Amr Kamal Abdelhakim 1, Dina Samir Mahmoud Shaker 2, Waleed Ibrahim Hamimy 3, Maha Abdelbar Attalla 4, Khalda Galal Radwan 5, Ahmed Kareem Mohammed Abdelhady 6
Authors affiliations:
Background & objectives: Several studies have shown that melatonin and vitamin C used as an analgesic adjuvant, can spare opioid use, resulting in a better analgesic profile with fewer adverse effects and shorter hospital stay. However, we found no studies which might have investigated the impact of a combination of both. We compared the effect of melatonin, vitamin C, and their combination on postoperative opioid consumption.
Methodology: Sixty patients scheduled for major abdominal surgeries were randomly allocated into three equal groups with 20 patients in each; melatonin group (Group M), vitamin C (Group C), and combined melatonin and vitamin C (Group CM). Two hours before surgery, all patients received the study medications orally, which was continued for 3 days postoperative at the same time of the first administration; Group M received 10 mg of melatonin, Group C received 1 gm of vitamin C, and Group MV received both melatonin (10 mg) and vitamin C (1 gm). The primary outcome was the total morphine consumption in 24 hours postoperatively, with patients, pain scores as measured by Numeric Pain Rating Scale (NPRS), the incidence of postoperative nausea and vomiting (PONV), and postoperative chronic pain as secondary outcomes.
Results: Postoperative morphine consumption (in the first 24 hours) was significantly lower in Group CM (16.7 ± 2.4 mg) than in Group M (20.98 ± 1.38 mg), and Group C (24.36 ± 3.12 mg) (P < 0.001). Group CM showed lower pain scores, decreased incidence of PONV, and a longer time to first request for analgesics. However, lower sedation scores were observed. There was no statistical difference among all groups regarding post-operative chronic pain incidence.
Conclusion: Combined use of melatonin and vitamin C in patients undergoing major abdominal surgery with mid-line incision provides a synergistic analgesic effect for the postoperative pain management with lower postoperative pain scores, less opioid consumption, and lower incidence of PONV compared to the use of any one of these.
Abbreviations: CPSP: Chronic Post-Surgical Pain, NPRS: Numeric Pain Rating Scale, PONV: postoperative nausea and vomiting, TAHBSO: Total Abdominal Hysterectomy with Bilateral Salpingo-Oophorectomy
Keywords: Melatonin; major abdominal surgery; Numeric Pain Rating Scale; opioid consumption; postoperative pain; PONV; vitamin C.
Citation: Abdelhakim AK, Shaker DSM, Hamimy WI, Attalla MA, Radwan KG, Abdelhady AKM. Anaesth. pain intensive care 2025;29(3):556-664. DOI: 10.35975/apic.v29i3.2790
Received: January 03, 2025; Revised: March 12, 2025; Accepted: March 22, 2025
Major abdominal surgeries cause changes in neuro-hormonal response, resulting in postoperative pain, various organ dysfunction, and prolonged hospitalization. Inadequate pain relief after major abdominal surgeries adversely affects the surgical outcome through slower rehabilitation and a more extended hospital stay.1
Chronic Post-Surgical Pain (CPSP) is defined as a persistent pain of duration longer than 2 months after the surgical procedure, excluding other causes of pain such as malignancy, infection, and pre-existing pain problems. Many procedures are likely to be associated with CPSP if the postoperative pain is not adequately controlled.
Major abdominal surgeries in previous studies have reported CPSP with an incidence ranging from 17% to 32%.2
Post-operative pain after major abdominal surgery is usually controlled through multimodal analgesia, which may be in the form of intravenous opioids, non-steroidal anti-inflammatory drugs, and paracetamol, together with regional blocks.3
There are many side effects of opioids, including sedation, dizziness, nausea, vomiting, constipation, physical dependence, tolerance, and respiratory depression.4
Therefore, many studies have been conducted to achieve effective postoperative pain control and improve patient satisfaction with minimal hazards. Several studies showed that the use of different adjuvants could spare opioid use, resulting in better rehabilitation, surgical outcomes, and less hospital stay.5
One of these adjuvants is melatonin. Melatonin (N-acetyl-5-methoxytryptamine) is an endogenous hormone the pineal gland produces and is essential to the circadian rhythm. Melatonin has proved to have a postoperative analgesic effect and to reduce opioid consumption. Administration of melatonin has dose-dependent long-term anti-nociceptive effects in acute, neuropathic, and inflammatory pain.6
Vitamin C is a water-soluble vitamin responsible for normal growth and development. It also has antioxidant, neuro-protective, and neuro-modulation effects. Vitamin C has been proven to reduce acute pain and the prevalence of complex regional pain syndromes with its anti-nociceptive effect.7 Certain studies have demonstrated that the combination of vitamin C with an opioid may diminish both opioid consumption for pain relief and the rate of opioid-related adverse effects.8
Several multimodal approaches have been advocated to reduce opioid consumption by using either melatonin or vitamin C.9 This is the first study conducted on using the combination of both in reducing opioid consumption in major abdominal surgeries.
This randomized, double-blinded, controlled study was conducted in the general surgery, uro-surgery, and gynecology operating rooms of Kasr Al Ainy Hospital, Cairo, Egypt, from January 2023 to July 2024. Ethical approval of the Institutional Ethics Committee (No. MD-15-2022) was obtained according to the ethical standards of the Declaration of Helsinki. The trial was registered at ClinicalTrials.gov (No. NCT06374771).
We enrolled 60 patients aged between 18 and 60 y, ASA- I–II, and scheduled for major abdominal surgeries with mid-line incisions, e.g., radical prostatectomy, nephrectomy, Total Abdominal Hysterectomy with Bilateral Salpingo-Oophorectomy (TAHBSO), hepatectomy, splenectomy, hepatic hydatid cyst excision. Informed signed consent was obtained from every participant. Those who refused to participate, were known allergic to the drugs used in the study, had a hepatic failure (Child class B- Child class C) or a renal impairment (creatinine >2mg/dL or chronic kidney disease on regular dialysis), or had a history of psychiatric or mental disorders, Chronic pain syndromes (neuropathic pain, post-herpetic neuralgia, Complex regional pain syndrome), or drug or alcohol abuse, were excluded.
Randomization was performed using computer-generated numbers with Random Allocation Software (https://random-allocation-software.software.informer.com/2.0/) in a 1:1:1 ratio. Sixty consequentially numbered opaque envelopes (twenty per group) containing group assignment and drug administration instructions were made. A research assistant was responsible for opening the envelopes and administering the assigned drug with no further involvement in the study. A blinded anesthesiologist was responsible for the anesthetic management, perioperative data collection, postoperative pain assessment, and analgesia administration.
Patients were allocated into three groups: the melatonin group (Group M), the vitamin C group (Group C), and the Melatonin and vitamin C group (Group CM).
The primary outcome was the total morphine consumption in the first 24 postoperative hours. Other outcomes included intraoperative fentanyl consumption, intraoperative and postoperative blood pressure (BP) and heart rate (HR), postoperative Richmond Agitation Sedation Scale (RASS), the time to first analgesic requirement, defined as the first recognized time point of Numeric Rating Scale (NRS) > 4 assessed during the first 24 h, the number of morphine boluses given after discharge from PACU, the frequency of postoperative chronic pain, and any adverse events.
2.1. Management Protocol
The day before surgery, participants were evaluated by acquiring a thorough medical history, physical examination, and investigations. Two hours before surgery, all patients will receive the study drugs orally: 10mg of melatonin for Group M (Melatonin 10 mg - Puritan’s Pride premium company), 1 gm of vitamin C for Group C (Sanso C 1000 mg - AUG pharma company), and 10mg of melatonin and 1 gm of vitamin C for Group CM.
The patients were transferred to the preparation room one hour before the procedure, and the NRS pain score was explained to all candidates (zero corresponds to no pain, and 10 indicates the worst unbearable pain). Baseline vital signs were recorded, including noninvasive measuring of systolic, mean, diastolic arterial blood pressures, heart rate, and oxygen saturation.
2.2. Anesthetic technique
Standard monitoring (non-invasive blood pressure, electrocardiography, and pulse oximetry) was applied upon arrival at the operating room. The patient received pre-induction ondansetron 8mg. Following pre-oxygenation via a face mask for 3 min, anesthesia was induced using propofol 1-2 mg/kg and fentanyl 2 µg/kg. Atracurium 0.5 mg/kg was administered to facilitate endotracheal intubation. After intubation, the capnography will be connected, and the patients will be mechanically ventilated to maintain end-tidal CO2 between 30-35 mmHg.
Anaesthesia was maintained with sevoflurane in 50% oxygen, and atracurium 0.1mg/kg every 20 min was administered to maintain muscle relaxation. After the skin incision, a bolus dose of morphine 100 mcg/kg was given. Fentanyl at 1µg/kg was administered as rescue analgesia if an intraoperative increase in heart rate or systolic blood pressure by 20% occurred after the exclusion of other causes rather than pain. Before skin closure, paracetamol 1gm was given intravenously. A bispectral index monitoring device was used to monitor the anesthesia depth. Readings within 40 to 55 were targeted throughout the procedure. Mean blood pressure and heart rate were maintained within ±20%.
2.3. Postoperative Management
Recovery time was recorded, defined as the time from extubation to the time of achieving Aldrete scores ≥ 9. The patient was transferred to the post-anesthesia care unit (PACU); Blood pressure, heart rate, and oxygen saturation were monitored every 15 min till discharge from the PACU. In PACU, 50mcg/kg of morphine was given by the physician to all patients. Then, all patients received a continuous morphine infusion of 5mcg/kg/hour through a disposable infusion pump 275ml with a constant flow of 5ml/hour (Zhejiang Fert Medical Device Co., Ltd). The patients were instructed to report any pain to a pain nurse, and if the NRS increased to more than 4, the pain nurse informed the anesthesiologist. Inadequate analgesia (NRS >4) was treated with a clinician bolus of 50mcg/kg with a time interval of 30 min at least between the additional doses till NRS decreased to less than 4. Excessive drowsiness or respiratory depression was treated by stopping the pump until appropriate recovery and by a decrease in the demand dose of 20%. 1 gm of paracetamol was administered intravenously every 8 hours. Postoperative hemodynamics, including systolic and diastolic blood pressures (SBP and DBP), HR, arterial oxygen saturation, NRS, and RASS, were recorded at 2, 4, 6, 12, and 24 hours. The total morphine consumption, including the continuous infusion, total boluses required, and the number of boluses, were recorded at
the end of the first 24 hours. The side effects of opioids were recorded, e.g., postoperative nausea and vomiting (PONV), pruritus, urine retention, and allergic reactions. Vomiting was treated with metoclopramide 10 mg. Ondansetron 8mg was given in persistent vomiting. Respiratory depression was treated with 1 mcg/kg of naloxone and oxygen supplied by an oxygen mask. NRS and patient satisfaction were recorded from the patient after 48, 72, 96 h, one month, and 3 months post-operative.
A blinded physician conducted CPSP assessments during patients’ visits to the pain clinic in the first and third postoperative months. They included the location, intensity, nature, duration of pain, and any aggravating or mitigating factors besides the used analgesic medication. CPSP was assessed using the Numeric Pain Rating Scale.
2.4. Statistical analysis
Using the G*power 3.1.9.2 program (Universitat Kiel, Germany) based on data from a previous study (10), a total sample of 54 (18 per group) subjects achieves 85% power to detect a difference of at least 5.5 mg in 24 h morphine consumption at an alpha error of 0.05. The common standard deviation is assumed to be 7. The number of envelopes will be increased to 60 (20 per group) to compensate for possible dropouts.
The Statistical Package for Social Science (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp). Categorical measurements were expressed as numbers and percentages (n [%]), and numerical measurements were expressed as mean (95% confidence intervals [CI]) and standard deviation [SD] if normally distributed or median, and interquartile range if abnormally distributed.
Shapiro Wilk’s test was used to evaluate the normal distribution of continuous data. Categorical measurements between groups were compared using the Chi-square test. Repeated measure analysis was used to assess the change over time of numerical measurements at different periods.
The Kolmogorov–Smirnov test was used to determine whether continuous variables had a specific distribution. Continuous data was analyzed using one-way analysis of variance (ANOVA) or Kruskal-Wallis test according to the normality of the data. Repeated measures were analyzed using ANOVA for repeated measures with post-hoc pairwise comparisons using Tukey’s tests. The level of statistical significance (P value) in all tests was considered as 0.05.
In this study, eighty-three patients undergoing elective major abdominal procedures with mid-line incisions under general anesthesia were enrolled. Twenty-three patients were excluded from the study, 15 of which didn’t fulfill the inclusion criteria, while eight declined to participate. The remaining sixty patients were allocated to three groups, twenty patients each, who finalized the study (Figure 1).
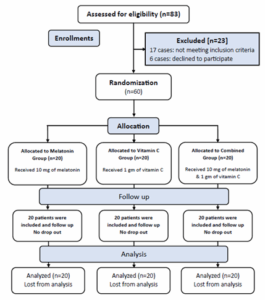
Figure 1: CONSORT flow diagram of the enrolled patients.
No significant differences were found between groups regarding patients’ demographic characteristics, recovery time, and surgery duration (Table 1). There were no significant differences between the groups regarding intraoperative or postoperative BP, HR, and arterial oxygen saturation.
The postoperative morphine consumption in the first 24 hours was significantly lower in Group CM than in Groups M and V, 16.7 mg ± 2.4, 20.98 mg ± 1.38, and 24.36 mg ± 3.12 in Groups VM, M, and V, respectively (P < 0.001). There was also a significant difference between Groups M and V regarding morphine consumption at 24 hours (P < 0.001) (Table 2). Intraoperative fentanyl consumption was lower in Groups VM and M compared to Group C (P < 0.001 and = 0.012, respectively). However, there was no significant difference between Group CM and M (P = 0.193).
The time to first analgesic requirement during the first 24 h was significantly longer in the VM Group, 12 ± 2.02 (P < 0.001). It was also longer in Group M than in Group C, 7.3 ± 3.2 and 5.1 ± 2.2 respectively (P < 0.001).
The NRS pain score was lower in Group CM than in groups M and C at 1, 2, 4, and 12 hours postoperative (P < 0.001). At 24 hours postoperative, the NRS pain score was lower in Group CM than in Group C (P < 0.001), while the difference between Group CM and Group M at 24 hours was not statistically significant (P = 0.07). The difference between Groups V and M was comparable except at 12 hours postoperative, as the NRS was higher in Group C (Figure 2).
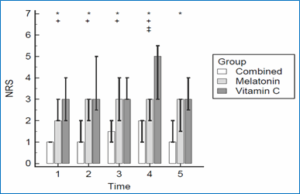
Figure 2: Numeric Rating Scale: Bars are medians, whiskers are 25th and 75th percentile; * denotes significance between Vitamin C and Combined groups; + denotes significance between Melatonin and Combined groups; ‡ denotes significance between Melatonin and Vitamin C groups; T1: 1 hour postoperative; T2: 2 hr; T3: 4 hr; T4: 12 hr; T5: 24 hr.
We found a significant difference between the three studied groups regarding PONV; the lowest incidence was in Group CM, and the highest one was in Group C (P < 0.001) (Table 2).
RASS score was lower in Group CM compared to Group C at 1, 2, 4, 12, and 24 hours postoperative (P < 0.001). It was also lower in Group M compared to Group C in all time points except at 12 hours postoperative (P = 0.106). RASS was lower in Group CM than in group M at 4 hours postoperative; however, it was comparable in both groups at 1, 2, 12, and 24 hours postoperative (Table 3).
None of the patients reported CPSP at one month or at three months postoperative.
In the present study, we found that 10 mg melatonin and 1 g vitamin C, given together two hours before surgery, reduced the postoperative morphine consumption, NSR scores, and the incidence of PONV compared to if either were given alone.
Administration of melatonin has a dose-dependent long-term anti-nociceptive effect in acute, neuropathic, and inflammatory pain. This is explained by exhibiting an effective change in pain threshold through attenuating nociceptive responses to various noxious stimuli. Several studies proved melatonin elevates β-endorphin levels within the central nervous system and interacts with the centrally located opioid, γ-aminobutyric acid (GABA), and N-methyl-D-aspartate receptor systems.10 Moreover, melatonin reduces the elevated expression of nuclear factor kappa B (NF-kB) and inhibits the enhanced level of pro-inflammatory cytokines IL-6 or TNF-a to modulate neuroinflammation.11
Our results are consistent with previous reports on using melatonin as an adjuvant analgesic in abdominal surgeries. In which it was compared with placebo or various adjuvant analgesics. Kiabi FH et al. investigated the postoperative analgesic effect of melatonin in patients undergoing elective cesarean section under spinal anesthesia; they showed that melatonin 10 mg given one hour preoperatively decreased postoperative pain intensity, the time to require analgesia, and the opioid requirements with a shorter time to resume mobility.12 Alkhfaji H et al. also reported the effect of melatonin on postoperative pain in patients who performed cesarean section; melatonin did not only decrease the postoperative pain severity and opioid requirements; it also reduced the intra- and postoperative blood loss.13 Borazan H et al. reported that 6 mg of melatonin given the night before surgery and repeated 1 hour preoperatively decreased intraoperative fentanyl requirements, postoperative pain severity, and tramadol consumption, with better postoperative sleep quality and higher sedation scores.14 Haryalchi et al. reported that 6 mg of melatonin orally given preoperatively had decreased the severity of postoperative pain.15 On the other side, Khezri M et al. tested two doses of melatonin in patients undergoing cesarean section, and they found that melatonin did not improve analgesia. Nevertheless, it increased the incidence of postoperative headache with the higher dose.16 Andersen LP et al. injected 10 mg of melatonin intravenously at the time of incision in patients undergoing laparoscopic cholecystectomy; they found no effect on pain intensity and analgesic usage.17 This controversy may be referred to the type of surgery and anesthesia or the different dosages or routes of administration.
Vitamin C could reduce postoperative pain through different mechanisms. They include its antioxidant characteristics, anti-inflammatory effect, neuroprotective effect, and involvement in collagen production.18 It is also crucial for producing neurotransmitters like dopamine, epinephrine, and serotonin; they are essential for cholinergic and GABAergic neural transmissions. These neurotransmitters also play a role in inhibiting pain pathways.19 It has proven to reduce the severity of acute pain and the prevalence of complex regional pain syndromes with its anti-nociceptive effect.7
Consistent with our results, Kumar A. et al. investigated the analgesic effect of vitamin C given orally at a dose of 2 gm administered the night before surgery and repeated 2 hours preoperatively in patients undergoing laparoscopic surgeries, they reported a reduction in postoperative pain intensity and opioid consumption.20 Aboelelea M. et al. also reported the pain-reducing effect of vitamin C and the associated lower postoperative morphine consumption in patients who performed laparoscopic sleeve gastrectomy.21 Jeon et al. injected 50 mg/kg of vitamin C immediately after anesthesia induction in patients undergoing laparoscopic colectomy; they found that vitamin C decreased the pain intensity in the first 24 hours postoperatively and reduced the total morphine consumption.22 Moon et al. reported that adding magnesium to vitamin C was more effective in reducing postoperative pain than using each agent separately. (23) A meta-analysis searched the effect of vitamin C on postoperative analgesic consumption. It stated that perioperative use of vitamin C has shown a significant reduction in pain score and opioid requirement up to postoperative 24 hours.19 Another meta-analysis has shown that the evidence on the postoperative pain-reducing effect of vitamin C and its role in reducing morphine consumption is of very low certainty, which cannot support a systematic use outside a research setting.24
Tunay et al. reported that preoperative administration of melatonin or vitamin C was associated with lower pain scores, analgesics consumption, and lower intensity of PONV compared to placebo in patients undergoing major abdominal surgeries; they suggest that the analgesic effect of the study drugs is referred to their interaction with morphine.10
Despite the several studies that have proven the pain-relieving effects of melatonin and vitamin C, it is still uncertain whether their combined analgesic effects are synergistic or additive in treating postoperative pain. To the extent of our knowledge, this is the first study that investigated the impact of combining both melatonin and vitamin C on postoperative pain, opioid consumption, and opioid adverse effects after major abdominal surgeries.
Managing postoperative pain is a significant concern, as studies show that up to 75% of patients report their pain levels as moderate, severe, or extreme. (25)Effective control of acute pain is vital in preventing the development of CPSP.26 Several studies have indicated a correlation between the severity of acute pain following surgery and the incidence of chronic pain.27 However, balancing effective pain management with the need to minimize side effects from analgesics remains a challenge for physicians. Therefore, despite the efficacy of opioids, cases of insufficient pain management are not uncommon due to their adverse effects.28 Current clinical guidelines advocate for a multimodal analgesia approach, which combines various analgesic medications and interventions. (29) In this context, combining melatonin and vitamin C could be a beneficial adjuvant to conventional analgesics.
First, our study is a single-center study. Second, we did not measure the concentration of vitamin C or melatonin in serum pre- and post-medication administration. Finally, the lower prevalence of CPSP in abdominal surgeries warrants further studies, especially in surgeries with a higher incidence of CPSP, e.g., radical mastectomy.
In patients undergoing major abdominal surgery with mid-line incision, combined use of melatonin and vitamin C provides a synergistic analgesic effect in postoperative pain management with lower postoperative pain scores, opioid consumption, and the incidence of PONV, compared to using any of them alone.
7. Data availability
The numerical data related to this study is available with the authors.
8. Acknowledgment
We want to sincerely thank Dr. Maha Mostafa, Associate Professor of Anesthesiology, Faculty of Medicine, Cairo University, for her kind help in statistical analysis.
9. Conflict of interest
The authors report no conflict of interest.
10. Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
11. Authors’ contribution
WH, KR, and MA were responsible for the idea's conception. WH, KR, Am K, and DS contributed to the study design. Ah K, Am K, and DS were responsible for data collection, analysis and interpretation, and manuscript writing. All authors read, revised, and approved the final manuscript. The corresponding author agreed to be accountable for all aspects of the work.
Authors affiliations:
- Amr Kamal Abdelhakim, MD, Assistant Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Cairo University, Cairo, Egypt: Email: Amr.kamal@kasralainy.edu.eg, ORCID: {0000-0003-4536-1312}
- Dina Samir Mahmoud Shaker, M.Sc, Assistant Lecturer of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Banha University, Qalubia, Egypt; Email: Dina.shaker@fmed.bu.edu.eg
- Waleed Ibrahim Hamimy, MD, Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Cairo University, Cairo, Egypt; hamimy2002@gmail.com
- Maha Abdelbar Attalla, MD, Professor of Anesthesiology, Intensive Care & Pain Management, Theodor Bilharz Research Institute, Cairo, Egypt; maha_atta_2008@hotmail.com, ORCID: {0000-0001-7619-0347}
- Khalda Galal Radwan, MD, Professor of Anesthesiology, Intensive Care & Pain Management, Theodor Bilharz Research Institute, Cairo, Egypt; Email: khalda.radwan31@gmail.com
- Ahmed Kareem Mohammed Abdelhady, MD, Assistant Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Cairo University, Cairo, Egypt; Email: Dr.ahmedkar@kasralainy.edu.eg, ORCID: {0000-0001-6321-3888}
ABSTRACT
Background & objectives: Several studies have shown that melatonin and vitamin C used as an analgesic adjuvant, can spare opioid use, resulting in a better analgesic profile with fewer adverse effects and shorter hospital stay. However, we found no studies which might have investigated the impact of a combination of both. We compared the effect of melatonin, vitamin C, and their combination on postoperative opioid consumption.
Methodology: Sixty patients scheduled for major abdominal surgeries were randomly allocated into three equal groups with 20 patients in each; melatonin group (Group M), vitamin C (Group C), and combined melatonin and vitamin C (Group CM). Two hours before surgery, all patients received the study medications orally, which was continued for 3 days postoperative at the same time of the first administration; Group M received 10 mg of melatonin, Group C received 1 gm of vitamin C, and Group MV received both melatonin (10 mg) and vitamin C (1 gm). The primary outcome was the total morphine consumption in 24 hours postoperatively, with patients, pain scores as measured by Numeric Pain Rating Scale (NPRS), the incidence of postoperative nausea and vomiting (PONV), and postoperative chronic pain as secondary outcomes.
Results: Postoperative morphine consumption (in the first 24 hours) was significantly lower in Group CM (16.7 ± 2.4 mg) than in Group M (20.98 ± 1.38 mg), and Group C (24.36 ± 3.12 mg) (P < 0.001). Group CM showed lower pain scores, decreased incidence of PONV, and a longer time to first request for analgesics. However, lower sedation scores were observed. There was no statistical difference among all groups regarding post-operative chronic pain incidence.
Conclusion: Combined use of melatonin and vitamin C in patients undergoing major abdominal surgery with mid-line incision provides a synergistic analgesic effect for the postoperative pain management with lower postoperative pain scores, less opioid consumption, and lower incidence of PONV compared to the use of any one of these.
Abbreviations: CPSP: Chronic Post-Surgical Pain, NPRS: Numeric Pain Rating Scale, PONV: postoperative nausea and vomiting, TAHBSO: Total Abdominal Hysterectomy with Bilateral Salpingo-Oophorectomy
Keywords: Melatonin; major abdominal surgery; Numeric Pain Rating Scale; opioid consumption; postoperative pain; PONV; vitamin C.
Citation: Abdelhakim AK, Shaker DSM, Hamimy WI, Attalla MA, Radwan KG, Abdelhady AKM. Anaesth. pain intensive care 2025;29(3):556-664. DOI: 10.35975/apic.v29i3.2790
Received: January 03, 2025; Revised: March 12, 2025; Accepted: March 22, 2025
1. INTRODUCTION
Major abdominal surgeries cause changes in neuro-hormonal response, resulting in postoperative pain, various organ dysfunction, and prolonged hospitalization. Inadequate pain relief after major abdominal surgeries adversely affects the surgical outcome through slower rehabilitation and a more extended hospital stay.1
Chronic Post-Surgical Pain (CPSP) is defined as a persistent pain of duration longer than 2 months after the surgical procedure, excluding other causes of pain such as malignancy, infection, and pre-existing pain problems. Many procedures are likely to be associated with CPSP if the postoperative pain is not adequately controlled.
Major abdominal surgeries in previous studies have reported CPSP with an incidence ranging from 17% to 32%.2
Post-operative pain after major abdominal surgery is usually controlled through multimodal analgesia, which may be in the form of intravenous opioids, non-steroidal anti-inflammatory drugs, and paracetamol, together with regional blocks.3
There are many side effects of opioids, including sedation, dizziness, nausea, vomiting, constipation, physical dependence, tolerance, and respiratory depression.4
Therefore, many studies have been conducted to achieve effective postoperative pain control and improve patient satisfaction with minimal hazards. Several studies showed that the use of different adjuvants could spare opioid use, resulting in better rehabilitation, surgical outcomes, and less hospital stay.5
One of these adjuvants is melatonin. Melatonin (N-acetyl-5-methoxytryptamine) is an endogenous hormone the pineal gland produces and is essential to the circadian rhythm. Melatonin has proved to have a postoperative analgesic effect and to reduce opioid consumption. Administration of melatonin has dose-dependent long-term anti-nociceptive effects in acute, neuropathic, and inflammatory pain.6
Vitamin C is a water-soluble vitamin responsible for normal growth and development. It also has antioxidant, neuro-protective, and neuro-modulation effects. Vitamin C has been proven to reduce acute pain and the prevalence of complex regional pain syndromes with its anti-nociceptive effect.7 Certain studies have demonstrated that the combination of vitamin C with an opioid may diminish both opioid consumption for pain relief and the rate of opioid-related adverse effects.8
Several multimodal approaches have been advocated to reduce opioid consumption by using either melatonin or vitamin C.9 This is the first study conducted on using the combination of both in reducing opioid consumption in major abdominal surgeries.
2. METHODOLOGY
This randomized, double-blinded, controlled study was conducted in the general surgery, uro-surgery, and gynecology operating rooms of Kasr Al Ainy Hospital, Cairo, Egypt, from January 2023 to July 2024. Ethical approval of the Institutional Ethics Committee (No. MD-15-2022) was obtained according to the ethical standards of the Declaration of Helsinki. The trial was registered at ClinicalTrials.gov (No. NCT06374771).
We enrolled 60 patients aged between 18 and 60 y, ASA- I–II, and scheduled for major abdominal surgeries with mid-line incisions, e.g., radical prostatectomy, nephrectomy, Total Abdominal Hysterectomy with Bilateral Salpingo-Oophorectomy (TAHBSO), hepatectomy, splenectomy, hepatic hydatid cyst excision. Informed signed consent was obtained from every participant. Those who refused to participate, were known allergic to the drugs used in the study, had a hepatic failure (Child class B- Child class C) or a renal impairment (creatinine >2mg/dL or chronic kidney disease on regular dialysis), or had a history of psychiatric or mental disorders, Chronic pain syndromes (neuropathic pain, post-herpetic neuralgia, Complex regional pain syndrome), or drug or alcohol abuse, were excluded.
Randomization was performed using computer-generated numbers with Random Allocation Software (https://random-allocation-software.software.informer.com/2.0/) in a 1:1:1 ratio. Sixty consequentially numbered opaque envelopes (twenty per group) containing group assignment and drug administration instructions were made. A research assistant was responsible for opening the envelopes and administering the assigned drug with no further involvement in the study. A blinded anesthesiologist was responsible for the anesthetic management, perioperative data collection, postoperative pain assessment, and analgesia administration.
Patients were allocated into three groups: the melatonin group (Group M), the vitamin C group (Group C), and the Melatonin and vitamin C group (Group CM).
The primary outcome was the total morphine consumption in the first 24 postoperative hours. Other outcomes included intraoperative fentanyl consumption, intraoperative and postoperative blood pressure (BP) and heart rate (HR), postoperative Richmond Agitation Sedation Scale (RASS), the time to first analgesic requirement, defined as the first recognized time point of Numeric Rating Scale (NRS) > 4 assessed during the first 24 h, the number of morphine boluses given after discharge from PACU, the frequency of postoperative chronic pain, and any adverse events.
2.1. Management Protocol
The day before surgery, participants were evaluated by acquiring a thorough medical history, physical examination, and investigations. Two hours before surgery, all patients will receive the study drugs orally: 10mg of melatonin for Group M (Melatonin 10 mg - Puritan’s Pride premium company), 1 gm of vitamin C for Group C (Sanso C 1000 mg - AUG pharma company), and 10mg of melatonin and 1 gm of vitamin C for Group CM.
The patients were transferred to the preparation room one hour before the procedure, and the NRS pain score was explained to all candidates (zero corresponds to no pain, and 10 indicates the worst unbearable pain). Baseline vital signs were recorded, including noninvasive measuring of systolic, mean, diastolic arterial blood pressures, heart rate, and oxygen saturation.
2.2. Anesthetic technique
Standard monitoring (non-invasive blood pressure, electrocardiography, and pulse oximetry) was applied upon arrival at the operating room. The patient received pre-induction ondansetron 8mg. Following pre-oxygenation via a face mask for 3 min, anesthesia was induced using propofol 1-2 mg/kg and fentanyl 2 µg/kg. Atracurium 0.5 mg/kg was administered to facilitate endotracheal intubation. After intubation, the capnography will be connected, and the patients will be mechanically ventilated to maintain end-tidal CO2 between 30-35 mmHg.
Anaesthesia was maintained with sevoflurane in 50% oxygen, and atracurium 0.1mg/kg every 20 min was administered to maintain muscle relaxation. After the skin incision, a bolus dose of morphine 100 mcg/kg was given. Fentanyl at 1µg/kg was administered as rescue analgesia if an intraoperative increase in heart rate or systolic blood pressure by 20% occurred after the exclusion of other causes rather than pain. Before skin closure, paracetamol 1gm was given intravenously. A bispectral index monitoring device was used to monitor the anesthesia depth. Readings within 40 to 55 were targeted throughout the procedure. Mean blood pressure and heart rate were maintained within ±20%.
2.3. Postoperative Management
Recovery time was recorded, defined as the time from extubation to the time of achieving Aldrete scores ≥ 9. The patient was transferred to the post-anesthesia care unit (PACU); Blood pressure, heart rate, and oxygen saturation were monitored every 15 min till discharge from the PACU. In PACU, 50mcg/kg of morphine was given by the physician to all patients. Then, all patients received a continuous morphine infusion of 5mcg/kg/hour through a disposable infusion pump 275ml with a constant flow of 5ml/hour (Zhejiang Fert Medical Device Co., Ltd). The patients were instructed to report any pain to a pain nurse, and if the NRS increased to more than 4, the pain nurse informed the anesthesiologist. Inadequate analgesia (NRS >4) was treated with a clinician bolus of 50mcg/kg with a time interval of 30 min at least between the additional doses till NRS decreased to less than 4. Excessive drowsiness or respiratory depression was treated by stopping the pump until appropriate recovery and by a decrease in the demand dose of 20%. 1 gm of paracetamol was administered intravenously every 8 hours. Postoperative hemodynamics, including systolic and diastolic blood pressures (SBP and DBP), HR, arterial oxygen saturation, NRS, and RASS, were recorded at 2, 4, 6, 12, and 24 hours. The total morphine consumption, including the continuous infusion, total boluses required, and the number of boluses, were recorded at
the end of the first 24 hours. The side effects of opioids were recorded, e.g., postoperative nausea and vomiting (PONV), pruritus, urine retention, and allergic reactions. Vomiting was treated with metoclopramide 10 mg. Ondansetron 8mg was given in persistent vomiting. Respiratory depression was treated with 1 mcg/kg of naloxone and oxygen supplied by an oxygen mask. NRS and patient satisfaction were recorded from the patient after 48, 72, 96 h, one month, and 3 months post-operative.
A blinded physician conducted CPSP assessments during patients’ visits to the pain clinic in the first and third postoperative months. They included the location, intensity, nature, duration of pain, and any aggravating or mitigating factors besides the used analgesic medication. CPSP was assessed using the Numeric Pain Rating Scale.
2.4. Statistical analysis
Using the G*power 3.1.9.2 program (Universitat Kiel, Germany) based on data from a previous study (10), a total sample of 54 (18 per group) subjects achieves 85% power to detect a difference of at least 5.5 mg in 24 h morphine consumption at an alpha error of 0.05. The common standard deviation is assumed to be 7. The number of envelopes will be increased to 60 (20 per group) to compensate for possible dropouts.
The Statistical Package for Social Science (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp). Categorical measurements were expressed as numbers and percentages (n [%]), and numerical measurements were expressed as mean (95% confidence intervals [CI]) and standard deviation [SD] if normally distributed or median, and interquartile range if abnormally distributed.
Shapiro Wilk’s test was used to evaluate the normal distribution of continuous data. Categorical measurements between groups were compared using the Chi-square test. Repeated measure analysis was used to assess the change over time of numerical measurements at different periods.
The Kolmogorov–Smirnov test was used to determine whether continuous variables had a specific distribution. Continuous data was analyzed using one-way analysis of variance (ANOVA) or Kruskal-Wallis test according to the normality of the data. Repeated measures were analyzed using ANOVA for repeated measures with post-hoc pairwise comparisons using Tukey’s tests. The level of statistical significance (P value) in all tests was considered as 0.05.
3. RESULTS
In this study, eighty-three patients undergoing elective major abdominal procedures with mid-line incisions under general anesthesia were enrolled. Twenty-three patients were excluded from the study, 15 of which didn’t fulfill the inclusion criteria, while eight declined to participate. The remaining sixty patients were allocated to three groups, twenty patients each, who finalized the study (Figure 1).

Figure 1: CONSORT flow diagram of the enrolled patients.
No significant differences were found between groups regarding patients’ demographic characteristics, recovery time, and surgery duration (Table 1). There were no significant differences between the groups regarding intraoperative or postoperative BP, HR, and arterial oxygen saturation.
| Table 1: Comparative demographic data and baseline characteristics | |||||
| Parameter | Melatonin group
(n = 20) |
Vitamin C group
(n = 20) |
Melatonin + Vit C
group (n = 20) |
P-value | |
| Age (yr) | 49.0 ± 12.1 | 44.2 ± 14.2 | 44.8 ± 11.3 | 0.43 | |
| Weight (kg) | 82.6 ± 12.1 | 84 ± 13.6 | 84.2 ± 13.4 | 0.91 | |
| Operative time (hr) | 2.6 ± 0.6 | 2.5 ± 0.6 | 2.4 ± 0.7 | 0.69 | |
| Recovery time (hr) | 5.2 ± 1.88 | 4.6 ± 1.67 | 5.6 ± 2.01 | 0.239 | |
| Gender | Male | 6 (30) | 8 (40) | 8 (40) | 0.75 |
| Female | 14 (70) | 12 (60) | 12 (60) | ||
| ASA | I | 9 (45) | 9 (45) | 11 (55) | 0.76 |
| II | 11 (55) | 11 (55) | 9 (45) | ||
| Surgery |
Partial hepatectomy | 3 (15) | 2 (10) | 3 (15) | 0.82 |
| Hepatic hydatid cyst excision | 2 (10) | 2 (10) | 3 (15) | ||
| TAHBSO | 8 (40) | 7 (35) | 5 (25) | ||
| Radical prostatectomy | 5 (25) | 6 (30) | 6 (30) | ||
| Nephrectomy | 2 (10) | 3 (15) | 3 (15) | ||
| ASA= American Society of Anesthesiologists, TAHBSO: Total Abdominal Hysterectomy with Bilateral Salpingo-Oophorectomy.
Data are presented as mean ± SD or numbers (%) |
|||||
The postoperative morphine consumption in the first 24 hours was significantly lower in Group CM than in Groups M and V, 16.7 mg ± 2.4, 20.98 mg ± 1.38, and 24.36 mg ± 3.12 in Groups VM, M, and V, respectively (P < 0.001). There was also a significant difference between Groups M and V regarding morphine consumption at 24 hours (P < 0.001) (Table 2). Intraoperative fentanyl consumption was lower in Groups VM and M compared to Group C (P < 0.001 and = 0.012, respectively). However, there was no significant difference between Group CM and M (P = 0.193).
The time to first analgesic requirement during the first 24 h was significantly longer in the VM Group, 12 ± 2.02 (P < 0.001). It was also longer in Group M than in Group C, 7.3 ± 3.2 and 5.1 ± 2.2 respectively (P < 0.001).
The NRS pain score was lower in Group CM than in groups M and C at 1, 2, 4, and 12 hours postoperative (P < 0.001). At 24 hours postoperative, the NRS pain score was lower in Group CM than in Group C (P < 0.001), while the difference between Group CM and Group M at 24 hours was not statistically significant (P = 0.07). The difference between Groups V and M was comparable except at 12 hours postoperative, as the NRS was higher in Group C (Figure 2).

Figure 2: Numeric Rating Scale: Bars are medians, whiskers are 25th and 75th percentile; * denotes significance between Vitamin C and Combined groups; + denotes significance between Melatonin and Combined groups; ‡ denotes significance between Melatonin and Vitamin C groups; T1: 1 hour postoperative; T2: 2 hr; T3: 4 hr; T4: 12 hr; T5: 24 hr.
We found a significant difference between the three studied groups regarding PONV; the lowest incidence was in Group CM, and the highest one was in Group C (P < 0.001) (Table 2).
| Table 2: Comparative analgesics consumption and PONV in the three groups | ||||
| Variables | Melatonin group
(n = 20) |
Vitamin C group
(n = 20) |
Melatonin + Vit C
group (n = 20) |
P-value |
| Postop morphine consumption (mg) | 20.98 ± 1.38 +‡ | 24.36 ± 3.12 * | 16.7 ± 2.4 | 0.001 |
| Fentanyl consumption (µg) | 169.6 ± 47.5 ‡ | 207.0 ± 39.2 * | 156 ± 30.3 | 0.001 |
| First time to rescue analgesia (hr) | 7.3 ± 3.2 +‡ | 5.1 ± 2.2 * | 12.5 ± 2.02 | 0.001 |
| Number of morphine boluses | 2 (1-2) +‡ | 2.5 (2-3) * | 1 (0-1) | 0.001 |
| · Zero | 0 | 0 | 8 (40) | |
| · 1 | 7 (35) | 1 (5) | 12 (60) | |
| · 2 | 11 (55) | 9 (45) | 0 | |
| · 3 | 2 (10) | 10 (50) | 0 | |
| PONV | 5 (25) +‡ | 13 (65) * | 2 (10) | 0.001 |
| data presented as mean ± SD, median (quartiles), or numbers (%); PONV: Postoperative Nausea and Vomiting; * denotes significance between Vitamin C group and Combined group; + denotes significance between Melatonin group and Combined group; ‡ denotes significance between Melatonin group and Vitamin C group. | ||||
RASS score was lower in Group CM compared to Group C at 1, 2, 4, 12, and 24 hours postoperative (P < 0.001). It was also lower in Group M compared to Group C in all time points except at 12 hours postoperative (P = 0.106). RASS was lower in Group CM than in group M at 4 hours postoperative; however, it was comparable in both groups at 1, 2, 12, and 24 hours postoperative (Table 3).
| Table 3: Comparative Richmond Agitation Sedation Scale (RASS) scores in the groups | ||||
| Postop time (hr) | Melatonin group
(n = 20) |
Vit C group
(n = 20) |
Melatonin + Vit C group
(n = 20) |
P-value |
| 1 | -1 (-1, 0) ‡ | 0 (0, 1) * | -1 (-1, -2) | < 0.001 |
| 2 | -1 (-1, 0) ‡ | 0 (0, 1) * | -1 (-2, -1) | < 0.001 |
| 4 | 0 (-1, 0) + | 0 (0, 1) * | -1 (-1, 0) | 0.029 |
| 12 | 0 (0, 0) ‡ | 1 (0, 1) * | 0 (-1, 0) | < 0.001 |
| 24 | 0 (-1, 0) ‡ | 0 (0, 1) * | -1 (-1, 0) | < 0.001 |
| Data presented as median (quartiles); *denotes significance between Vitamin C group and Combined group; + denotes significance between Melatonin group and Combined group; ‡ denotes significance between Melatonin group and Vitamin C group | ||||
None of the patients reported CPSP at one month or at three months postoperative.
4. DISCUSSION
In the present study, we found that 10 mg melatonin and 1 g vitamin C, given together two hours before surgery, reduced the postoperative morphine consumption, NSR scores, and the incidence of PONV compared to if either were given alone.
Administration of melatonin has a dose-dependent long-term anti-nociceptive effect in acute, neuropathic, and inflammatory pain. This is explained by exhibiting an effective change in pain threshold through attenuating nociceptive responses to various noxious stimuli. Several studies proved melatonin elevates β-endorphin levels within the central nervous system and interacts with the centrally located opioid, γ-aminobutyric acid (GABA), and N-methyl-D-aspartate receptor systems.10 Moreover, melatonin reduces the elevated expression of nuclear factor kappa B (NF-kB) and inhibits the enhanced level of pro-inflammatory cytokines IL-6 or TNF-a to modulate neuroinflammation.11
Our results are consistent with previous reports on using melatonin as an adjuvant analgesic in abdominal surgeries. In which it was compared with placebo or various adjuvant analgesics. Kiabi FH et al. investigated the postoperative analgesic effect of melatonin in patients undergoing elective cesarean section under spinal anesthesia; they showed that melatonin 10 mg given one hour preoperatively decreased postoperative pain intensity, the time to require analgesia, and the opioid requirements with a shorter time to resume mobility.12 Alkhfaji H et al. also reported the effect of melatonin on postoperative pain in patients who performed cesarean section; melatonin did not only decrease the postoperative pain severity and opioid requirements; it also reduced the intra- and postoperative blood loss.13 Borazan H et al. reported that 6 mg of melatonin given the night before surgery and repeated 1 hour preoperatively decreased intraoperative fentanyl requirements, postoperative pain severity, and tramadol consumption, with better postoperative sleep quality and higher sedation scores.14 Haryalchi et al. reported that 6 mg of melatonin orally given preoperatively had decreased the severity of postoperative pain.15 On the other side, Khezri M et al. tested two doses of melatonin in patients undergoing cesarean section, and they found that melatonin did not improve analgesia. Nevertheless, it increased the incidence of postoperative headache with the higher dose.16 Andersen LP et al. injected 10 mg of melatonin intravenously at the time of incision in patients undergoing laparoscopic cholecystectomy; they found no effect on pain intensity and analgesic usage.17 This controversy may be referred to the type of surgery and anesthesia or the different dosages or routes of administration.
Vitamin C could reduce postoperative pain through different mechanisms. They include its antioxidant characteristics, anti-inflammatory effect, neuroprotective effect, and involvement in collagen production.18 It is also crucial for producing neurotransmitters like dopamine, epinephrine, and serotonin; they are essential for cholinergic and GABAergic neural transmissions. These neurotransmitters also play a role in inhibiting pain pathways.19 It has proven to reduce the severity of acute pain and the prevalence of complex regional pain syndromes with its anti-nociceptive effect.7
Consistent with our results, Kumar A. et al. investigated the analgesic effect of vitamin C given orally at a dose of 2 gm administered the night before surgery and repeated 2 hours preoperatively in patients undergoing laparoscopic surgeries, they reported a reduction in postoperative pain intensity and opioid consumption.20 Aboelelea M. et al. also reported the pain-reducing effect of vitamin C and the associated lower postoperative morphine consumption in patients who performed laparoscopic sleeve gastrectomy.21 Jeon et al. injected 50 mg/kg of vitamin C immediately after anesthesia induction in patients undergoing laparoscopic colectomy; they found that vitamin C decreased the pain intensity in the first 24 hours postoperatively and reduced the total morphine consumption.22 Moon et al. reported that adding magnesium to vitamin C was more effective in reducing postoperative pain than using each agent separately. (23) A meta-analysis searched the effect of vitamin C on postoperative analgesic consumption. It stated that perioperative use of vitamin C has shown a significant reduction in pain score and opioid requirement up to postoperative 24 hours.19 Another meta-analysis has shown that the evidence on the postoperative pain-reducing effect of vitamin C and its role in reducing morphine consumption is of very low certainty, which cannot support a systematic use outside a research setting.24
Tunay et al. reported that preoperative administration of melatonin or vitamin C was associated with lower pain scores, analgesics consumption, and lower intensity of PONV compared to placebo in patients undergoing major abdominal surgeries; they suggest that the analgesic effect of the study drugs is referred to their interaction with morphine.10
Despite the several studies that have proven the pain-relieving effects of melatonin and vitamin C, it is still uncertain whether their combined analgesic effects are synergistic or additive in treating postoperative pain. To the extent of our knowledge, this is the first study that investigated the impact of combining both melatonin and vitamin C on postoperative pain, opioid consumption, and opioid adverse effects after major abdominal surgeries.
Managing postoperative pain is a significant concern, as studies show that up to 75% of patients report their pain levels as moderate, severe, or extreme. (25)Effective control of acute pain is vital in preventing the development of CPSP.26 Several studies have indicated a correlation between the severity of acute pain following surgery and the incidence of chronic pain.27 However, balancing effective pain management with the need to minimize side effects from analgesics remains a challenge for physicians. Therefore, despite the efficacy of opioids, cases of insufficient pain management are not uncommon due to their adverse effects.28 Current clinical guidelines advocate for a multimodal analgesia approach, which combines various analgesic medications and interventions. (29) In this context, combining melatonin and vitamin C could be a beneficial adjuvant to conventional analgesics.
5. LIMITATIONS
First, our study is a single-center study. Second, we did not measure the concentration of vitamin C or melatonin in serum pre- and post-medication administration. Finally, the lower prevalence of CPSP in abdominal surgeries warrants further studies, especially in surgeries with a higher incidence of CPSP, e.g., radical mastectomy.
6. CONCLUSION
In patients undergoing major abdominal surgery with mid-line incision, combined use of melatonin and vitamin C provides a synergistic analgesic effect in postoperative pain management with lower postoperative pain scores, opioid consumption, and the incidence of PONV, compared to using any of them alone.
7. Data availability
The numerical data related to this study is available with the authors.
8. Acknowledgment
We want to sincerely thank Dr. Maha Mostafa, Associate Professor of Anesthesiology, Faculty of Medicine, Cairo University, for her kind help in statistical analysis.
9. Conflict of interest
The authors report no conflict of interest.
10. Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
11. Authors’ contribution
WH, KR, and MA were responsible for the idea's conception. WH, KR, Am K, and DS contributed to the study design. Ah K, Am K, and DS were responsible for data collection, analysis and interpretation, and manuscript writing. All authors read, revised, and approved the final manuscript. The corresponding author agreed to be accountable for all aspects of the work.
12. REFERENCES
- Wan HY, Li SY, Ji W, Yu B, Jiang N. Fascia Iliaca Compartment Block for Perioperative Pain Management of Geriatric Patients with Hip Fractures: A Systematic Review of Randomized Controlled Trials. Pain Res Manag. 2020 Nov 25;2020:8503963. PubMed DOI: 1155/2020/8503963
- Correll D. Chronic postoperative pain: Recent findings in understanding and management. F1000Res. 2017 Jul 4:6:1054. PubMed DOI: 12688/f1000research.11101.1
- Capdevila X, Moulard S, Plasse C, Peshaud JL, Molinari N, Dadure C, et al. Effectiveness of Epidural Analgesia, Continuous Surgical Site Analgesia, and Patient-Controlled Analgesic Morphine for Postoperative Pain Management and Hyperalgesia, Rehabilitation, and Health-Related Quality of Life After Open Nephrectomy: A Prospective, Randomized, Controlled Study. Anesth Analg. 2017 Jan 1; 124(1):336–45. PubMed DOI: 1213/ANE.0000000000001688
- Pirie K, Traer E, Finniss D, Myles PS, Riedel B. Current approaches to acute postoperative pain management after major abdominal surgery: a narrative review and future directions. Br J Anaesth. 2022 Sep 1; 129(3):378–93. PubMed DOI: 1016/j.bja.2022.05.029
- Mercadante S, Arcuri E, Santoni A. Opioid-Induced Tolerance and Hyperalgesia. CNS Drugs. 2019 Oct 1;33(10):943–55. PubMed DOI: 1007/s40263-019-00660-0
- Yousaf F, Seet E, Venkatraghavan L, Abrishami A, Chung F. Efficacy and safety of melatonin as an anxiolytic and analgesic in the perioperative period: a qualitative systematic review of randomized trials. Anesthesiology. 2010;113(4):968–76. PubMed DOI: 1097/ALN.0b013e3181e7d626
- Lu R, Kallenborn-Gerhardt W, Geisslinger G, Schmidtko A. Additive antinociceptive effects of a combination of vitamin C and vitamin E after peripheral nerve injury. PLoS One. 2011 Dec 14;6(12). PubMed DOI: 1371/journal.pone.0029240
- Jain SK, Dar MY, Kumar S, Yadav A, Kearns SR. Role of anti-oxidant (vitamin-C) in post-operative pain relief in foot and ankle trauma surgery: A prospective randomized trial. Foot Ankle Surg. 2019 Aug 1;25(4):542–5. PubMed DOI: 1016/j.fas.2018.05.001
- Jeon Y, Park JS, Moon S, Yeo J. Effect of Intravenous High Dose Vitamin C on Postoperative Pain and Morphine Use after Laparoscopic Colectomy: A Randomized Controlled Trial. Pain Res Manag. 2016;2016. PubMed DOI: 1155/2016/9147279
- Tunay DL, Ilgınel MT, Ünlügenç H, Tunay M, Karacaer F, Biricik E. Comparison of the effects of preoperative melatonin or vitamin C administration on postoperative analgesia. Bosn J Basic Med Sci. 2020;20(1):117–24. PubMed DOI: 17305/bjbms.2019.4379
- Madsen BK, Zetner D, Møller AM, Rosenberg J. Melatonin for preoperative and postoperative anxiety in adults. Cochrane Database Syst Rev. 2020 Dec 8 ;12(12). PubMed DOI: 1002/14651858.CD009861.pub3
- Kiabi FH, Emadi SA, Jamkhaneh AE, Aezzi G, Ahmadi NS. Effects of preoperative melatonin on postoperative pain following cesarean section: A randomized clinical trial. Ann Med Surg (Lond). 2021 Jun 1;66. PubMed DOI: 1016/j.amsu.2021.102345
- Alkhfaji H, Kahloul M, Askar TRM, Alhamaidah MF, Ali Hussein H. Impact of Melatonin as a Premedication Agent in Caesarean Section on Blood Loss and Postoperative Pain Level. Anesthesiol Res Pract. 2023 Dec; 12:2023:8102111. PubMed DOI: 1155/2023/8102111
- Borazan H, Tuncer S, Yalcin N, Erol A, Otelcioglu S. Effects of preoperative oral melatonin medication on postoperative analgesia, sleep quality, and sedation in patients undergoing elective prostatectomy: a randomized clinical trial. J Anesth. 2010 Apr;24(2):155–60. PubMed DOI: 1007/s00540-010-0891-8
- Haryalchi K, Ghanaei MM, Rajabi M, Ghazizadeh M, Aghajanpour F, Koochakpoor P, et al. Preventive Dose of Melatonin on Postoperative Pain in Total Abdominal Hysterectomy: A Clinical Trial Study. J Obstetr Gynecol Cancer Res. 2024;9(1):2476–5848. Full Text
- Khezri MB, Reihany MD, Oveisy S, Mohammadi N. Evaluation of the Analgesic Efficacy of Melatonin in Patients Undergoing Cesarean Section Under S pinal Anesthesia: A Prospective Randomized Double-blind Study. Iran J Pharm Res. 2016 Sep 1;15(4):963. PubMed
- Andersen LPH, Kücükakin B, Werner MU, Rosenberg J, Gögenur I. Absence of analgesic effect of intravenous melatonin administration during daytime after laparoscopic cholecystectomy: a randomized trial. J Clin Anesth. 2014;26(7):545–50. PubMed DOI: 1016/j.jclinane.2014.03.008
- Jain SK, Dar MY, Kumar S, Yadav A, Kearns SR. Role of anti-oxidant (vitamin-C) in post-operative pain relief in foot and ankle trauma surgery: A prospective randomized trial. Foot Ankle Surg. 2019 Aug 1;25(4):542–5. PubMed DOI: 1016/j.fas.2018.05.001
- Hung KC, Lin YT, Chen KH, Wang LK, Chen JY, Chang YJ, et al. The Effect of Perioperative Vitamin C on Postoperative Analgesic Consumption: A Meta-Analysis of Randomized Controlled Trials. Nutrients. 2020 Oct 1;12(10):1–17. PubMed DOI: 3390/nu12103109
- Vitamin C Premedication Reduces Postoperative Rescue Analgesic Requirement after Laparoscopic Surgeries. J Anesth Crit Care. 2016;5(1). DOI: 15406/jaccoa.2016.05.00175
- Aboelela M, Abedelkhalek M, Mamoun M. Role of vitamin C in multimodal analgesia for sleeve gastrectomy: a prospective randomized controlled trail. Anaesthesia, Pain & Intensive Care. 2023 Dec 1;27(1):82–8. DOI: 35975/apic.v27i1.2110
- Jeon Y, Park JS, Moon S, Yeo J. Effect of Intravenous High Dose Vitamin C on Postoperative Pain and Morphine Use after Laparoscopic Colectomy: A Randomized Controlled Trial. Pain Res Manag. 2016, 9147279, 6 pages, 2016. PubMed DOI: 1155/2016/9147279
- Moon S, Lim S, Yun J, Lee W, Kim M, Cho K, et al. Additional effect of magnesium sulfate and vitamin C in laparoscopic gynecologic surgery for postoperative pain management: a double-blind randomized controlled trial. Anesth Pain Med (Seoul). 2020 Jan 1;15(1):88–95. PubMed DOI: 17085/apm.2020.15.1.88
- Suter M, Bollen Pinto B, Belletti A, Putzu A. Efficacy and safety of perioperative vitamin C in patients undergoing noncardiac surgery: a systematic review and meta-analysis of randomised trials. Br J Anaesth. 2022 Apr 1;128(4):664–78. PubMed DOI: 1016/j.bja.2021.11.039
- Gan TJ, Habib AS, Miller TE, White W, Apfelbaum JL. Incidence, patient satisfaction, and perceptions of post-surgical pain: results from a US national survey. Curr Med Res Opin. 2014 Jan;30(1):149–60. PubMed DOI: 1185/03007995.2013.860019
- Gjeilo KH, Stenseth R, Klepstad P. Risk factors and early pharmacological interventions to prevent chronic postsurgical pain following cardiac surgery. Am J Cardiovasc Drugs. 2014 Oct 1;14(5):335–42. PubMed DOI: 1007/s40256-014-0083-2
- Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. 2006 May 13;367(9522):1618–25. PubMed DOI: 1016/S0140-6736(06)68700-X
- Ayatollahi V, Dehghanpour Farashah S, Behdad S, Vaziribozorg S, Rabbani Anari M. Effect of intravenous vitamin C on postoperative pain in uvulopalatopharyngoplasty with tonsillectomy. Clin Otolaryngol. 2017 Feb 1;42(1):139–43. PubMed DOI: 1111/coa.12684
- Chou R, Gordon DB, De Leon-Casasola OA, Rosenberg JM, Bickler S, Brennan T, et al. Management of Postoperative Pain: A Clinical Practice Guideline From the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016 Feb 1;17(2):131–57. PubMed DOI: 1016/j.jpain.2015.12.008