff in LN patients versus control groups as a potential diagnostic marker
Present study revealed that the concentration of IL-35 was high in males with or without bacterial infections compared to females. The mean of females and males with LN was 90.2 ± 1pg/mL and 86.7 ± 2.2pg/mL respectively. In KF Group mean values were 75.2 ± 17.5 pg/mL and 70.9 ± 8.4 pg/mL, and in healthy groups the mean value was 64.3 ± 13.2 pg/mL and 62.4 ± 11.4 pg/mL for females and males respectively. The values for female LN patients without bacterial infect compared with those with bacterial infection were 76.8 ± 2.4 pg/mL, KF Group 62.1 ± 6.1 pg/mL and healthy groups 58.8 ± 0.00 pg/mL. while mean of males to LN was 74.0 ± 2.1 pg/mL, KF Group 59.2 ± 7.4 pg/mL, and healthy groups 48.5 ± 0.00 pg/mL. The differences in the results of these two subgroups were statistically not significant (P ≤ 0.05) as given in Table 2.
3.1.4. Correlations between parameters in LN patients
The correlations between three immunological parameters have been deducted. The results of the present study demonstrated that there were positive correlations between all parameter in patients suffering from LN.
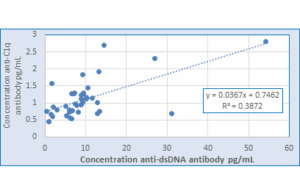
Figure 7: Correlation between concentration anti-dsDNA and anti-C1q antibody levels in LN Patients
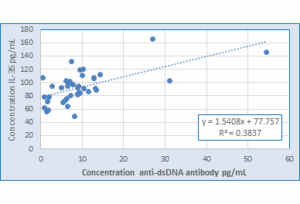
Figure 8: Correlation between concentration of anti-dsDNA antibody and IL-35 Levels in LN patients
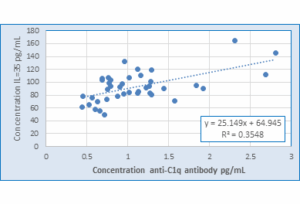
Figure 9: Correlation between concentration of anti-C1q antibody and IL-35 Levels in LN patients
The present study explains that the females are more affected than males, because of sex hormones, and X-chromosome genes in females with high effect and sensitivity to autoimmune disease.
According to studies by Moulton VR.,11 females are more likely to develop autoimmune inflammatory diseases than the males, and the estrogen increases severity of these diseases. The immune system is impacted by this condition, which is linked to steroid hormones, particularly progesterone and estrogen. Lower progesterone levels in lupus-affected women, indicates that a relative difference for estrogen may be an indicator that influences certain female patients' to immunological reactivity.11 Woo et al,12 suggested that the incidence of SLE in females was 100%. This could be because of the possible roles of gender-based differences in environmental exposures or sensitivity to these exposures, as well as genetic variations on the X chromosome and sex hormone milieus, as risk factors for incident SLE. High female to male ratio indicates that gender variables influence the development and severity of the disease.13 Depending on the relationship between the age and other factors implicated in the pathogenesis of SLE, the ratio of female to male in this condition might range from 9:1.14 A previous study demonstrated that the percentage of females (57.4 %) with LN is slightly higher than males (42.6 %).15 These findings could be explained by hormonal differences between the genders. Estradiol acts through its receptors and contributes to the sexual orientation in SLE. Most of the studies in the world revealed wide range of female to male differences.16 Previous studies show that the female to male ratio was very high. 17, 18 The current study demonstrated that in patients with LN, the men had more severe disease than women. This disagrees with studies of patients with chronic kidney disease, which show a more accelerated loss of kidney function and increased glomerular sclerosis in men compared to women.19
The demographic distribution of data from the present study showed that SLE was more common in older women aged > 45 years, and teenagers than in women in the fertile period (20–45 years), which may be linked to anxiety and psychological problems. This finding is consistent with research from Western Asia, Eastern Asia, and Northern Africa that shows that SLE disease can affect patient of all ages, but it most commonly affects those between the ages 20 and 45.20
In addition, variation in environmental exposures and nutrition may actually lead to an increase in older age groups. In the three cohorts studied (pediatric onset [less than 18 years old], adult-onset [18–50 years old], and delayed-onset [more than 50 years old]), the ThiQar-Iraq study found an elevated female to male ratio.21
Remarkably, the study conducted by Trentin et al.,22 the age group (20-45) years whether females or males was more affected statistically compare other age groups. This study found that females out-numbered males in LN patients, which is comparable with a local study in Baghdad,23 which found that females out-numbered males, with females 32(80%) and males 8(20) with ages ranging from 19 to 44 years.
These antibodies are limited to the mitochondria and nucleus, and DNases rapidly degrade them in the endosomes and cytoplasm. Hence, for this reason, these antibodies are not assessable to the immune system. The nucleus materials, including DNA, can be released after cell apoptosis due to exposure to drugs, infection, and ultraviolet light resulting in recognition by anti-DNA antibodies.24 Patients' serum levels of anti-dsDNA antibodies are higher than those of the control group. These findings suggest that the increase in anti-dsDNA antibody concentrations previous to SLE exacerbations may be the result of a controlled immune response or simply the result of polyclonal B-cell activation.25 In our study, 71% of cases with LN occurred within five years after getting a diagnosis of SLE. The development of LN may have been directly influenced by the immunological pathogenic processes that caused SLE initially. Late in the course of the diseases, LN can develop. Patients with late-onset LN did not differ in their disease profile or response to treatment, according to previous studies.26 Numerous studies have evaluated the relationship between anti-DNA antibodies and the renal activity (but not chronicity) index, as well as a possible association with WHO type IV (diffuse proliferative) glomerulonephritis.27 Proliferative lupus nephritis (PLN) is associated with elevated levels of C-reactive protein and anti-dsDNA antibody, and almost all of PLN patients have higher levels of anti-dsDNA antibody than controls at any one time (78% versus 5%).28 In LN, the immune system targets the kidneys, Genetic factors Certain genetic predispositions can increase the likelihood of developing these autoantibodies, Environmental triggers Factors such as UV-radiation, infections, and certain drugs can trigger the immune response that leads to increased production of ANAs and anti-dsDNA antibodies.29 Extensive research has established the involvement of anti-dsDNA antibodies in causing damage to the kidneys, skin, and brain in individuals with SLE. The production of these antibodies is influenced by various factors, including abnormalities in dendritic cells, B cells, or T cells, as well as a deficiency of a DNase enzyme responsible for clearing released nuclear materials. However, further studies are required to fully understand these mechanisms. The detrimental effects of penetrating autoantibodies in initiating complex inflammatory and fibrotic processes underscore the role of anti-DNA antibodies in harming target cells and organs,24 increased production of ANAs and anti-dsDNA contribute to LN by Direct binding to renal antigens. Anti-dsDNA antibodies can bind directly to antigens in the kidney or form complexes that deposit in the kidneys, leading to inflammation and damage, Cross-reactive antigens Some renal antigens may cross-react with anti-dsDNA antibodies, exacerbating kidney injury and Induction of inflammatory pathways the binding of these antibodies to renal tissues can activate inflammatory pathways, leading to further kidney damage, the anti-dsDNA antibody test is generally considered the gold standard in serology for identifying and assessing disease activity in patients with SLE.30 Loss of immunological tolerance and the generation of autoantibodies play a crucial part in the pathophysiology of SLE at the auto-antibody level. LN has been closely linked to anti-dsDNA auto-antibodies, and a variety of mechanisms supporting their nephritogenic potential have been suggested.31
The complement component works to degrade these complexes, perhaps most SLE patients have a deficiency in the complement component, and these antibodies will accumulate and appear higher in patients.32 Although auto-antibodies against C1r and C1s, which are less common and show no correlation, those against C1q have a significant correlate with the severity of the disease and immunological activity. Auto-antibodies against C1q are common in LN patients,33 and The classical pathway's complementary elements vary according on disease activity. Decreases in C1q and C3 levels have been linked to active renal disease; in contrast to other complement factor levels, C1q levels were found to be the best predictor of the later development of renal flare.34 Because the complement system plays an opsonizing role in the removal of auto-antigens and apoptotic bodies in physiological conditions, a malfunction in its functioning may result in a failure to identify and remove cell debris and auto-antigens in time, which can then trigger an immune response and generate auto-antibodies. Auto-antibodies sequentially link to complement components, which primarily act to damage tissue, particularly the kidney.35
LN is a serious complication of SLE in which multiple cytokines cause progression of the disease. Hence, early detection and treatment can significantly improve renal outcome. Identifying biomarker of renal involvement in SLE could replace renal biopsy, and avoid its complications.36 The immune response in LN involves the formation of immune complexes, activation of the complement system, release of proinflammatory cytokines and chemokine, infiltration of leukocytes, and the development of interstitial nephritis and fibrosis.37 The production of proinflammatory cytokines and chemokine, along with an increase in leukocytes, intensifies interstitial nephritis and promotes fibrosis in the kidneys.38 These inflammatory processes contribute to the progression of renal damage in LN.39 It is remains unknown if IL-35 suggests a role in renal association in LN. Compared to SLE patients without nephritis, LN patients had considerably lower serum levels of IL-35. According to earlier research, chemokines reduce inflammation by attracting T-cells to the target organ.40 Our findings were consistent with Li et al.,41 who show that human IL-35 is expressed in response to inflammatory stimuli but is not compatible with tissues. This suggests that human IL-35's function is to inhibit full-blown inflammation rather than to prevent inflammation from starting in the first place. indicating an immuno-regulatory function of IL-35 against an inflammatory environment. Our results, were consistent with those of Göschl et al.,42 who demonstrated that CD4+CD25-Foxp3+T cells are elevated in SLE patients with renal involvement and in urine sediment samples of patients with active glomerulo-nephritis, and that they are also linked to the degree of proteinuria. This is because Treg cells are a major source of IL-35. While He et al.43 found that elevated anti-dsDNA indicated inverse relationships with serum IL-35 levels in patients with SLE, our research found that LN patients with positive anti-dsDNA had considerably higher serum IL-35 than SLE patients without nephritis. Our findings were consistent with those of Qiu et al.,44,45 who proposed that elevated IL-35 levels may protect immune systems from pathogenic factors to prevent tissue and organ damage. This could be a compensatory but ineffective attempt to lessen the disease's inflammatory burden, which proinflammatory factors may antagonized.
The results explain that bacteria play an important role in increasing the concentration of interleukin 35 in patients with LN by stimulating the processing of molecular mimicry. These processes between a molecule from infection agents and LN-related autos have been proposed as mechanisms and how they lead to the breakdown of self-tolerance. In addition, tissues are injured by bacteria leading to damage to the kidney. Bacteria are also considered one of the causes of kidney failure Acute or chronic inflammation associated with glomerulonephritis may damage the kidneys. For this reason, we observe a higher level of interleukin 35 in patients with LN than in patients with renal failure, as patients with renal failure are constantly undergoing hemodialysis ranging from 2-3 times a week.
It was concluded from this study that immune-modulatory markers are good for predicting and diagnosing patients with lupus nephritis. The increase in interleukin-35 in patients with lupus nephritis compared with renal failure and healthy groups is considered a predictive sign for detecting the disease, in addition to the role of bacteria in raising interleukin 35 in the subgroup in both patients with lupus nephritis and renal failure compared to healthy groups.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Acknowledgement
We would like to thank all the field and lab teams (Al-Amin Research Center In Al-Najaf, Iraq) to help us provide all research needs.
8. Conflict of interest
The authors declare no conflicts of interest.
9. Authors contributions
All the authors contributed equally to this study, revised the paper, and approved the final version of the manuscript.
10. REFERENCES
Authors affiliations:
- Angham Najah Al-Khafaji, Kufa Technical Institute /Al-Furat Al-Awsat Technical University, Al-Najaf, Iraq; Email: kin.angh22@atu.edu.iq
- Zahraa Yosif Motaweq, Faculty of Science, Kufa University, Al-Najaf, Iraq; Email: Zahraa.mutawak@uokufa.edu.iq
ABATRACT
Background & Objective: Lupus Nephritis (LN) is considered a serious manifestation of SLE (SLE). which is diagnosed through Immunological biomarker. Interleukin-35 (IL-35), a member of the interleukin-12 family, has been identified as immunosuppressive and anti-inflammatory cytokine. In the past ten years, the role of IL-35 in inflammatory and autoimmune diseases has been studied extensively. We aimed to detect LN using immunological markers and to evaluate serum levels of iL-35 in LN patients with and without bacteria.
Methodology: Serum levels of iL-35 were measured in 120 adults, including 60 patients with LN, 30 patients with kidney failure and 30 from healthy volunteers using ELISA.
Results: Females were found to be highly infected with LN compared with males. LN was more prevalent at the age of 15-45 years and more highly infected with the disease compared with other ages. Anti-dsDNA and Anti-C1q antibodies are good Immunological markers to consider to detect LN patients. Levels of serum IL-35 were significantly higher (P < 0.05) in the LN group compared with those with kidney failure (KF Group) and healthy groups. IL-35 levels were significantly higher in LN patients - (mean 90.36 ± 20.02 pg/mL), than Kidney failure group (77.15 ± 25.7 pg/mL) and the Healthy group (70.76 ± 17.96 pg/mL) (P ≤ 0.001). The concentration of IL-35 was high in males infected and non-infected with bacteria as compared to females. The differences in the results of these two subgroups were statistically not significant at a mean probability of P ≤ 0.05.
Conclusion: It was concluded from the results of this study that the increase in interleukin-35 in patients with lupus nephritis compared with renal failure and healthy groups is considered a predictive sign for detecting the disease, in addition to the role of bacteria in raising interleukin 35 in the subgroups lupus nephritis and renal failure compared to the healthy groups.
Abbreviations: APC: antigen presenting cells, IL-35: Interlukein-35, SLE; SLE, LN: Lupus nephritis, ELISA: Enzyme Linked Immuno-Sorbent Assay, dsDNA: double stranded DNA, OD: optical density, ROC: Receiver operator characteristic
Keywords: Anti-dsDNA; Anti C1q; ELIZA; Interlukein-35; Lupus Nephritis; SLE
Citation: Al-Khafaji AN, Motaweq ZY. Evaluation and management of interlukein-35 (IL-35) in prognosis of patient with lupus nephritis. Anaesth. pain intensive care 2025;29(4):308-17. DOI: 10.35975/apic.v29i4.xxxx
Received: March 07, 2025; Revised: April 27, 2025; Accepted: April 27, 2025
1. INTRODUCTION
Lupus nephritis (LN) is a common complication of SLE (SLE). Mild sub-nephrotic proteinuria to diffuse progressive glomerulonephritis leading to chronic kidney disease are the results. LN usually manifests first in the course of SLE. Proteinuria, hematuria, edema of the lower extremities, increased creatinine levels and hypertension are the signs of LN. In order to diagnose LN and rule out alternative causes, a histopathological biopsy is essential. The six LN classes are revealed by the histopathology biopsy used in this investigation.1 T-cells play a major role in the pathogenesis of SLE, by promoting inflammation through the release of pro-inflammatory cytokines, inducing B-cells to produce auto-antibodies, and maintaining the disease through an accumulation of auto-reactive memory T-cells.
T-follicular helper-cells are essential for germinal center induction, proliferation, isotype-switching, and somatic hyper-mutation, as well as for direct infiltration and cytotoxicity to renal parenchymal tissues. However, the ratios of specific T-cell subsets and their function are aberrant in LN patients. The two main subpopulations of T-lymphocytes are CD4+ helper T-cells and CD8+ cytotoxic T-cells.2 Numerous mononuclear cells, CD4+ T-cells and CD8+ T-cells have been seen in the tubulointerstitial lesions in patients with LN.3
The most reliable method for identifying, categorizing and predicting LN is renal biopsy. However, it cannot be extensively used due to a number of disadvantages, such as it being an invasive procedure, bleeding risk, and potential of sample error.4 Furthermore, serial biopsies are not possible due to the invasive nature and the possibility of complications linked with the procedure.5
Anti-double stranded DNA (dsDNA) antibodies play an important role in the diagnosis, classification and management of SLE.6 High titers of anti-dsDNA antibodies are associated with the presence of SLE and are frequently used to follow the progress of LN.7
Interleukin-35 is a unique inhibitory cytokine that is part of the IL-12 cytokine family containing IL-12 subunit a chain (P35) subunits and Epstein-Barr virus-induced gene 3 (EBI3). It was discovered that, in contrast to other members of the IL-12 family, which are known to be mainly secreted by activated antigen presenting cells (APC), it is secreted by a variety of cell types and in a wide range of tissues, including monocytes, smooth muscle cells, endothelial cells, dendritic cells (DCs), and regulatory B-cells.
Numerous autoimmune diseases, inflammatory diseases, bacterial and viral infectious diseases, and tumors have also been shown to be significantly regulated by IL-35.8 Surprisingly, its biological functions differ from those of its siblings primarily in that it improves Treg-cell differentiation and immune suppression by transforming native T-cells into an IL-35 producing induced Treg-cell population known as iTr35,9 which directly inhibits effector T-cell proliferation in an APC-free culture in vitro.10
The present study aimed to estimate the levels of IL-35 in the serum of patients with and without bacterial infections. In addition, we sought to examine any possible variations between males and females at these levels. This research will help better understanding of how IL-35 functions in autoimmune nephritis and how gender affects these levels.
2. METHODOLOGY
A case control study design was used for this study. We collected 120 samples; e.g., 60 from LN patients, 30 from kidney failure patients (KF Group) and 30 healthy adults, with age range 11-70 years. Clinical samples were collected from Al-Sader Medical City in Al-Najaf Province from December 2023 to May 2024. The research was preapproved by the Institutional Higher Scientific and Ethical Committee. All participants gave their written informed permission.
2.1. Specimen Collection
Venous blood samples 15 mL were drawn from all LN patients and healthy groups. Blood of all samples, 4 mL was transmitted into vacuum gel tubes and left at room temperature for at least 30 min for clotting, then centrifuged at 4000 rpm for 10 min. The serum 0.5 mL was poured into Eppendrff tubes for estimating the levels of anti-dsDNA, anti-C1q, and IL-35, and frozen at -20°C until used.
2.2. Estimation of serum biomarker levels
Enzyme Linked Immuno-sorbent Assay (ELISA) kit (Mindray; Germany) uses Sandwich-ELISA as the technique. The Micro ELIZA strip plate provided in this kit has been pre-coated with an antibody specific to (anti-dsDNA, anti-C1q, IL-35). Added, standards or samples to the suitable Micro ELIZA strip plate wells, combines to the specific antibody. Then, added a Horseradish Peroxidase (HRP)-conjugated antibody specific for biomarker to each Micro ELIZA strip plate well and incubated. Free components are washed method. Added the substrate solution (TMB) to all well. Only those wells that contain biomarker and HRP-conjugated double stranded antibody will appear blue in color and then turn yellow after the addition of the stop solution. The optical density (OD) is measured with spectrophotometer (Mindray; Germany) at a wavelength of 450 nm. The value of OD is relative estimate to the concentration of biomarker. One can calculate the concentration of biomarker in the samples by comparing the OD of the samples to the standard curve, as demonstrated in Figures 1 (a, b and c).

Figure 1 (a): The standard curve of biomarker IL-35

Figure 1 (b): The standard curve of biomarker anti-C1q

Figure 1 (c): The standard curve of biomarker
anti-dsDNA.
2.3. Statistical Analysis
This is a case control study. Data were tested using statistical software IBM SPSS. The data were accessible as mean ± standard deviation. The Kolmogorov-Smirnov test for Normal distribution uses the normal distribution. Comparison of groups using Chi-square have been done in order to investigate the association of the categorical and numerical variables, respectively, ANOVA, ROC curve analysis, Pearson’s correlation coefficient (r) test was used to evaluate the degree of relationship between two sets of variables. P < 0.05 was considered significant.
3. RESULTS
A total of 120 samples were collected and divided into two groups, the patients group and the control group.
The patient group included 60 patients suffering from LN, diagnosed by a specialist physician, and with laboratory findings. While the control group was divided into two group (KF Group and Healthy adults).
The majority of LN patients were females; 45 (75%) vs. 15 (25%) males. The age of patients in LN group ranged between 11-70 years (Figure 2).

Figure 2: Distribution of LN patients according to age group
3.1. Immunological Marker to Detect LN
3.1.1. Anti-dsDNA antibody
The results of present study have illustrated a significant difference in the serum level of anti-dsDNA Ab in patients infected with LN, compare with control groups. The anti-dsDNA Ab level was elevated in the serum of LN patients; e.g., 10.47 ± 8.85 pg/mL versus 9.25 ± 4.3 pg/mL in KF Group and 4.37 ± 1.64 pg/mL in Healthy group (P = 0.001) as show in Figure 3.

Figure 3: Anti-dsDNA antibody levels in serum of patients with LN compared with Control Groups (P = 0.05 - Significant)
3.1.2. Anti-C1q Antibody
Figure 4 shows the serum levels of Anti-C1q antibody in LN group compared with control group. The results demonstrate that there was a significant decrease (P ≤ 0.002) in patients mean serum levels; e.g., 0.90 ± 0.27 pg/mL in LN group versus 0.94 ± 0.21 pg/mL in KF Group and 1.19 ± 0.5 pg/mL in Healthy patient group.

Figure 4: Anti-C1q antibody levels in serum of patients infect LN compared with Control Groups (P = 0.05 Significant)
3.1.3. Evaluation of serum IL-35 levels
The results showed that mean serum levels of IL-35 were significantly higher in LN group (85.40 ± 9.7 pg/mL),
compared to KF Group (67.60 ± 12.1 pg/mL) and Healthy group (62.80 ± 12.2 pg/mL). The differences were significant (P = 0.0001) as show in Figure 5.

Figure 5: Interleukin-35 levels of serum in LN patients compared with control groups (P = 0.05 Significant)
Being higher in LN cases, the IL-35 levels were analyzed using receiver operator characteristic curve (ROC) analysis in order to find cutoff value for IL-35 that can predict positive diagnosis of LN. The cutoff value of IL-35 was > 85.69. However, it had confidence interval (Cl) 0.86-0.98 % since the area under curve (AUC) was 0.923 despite that ROC test was highly significant (P= 0.0001). The sensitivity and specificity of IL-35 cutoff value were 77.5% and 96%, respectively as shown in Figure 6 and Table 1.

Figure 6: ROC curve analysis to identify the IL-35 to find the cutoff in LN patients versus control groups as a potential diagnostic marker
| Table 1: Characteristics of ROC curve in lupus nephritis and control groups | ||||||
| Parameter | Cut off | AUC | 95% (CI) | Sensitivity % | Specificity % | P-value |
| IL-35 | 85.69 | 0.923 | 0.86-0.98 | 77.5 | 96 | < 0.0001 |
| AUC: area under curve; CI: confidence interval; values where significant at P ≤ 0.05 | ||||||
Present study revealed that the concentration of IL-35 was high in males with or without bacterial infections compared to females. The mean of females and males with LN was 90.2 ± 1pg/mL and 86.7 ± 2.2pg/mL respectively. In KF Group mean values were 75.2 ± 17.5 pg/mL and 70.9 ± 8.4 pg/mL, and in healthy groups the mean value was 64.3 ± 13.2 pg/mL and 62.4 ± 11.4 pg/mL for females and males respectively. The values for female LN patients without bacterial infect compared with those with bacterial infection were 76.8 ± 2.4 pg/mL, KF Group 62.1 ± 6.1 pg/mL and healthy groups 58.8 ± 0.00 pg/mL. while mean of males to LN was 74.0 ± 2.1 pg/mL, KF Group 59.2 ± 7.4 pg/mL, and healthy groups 48.5 ± 0.00 pg/mL. The differences in the results of these two subgroups were statistically not significant (P ≤ 0.05) as given in Table 2.
| Table 2: Levels of IL-35 in patient with lupus nephritis compared with control groups (with & without bacterial infection) according to gender | |||||
| IL-35 | Gender | Lupus Nephritis (pg/mL) | KF Group (pg/mL) | Healthy groups (pg/mL) | P-value |
| With bacterial infection | Female | 90.2 ± 1 | 75.2 ± 17.5 | 64.3 ± 13.2 | 0.0001 |
| Male | 86.7 ± 2.2 | 70.9 ± 8.4 | 62.4 ± 11.4 | 0.001 | |
| Without bacterial infection | Female | 76.8 ± 2.4 | 62.1 ± 6.1 | 58.8 ± 0.00 | 0.001 |
| Male | 74.0 ± 2.1 | 59.2 ± 7.4 | 48.5 ± 0.00 | 0.001 | |
| Data presented as Mean ± SD; P < 0.05 considered as significant | |||||
3.1.4. Correlations between parameters in LN patients
The correlations between three immunological parameters have been deducted. The results of the present study demonstrated that there were positive correlations between all parameter in patients suffering from LN.
- There was a highly positive correlation between anti-dsDNA antibody and anti-C1q antibody, anti-dsDNA antibody and IL-35, of LN patients r = 0.623** and P = 0.000018, r=0.619** and P = 0.00002, , as shown in Table 3 and Figures 7 and 8 respectively.
| Table 3: Correlation between parameter in lupus nephritis patients | ||
| Parameter | Pearson Correlation (r) | P-value |
| Anti-dsDNA , Anti-C1q | 0.623** | 0.000018 |
| Anti-dsDNA, IL-35 | 0.619** | 0.00002 |
| Anti-C1q, IL-35 | 0.597** | 0.00005 |
| r: person correlation coefficient; ** Correlation is significant at P < 0.01 | ||

Figure 7: Correlation between concentration anti-dsDNA and anti-C1q antibody levels in LN Patients

Figure 8: Correlation between concentration of anti-dsDNA antibody and IL-35 Levels in LN patients
- A strong positive correlation was observed between anti-C1q antibody and IL-35; the r = 0.597** and P = 0.00005, as depicted in Table 3 and Figure 9.
| Table 3: Correlation between parameter in lupus nephritis patients | ||
| Parameter | Pearson Correlation (r) | P-value |
| Anti-dsDNA , Anti-C1q | 0.623** | 0.000018 |
| Anti-dsDNA, IL-35 | 0.619** | 0.00002 |
| Anti-C1q, IL-35 | 0.597** | 0.00005 |
| r: person correlation coefficient; ** Correlation is significant at P < 0.01 | ||

Figure 9: Correlation between concentration of anti-C1q antibody and IL-35 Levels in LN patients
4. DISCUSSION
The present study explains that the females are more affected than males, because of sex hormones, and X-chromosome genes in females with high effect and sensitivity to autoimmune disease.
According to studies by Moulton VR.,11 females are more likely to develop autoimmune inflammatory diseases than the males, and the estrogen increases severity of these diseases. The immune system is impacted by this condition, which is linked to steroid hormones, particularly progesterone and estrogen. Lower progesterone levels in lupus-affected women, indicates that a relative difference for estrogen may be an indicator that influences certain female patients' to immunological reactivity.11 Woo et al,12 suggested that the incidence of SLE in females was 100%. This could be because of the possible roles of gender-based differences in environmental exposures or sensitivity to these exposures, as well as genetic variations on the X chromosome and sex hormone milieus, as risk factors for incident SLE. High female to male ratio indicates that gender variables influence the development and severity of the disease.13 Depending on the relationship between the age and other factors implicated in the pathogenesis of SLE, the ratio of female to male in this condition might range from 9:1.14 A previous study demonstrated that the percentage of females (57.4 %) with LN is slightly higher than males (42.6 %).15 These findings could be explained by hormonal differences between the genders. Estradiol acts through its receptors and contributes to the sexual orientation in SLE. Most of the studies in the world revealed wide range of female to male differences.16 Previous studies show that the female to male ratio was very high. 17, 18 The current study demonstrated that in patients with LN, the men had more severe disease than women. This disagrees with studies of patients with chronic kidney disease, which show a more accelerated loss of kidney function and increased glomerular sclerosis in men compared to women.19
The demographic distribution of data from the present study showed that SLE was more common in older women aged > 45 years, and teenagers than in women in the fertile period (20–45 years), which may be linked to anxiety and psychological problems. This finding is consistent with research from Western Asia, Eastern Asia, and Northern Africa that shows that SLE disease can affect patient of all ages, but it most commonly affects those between the ages 20 and 45.20
In addition, variation in environmental exposures and nutrition may actually lead to an increase in older age groups. In the three cohorts studied (pediatric onset [less than 18 years old], adult-onset [18–50 years old], and delayed-onset [more than 50 years old]), the ThiQar-Iraq study found an elevated female to male ratio.21
Remarkably, the study conducted by Trentin et al.,22 the age group (20-45) years whether females or males was more affected statistically compare other age groups. This study found that females out-numbered males in LN patients, which is comparable with a local study in Baghdad,23 which found that females out-numbered males, with females 32(80%) and males 8(20) with ages ranging from 19 to 44 years.
These antibodies are limited to the mitochondria and nucleus, and DNases rapidly degrade them in the endosomes and cytoplasm. Hence, for this reason, these antibodies are not assessable to the immune system. The nucleus materials, including DNA, can be released after cell apoptosis due to exposure to drugs, infection, and ultraviolet light resulting in recognition by anti-DNA antibodies.24 Patients' serum levels of anti-dsDNA antibodies are higher than those of the control group. These findings suggest that the increase in anti-dsDNA antibody concentrations previous to SLE exacerbations may be the result of a controlled immune response or simply the result of polyclonal B-cell activation.25 In our study, 71% of cases with LN occurred within five years after getting a diagnosis of SLE. The development of LN may have been directly influenced by the immunological pathogenic processes that caused SLE initially. Late in the course of the diseases, LN can develop. Patients with late-onset LN did not differ in their disease profile or response to treatment, according to previous studies.26 Numerous studies have evaluated the relationship between anti-DNA antibodies and the renal activity (but not chronicity) index, as well as a possible association with WHO type IV (diffuse proliferative) glomerulonephritis.27 Proliferative lupus nephritis (PLN) is associated with elevated levels of C-reactive protein and anti-dsDNA antibody, and almost all of PLN patients have higher levels of anti-dsDNA antibody than controls at any one time (78% versus 5%).28 In LN, the immune system targets the kidneys, Genetic factors Certain genetic predispositions can increase the likelihood of developing these autoantibodies, Environmental triggers Factors such as UV-radiation, infections, and certain drugs can trigger the immune response that leads to increased production of ANAs and anti-dsDNA antibodies.29 Extensive research has established the involvement of anti-dsDNA antibodies in causing damage to the kidneys, skin, and brain in individuals with SLE. The production of these antibodies is influenced by various factors, including abnormalities in dendritic cells, B cells, or T cells, as well as a deficiency of a DNase enzyme responsible for clearing released nuclear materials. However, further studies are required to fully understand these mechanisms. The detrimental effects of penetrating autoantibodies in initiating complex inflammatory and fibrotic processes underscore the role of anti-DNA antibodies in harming target cells and organs,24 increased production of ANAs and anti-dsDNA contribute to LN by Direct binding to renal antigens. Anti-dsDNA antibodies can bind directly to antigens in the kidney or form complexes that deposit in the kidneys, leading to inflammation and damage, Cross-reactive antigens Some renal antigens may cross-react with anti-dsDNA antibodies, exacerbating kidney injury and Induction of inflammatory pathways the binding of these antibodies to renal tissues can activate inflammatory pathways, leading to further kidney damage, the anti-dsDNA antibody test is generally considered the gold standard in serology for identifying and assessing disease activity in patients with SLE.30 Loss of immunological tolerance and the generation of autoantibodies play a crucial part in the pathophysiology of SLE at the auto-antibody level. LN has been closely linked to anti-dsDNA auto-antibodies, and a variety of mechanisms supporting their nephritogenic potential have been suggested.31
The complement component works to degrade these complexes, perhaps most SLE patients have a deficiency in the complement component, and these antibodies will accumulate and appear higher in patients.32 Although auto-antibodies against C1r and C1s, which are less common and show no correlation, those against C1q have a significant correlate with the severity of the disease and immunological activity. Auto-antibodies against C1q are common in LN patients,33 and The classical pathway's complementary elements vary according on disease activity. Decreases in C1q and C3 levels have been linked to active renal disease; in contrast to other complement factor levels, C1q levels were found to be the best predictor of the later development of renal flare.34 Because the complement system plays an opsonizing role in the removal of auto-antigens and apoptotic bodies in physiological conditions, a malfunction in its functioning may result in a failure to identify and remove cell debris and auto-antigens in time, which can then trigger an immune response and generate auto-antibodies. Auto-antibodies sequentially link to complement components, which primarily act to damage tissue, particularly the kidney.35
LN is a serious complication of SLE in which multiple cytokines cause progression of the disease. Hence, early detection and treatment can significantly improve renal outcome. Identifying biomarker of renal involvement in SLE could replace renal biopsy, and avoid its complications.36 The immune response in LN involves the formation of immune complexes, activation of the complement system, release of proinflammatory cytokines and chemokine, infiltration of leukocytes, and the development of interstitial nephritis and fibrosis.37 The production of proinflammatory cytokines and chemokine, along with an increase in leukocytes, intensifies interstitial nephritis and promotes fibrosis in the kidneys.38 These inflammatory processes contribute to the progression of renal damage in LN.39 It is remains unknown if IL-35 suggests a role in renal association in LN. Compared to SLE patients without nephritis, LN patients had considerably lower serum levels of IL-35. According to earlier research, chemokines reduce inflammation by attracting T-cells to the target organ.40 Our findings were consistent with Li et al.,41 who show that human IL-35 is expressed in response to inflammatory stimuli but is not compatible with tissues. This suggests that human IL-35's function is to inhibit full-blown inflammation rather than to prevent inflammation from starting in the first place. indicating an immuno-regulatory function of IL-35 against an inflammatory environment. Our results, were consistent with those of Göschl et al.,42 who demonstrated that CD4+CD25-Foxp3+T cells are elevated in SLE patients with renal involvement and in urine sediment samples of patients with active glomerulo-nephritis, and that they are also linked to the degree of proteinuria. This is because Treg cells are a major source of IL-35. While He et al.43 found that elevated anti-dsDNA indicated inverse relationships with serum IL-35 levels in patients with SLE, our research found that LN patients with positive anti-dsDNA had considerably higher serum IL-35 than SLE patients without nephritis. Our findings were consistent with those of Qiu et al.,44,45 who proposed that elevated IL-35 levels may protect immune systems from pathogenic factors to prevent tissue and organ damage. This could be a compensatory but ineffective attempt to lessen the disease's inflammatory burden, which proinflammatory factors may antagonized.
The results explain that bacteria play an important role in increasing the concentration of interleukin 35 in patients with LN by stimulating the processing of molecular mimicry. These processes between a molecule from infection agents and LN-related autos have been proposed as mechanisms and how they lead to the breakdown of self-tolerance. In addition, tissues are injured by bacteria leading to damage to the kidney. Bacteria are also considered one of the causes of kidney failure Acute or chronic inflammation associated with glomerulonephritis may damage the kidneys. For this reason, we observe a higher level of interleukin 35 in patients with LN than in patients with renal failure, as patients with renal failure are constantly undergoing hemodialysis ranging from 2-3 times a week.
5. CONCLUSION
It was concluded from this study that immune-modulatory markers are good for predicting and diagnosing patients with lupus nephritis. The increase in interleukin-35 in patients with lupus nephritis compared with renal failure and healthy groups is considered a predictive sign for detecting the disease, in addition to the role of bacteria in raising interleukin 35 in the subgroup in both patients with lupus nephritis and renal failure compared to healthy groups.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Acknowledgement
We would like to thank all the field and lab teams (Al-Amin Research Center In Al-Najaf, Iraq) to help us provide all research needs.
8. Conflict of interest
The authors declare no conflicts of interest.
9. Authors contributions
All the authors contributed equally to this study, revised the paper, and approved the final version of the manuscript.
10. REFERENCES
- Zaman GS. Introduction and physiology of lupus. In: IntechOpen; 2017;5:31. DOI: 5772/intechopen.68635
- Nakayamada S, Tanaka Y. Clinical relevance of T follicular helper cells in SLE. Expert Rev Clin Immunol. 2021;17(10):1143–50. PubMed DOI: 1080/1744666X.2021.1976146
- Stanley S, Mok CC, Vanarsa K, Habazi D, Li J, Pedroza C, et al. Identification of low-abundance urinary biomarkers in lupus nephritis using electrochemiluminescence immunoassays. Arthritis Rheumatol. 2019;71(5):744–55. PubMed DOI: 1002/art.40813
- Caster DJ, Merchant ML, Klein JB, Powell DW. Precision medicine in lupus nephritis: can biomarkers get us there? Transl Res. 2018;201:26–39. PubMed DOI: 1016/j.trsl.2018.08.002
- Hassan LA, Majeed AA, Darweesh MF. The role of IL-12 in the aetiology of SLE and its connection to HBV infection in Iraqi patients. J Commun Dis. 2022;54(1):41–46. DOI: 24321/0019.5138.202248
- Admou B, Eddehbi FE, Elmoumou L, Elmojadili S, Salami A, Oujidi M, et al. Anti-double stranded DNA antibodies: a rational diagnostic approach in limited-resource settings. Pract Lab Med. 2022;31(6):e00285. PubMed DOI: 1016/j.plabm.2022.e00285
- Su LC, Liu XY, Huang AF, Xu WD. Emerging role of IL-35 in inflammatory autoimmune diseases. Autoimmun Rev. 2018;17(7):665–73. PubMed DOI: 1016/j.autrev.2018.01.017
- Collison LW, Delgoffe GM, Guy CS, Vignali KM, Chaturvedi V, Fairweather D, et al. The composition and signaling of the IL-35 receptor are unconventional. Nat Immunol. 2012;13(3):290–9. PubMed DOI: 1038/ni.2227
- Collison LW, Chaturvedi V, Henderson AL, Giacomin PR, Guy C, Bankoti J, et al. IL-35-mediated induction of a potent regulatory T cell population. Nat Immunol. 2010;11(12):1093–101. PubMed DOI: 1038/ni.1952
- Cutolo M, Straub RH. Sex steroids and autoimmune rheumatic diseases: state of the art. Nat Rev Rheumatol. 2020;16(11):628–44. PubMed DOI: 1038/s41584-020-0503-4
- Moulton VR. Sex hormones in acquired immunity and autoimmune disease. Front Immunol. 2018;9:2279. PubMed DOI: 3389/fimmu.2018.02279
- Woo JM, Parks CG, Jacobsen S, Costenbader KH, Bernatsky S. The role of environmental exposures and gene–environment interactions in the etiology of systemic lupus erythematosus. J Intern Med. 2022;291(6):755–78. PubMed DOI: 1111/joim.13448
- Lasrado N, Jia T, Massilamany C, Franco R, Illes Z, Reddy J. Mechanisms of sex hormones in autoimmunity: focus on EAE. Biol Sex Differ. 2020;11(12):1–4. PubMed DOI: 1186/s13293-020-00325-4
- Sakkas LI, Chikanza IC. Sex bias in immune response: it is time to include the sex variable in studies of autoimmune rheumatic diseases. Rheumatol Int. 2024;44(2):203–9. PubMed DOI: 1007/s00296-023-05446-8
- Constantin AM, Baicus C. Estradiol in systemic lupus erythematosus. Acta Endocrinol (Buchar). 2023;19(2):274. PubMed DOI: 4183/aeb.2023.274
- Margery-Muir AA, Bundell C, Nelson D, Groth DM, Wetherall JD. Gender balance in patients with systemic lupus erythematosus. Autoimmun Rev. 2017;16(3):258–68. PubMed DOI: 1016/j.autrev.2017.01.007
- Kosałka-Węgiel J, Dziedzic R, Siwiec-Koźlik A, Spałkowska M, Milewski M, Żuk-Kuwik J, et al. Clinical and laboratory characteristics of early-onset and delayed-onset lupus nephritis patients: a single-center retrospective study. Rheumatol Int. 2024;44(7):1283–94. PubMed DOI: 1007/s00296-024-05579-4
- Al-Yasiri DKH, Darweesh MF. The role of miR-21 that related with IL-12 and INF-γ as diagnostic biomarker among patients with lupus nephritis and type 1 diabetes disease. Univ Kufa Fac Sci Theses. 2023.
- Mahmood SB, Aziz M, Malepati D, Lee-Smith W, Clark J, Brearley A, et al. Evaluating sex differences in the characteristics and outcomes of lupus nephritis: a systematic review and meta-analysis. Glomerul Dis. 2024;4(1):19–32. PubMed DOI: 1159/000535981
- Rathoon AI, Gurusamy V, Ganesan V, Arivazhagan S, Yaswanth C. A long-term follow-up study of lupus nephritis in a single tertiary care centre. Indian J Nephrol. 2022;32(6):595–9. PubMed DOI: 4103/ijn.ijn_501_21
- Sabr NH, Obaid KA, Nisan AH. Hepatomegaly, splenomegaly and lymphadenopathy in Iraqi patients with systemic lupus erythematosus. Univ Thi-Qar J Med. 2020;19(1):1. DOI: 32792/jmed.v19i1.100
- Trentin F, Signorini V, Manca ML, Cascarano G, Gualtieri L, Schilirò D, et al. Gender differences in SLE: report from a cohort of 417 Caucasian patients. Lupus Sci Med. 2023;10(1):e000880. PubMed DOI: 1136/lupus-2022-000880
- Ibrahim NA, Allawi AA, Ghudhaib KK, Hammoudi FA. Estimation of some immunological markers of Iraqi patients in systemic lupus erythematosus with lupus nephritis. Medico Legal Update. 2020;20(4) 643-9,. DOI: 37506/mlu.v20i4.1892
- Wang X, Xia Y. Anti-double stranded DNA antibodies: origin, pathogenicity, and targeted therapies. Front Immunol. 2019;10:1667. PubMed DOI: 3389/fimmu.2019.01667
- ter Borg EJ, Horst G, Hummel E, Limburg PC, Kallenberg CG. Rises in anti-double stranded DNA antibody levels prior to exacerbations of systemic lupus erythematosus are not merely due to polyclonal B cell activation. Clin Immunol Immunopathol. 1991;59(1):117–28. PubMed DOI: 1016/0090-1229(91)90086-p
- Delfino J, Santos TA, Skare TL. Comparison of lupus patients with early and late onset nephritis: a study in 71 patients from a single referral center. Adv Rheumatol. 2020;60(1):5. PubMed DOI: 1186/s42358-019-0105-5
- Okamura M, Kanayama Y, Amastu K, Negoro N, Kohda S, Takeda T, et al. Significance of ELISA for antibodies to double stranded and single stranded DNA in lupus nephritis: correlation with severity of renal histology. Ann Rheum Dis. 1993;52(1):14–20. PubMed DOI: 1136/ard.52.1.14
- Olson SW, Lee JJ, Prince LK, Baker TP, Papadopoulos P, Edison J, et al. Elevated subclinical double-stranded DNA antibodies and future proliferative lupus nephritis. Clin J Am Soc Nephrol. 2013;8(10):1702–8. PubMed DOI: 2215/CJN.01910213
- Fu SM, Sung SS, Wang H, Zhao Z, Gaskin F. Mechanisms of renal damage in systemic lupus erythematosus. In: Lahita RG, editor. Systemic Lupus Erythematosus. Academic Press; 2021. p. 313–24. DOI: 1016/B978-0-12-801917-7.00033-4
- Choi SE, Park DJ, Kang JH, Lee SS. Significance of co-positivity for anti-dsDNA, -nucleosome, and -histone antibodies in patients with lupus nephritis. Ann Med. 2023;55(1):1009–17. PubMed DOI: 1080/07853890.2023.2187076
- Shang X, Ren L, Sun G, Yu T, Yao Y, Wang L, et al. Anti-dsDNA, anti-nucleosome, anti-C1q, and anti-histone antibodies as markers of active lupus nephritis and systemic lupus erythematosus disease activity. Immun Inflamm Dis. 2021;9(2):407–18. PubMed DOI: 1002/iid3.401
- Torres-Ruiz J, Barrera-Vargas A, Ortiz-Hernandez R, Alcocer-Varela J, Ponce-de-León A, Gómez-Martín D. Microbiological and immunological profile of patients with severe lupus flares related to bloodstream infections: a retrospective cohort study. Lupus. 2018;27(2):312–8. PubMed DOI: 1177/0961203317720527
- Skopelja-Gardner S, Colonna L, Hermanson P, Sun X, Tanaka L, Tai J, et al. Complement deficiencies result in surrogate pathways of complement activation in novel polygenic lupus-like models of kidney injury. J Immunol. 2020;204(10):2627–40. PubMed DOI: 4049/jimmunol.1901473
- Yin Y, Wu X, Shan G, Zhang X. Diagnostic value of serum anti-C1q antibodies in patients with lupus nephritis: a meta-analysis. Lupus. 2012;21(10):1088–97. PubMed DOI: 1177/0961203312451202
- Abdulla NY, Motaweq ZY, Alrufaie ZMM, Zghair LS. Phenotypic and genotypic study of biofilm formation in multidrug-resistant bacteria isolated from urinary tract infection from diabetes patients. AIP Conf Proc. 2024;3092(1):020006. DOI: 1063/5.0199864
- El-Hewala A, Nageeb GS, El-Shahawy EE, Sharaf DM, Omran AA, El-Messallamy FA, et al. Anti-C1q and anti-dsDNA antibodies in systemic lupus erythematosus: relationship with disease activity and renal involvement in Sharkia governorate, Egypt. Egypt Rheumatol. 2011;33(4):203–8. DOI: 1016/j.ejr.2011.07.005
- Zickert A, Amoudruz P, Sundström Y, Rönnelid J, Malmström V, Gunnarsson I. IL-17 and IL-23 in lupus nephritis: association with histopathology and response to treatment. BMC Immunol. 2015;16(1):1. PubMed DOI: 1186/s12865-015-0070-7
- Maria NI, Davidson A. Protecting the kidney in systemic lupus erythematosus: from diagnosis to therapy. Nat Rev Rheumatol. 2020;16(5):255–67. PubMed DOI: 1038/s41584-020-0401-9
- Yuan Q, Tang B, Zhang C. Signaling pathways of chronic kidney diseases, implications for therapeutics. Signal Transduct Target Ther. 2022;7(1):182. PubMed DOI: 1038/s41392-022-01036-5
- Li X, Mai J, Virtue A, Yin Y, Gong R, Sha X, et al. IL-35 is a novel responsive anti-inflammatory cytokine: a new system of categorizing anti-inflammatory cytokines. PLoS One. 2012;7(3):e33628. PubMed DOI: 1371/journal.pone.0033628
- Ye H, Cao P, Lin J, Yang X, Guo Q, Mao H, et al. Long-term clinical outcomes of lupus nephritis patients undergoing peritoneal dialysis: a matched, case-control study. Perit Dial Int. 2019;39(6):570–3. PubMed DOI: 3747/pdi.2019.00041
- Göschl L, Scheinecker C, Bonelli M. Treg cells in autoimmunity: from identification to Treg-based therapies. Semin Immunopathol. 2019;41(3):301–14. PubMed DOI: 1007/s00281-019-00741-8
- He D, Liu M, Liu B. Interleukin-35 as a new biomarker of renal involvement in lupus nephritis patients. Tohoku J Exp Med. 2018;244(4):263–70. PubMed DOI: 1620/tjem.244.263
- Qiu F, Song L, Yang N, Li X. Glucocorticoid downregulates expression of IL-12 family cytokines in systemic lupus erythematosus patients. Lupus. 2013;22(10):1011–6. PubMed DOI: 1177/0961203313498799
- Majeed HT, Motaweq ZY. Phylogenetic group of Escherichia coli isolated from inflammatory bowel disease in Al Najaf province. Egypt J Med Microbiol. 2024;33(4):129–34. DOI: 21608/ejmm.2024.307468.1293