Anh Minh Nguyen 1, Phuoc Trong Do 2, Dung Hoang Tuan Bui 3, Viet-Thang Le 4
Author affiliations:
Background & objective: Neuropathic pain (NP) is perhaps one of the most difficult to manage pain syndrome. Pharmacologic management is often not successful and various other pain management modalities have to be used. This study evaluated the effectiveness of transcranial direct current stimulation (tDCS) targeting the dorsolateral prefrontal cortex (DLPFC) for treating NP, which followed Spinal Cord Injury (SCI).
Methodology: We enrolled 25 patients, 18-80 years old, from both genders, with NP, which followed SCI, From March 2023 to July 2024, in this retrospective study. Single-pulse transcranial magnetic stimulation (TMS) was used to locate the primary motor cortex (M1) and DLPFC regions before treatment. Patients received tDCS with the anode over the DLPFC and the cathode over the contralateral orbitofrontal (COF) region, with 2 mA current for 20 min daily, five days a week, for two weeks. Pain levels were assessed using Douleur Neuropathique 4 (DN4) and Numeric Rating Scale (NRS) before treatment, immediately after treatment, and then on monthly basis for three months. A responder was defined as a reduction of ≥ 2 points on the NRS or a 50% reduction in pain intensity. Additionally, side effects were diligently noted after each tDCS session.
Results: DN4 scores decreased from 5.8 to 3.3 and VAS from 5.9 to 3.2 after two weeks (P < 0.05). This effect lasted for two months post-treatment. Common side effects included drowsiness and transient scalp tingling, while less frequent side effects like headache, dizziness, and fatigue were mild and self-resolving.
Conclusion: tDCS stimulation on the DLPFC region can effectively alleviate NP symptoms after SCI in the short term, with no severe side effects. Further research is needed to optimize treatment regimens.
Abbreviations: COF: contralateral orbitofrontal, DN4: Douleur Neuropathique 4, DLPFC: dorsolateral prefrontal cortex, NP: Neuropathic pain, SCI: Spinal Cord Injury, tDCS: transcranial direct current stimulation, TMS: transcranial magnetic stimulation
Keywords: Neuropathic Pain; Spinal Cord Injury; Transcranial Direct Current Stimulation; Dorsolateral Prefrontal Cortex
Citation: Nguyen AM, Do PT, Bui DHT, Le VT. Transcranial direct current stimulation in spinal cord-injured patients with neuropathic pain. Anaesth. pain intensive care 2025;29(3):480-484.
DOI: 10.35975/apic.v29i3.2761
Received: February 23, 2025; Revised: March 04, 2025; Accepted: March 05, 2025
NP is a prevalent and debilitating condition affecting approximately 40% of patients following SCI1. The pathophysiology of NP involves complex mechanisms, including aberrant nociceptive signaling and central sensitization, which contribute to the chronic and often refractory nature of this pain.1 Traditional pharmacological treatments, such as opioids and anticonvulsants, often provide limited relief and are associated with significant side effects.2 Therefore, there is a growing interest in non-invasive neuromodulation techniques, such as tDCS, as potential therapeutic options for NP management.3-8
tDCS is a form of non-invasive brain stimulation that modulates cortical excitability through the application of a low-intensity direct current via electrodes placed on the scalp. The DLPFC has been identified as a key target for tDCS in the treatment of NP due to its role in pain modulation and cognitive-emotional processing of pain. Previous studies have demonstrated that tDCS targeting the DLPFC can significantly reduce pain intensity and improve quality of life in patients with chronic pain conditions, including NP following SCI.
Recent research has further elucidated the mechanisms underlying the analgesic effects of tDCS. It has been shown that tDCS can modulate the activity of the prefrontal cortex and its connectivity with other brain regions involved in pain perception, such as the thalamus and somatosensory cortex. Additionally, tDCS has been found to influence the release of neurotransmitters and neurotrophic factors, which may contribute to its pain-relieving effects. These findings suggest that tDCS not only provides symptomatic relief but also targets the underlying neural mechanisms of NP.
Despite the promising results, there is still a need for further research to optimize tDCS protocols and determine the long-term efficacy and safety of this intervention. Our study aims to build on previous findings by investigating the effects of tDCS targeting the DLPFC on NP in patients with SCI. Specifically, we aim to assess the impact of tDCS on pain intensity as well as to explore potential predictors of treatment response.
2.1. Patient selection
It was a retrospective study. All patients were 18 to 80 years old, met the diagnostic criteria for SCI with NP status and were treated from March 2023 to July 2024. We included patients diagnosed with SCI at least 1 month prior; with an exact cause of SCI (trauma, myelitis or spinal cord surgery); clinical manifestations consistent with the definitions of quadriplegia and/or hemiplegia in the “International Standards for Neurological Classification of SCI” (2011); sensory and motor impairment at or below the level of injury and evidence of SCI on magnetic resonance imaging.
Patients had NP at or below the level of SCI with an average NRS score ≥4 and stable in the past week (Diagnostic classification criteria recommended by the International Association for the Study of Pain in 2008).
Exclusion criteria included patients with metal implants in the head (except dental implants); patients with epilepsy, cognitive disturbances, inability to cooperate, unstable vital signs, a history of unmanaged substance abuse or acute dermatological condition affecting the scalp.
2.2. tDCS Therapy Administration
Patients from the outpatient clinic, after being evaluated by pain specialists and determined to meet the above criteria for participating in the study, were explained the proceedings. In a designated treatment room, metallic items were removed from their head and face, donning a head-hugging fabric hat.
M1 region was identified as the site eliciting the strongest contralateral thumb interstitial muscle contraction with the lowest stimulation intensity of TMS machine.
DLPFC position was determined by 5 cm anterior shifting from the M1 position and parallel to the midline and marked on the hat using a non-fading pen.
Trained physicians administered tDCS using a programmable, battery-powered constant current generator (Neurostim, Neurosoft LLC®, 5, Voronin str., Ivanovo, 153032, Russia). Carbon rubber electrodes, embedded in porous bags (both 5 × 7 cm), soaked in isotonic saline and coated with conductive ultrasound gel (to minimize scalp/hair resistance <10 kOhms), delivered electrical stimulation.
All patients received stimulation from the positive (anode) electrode placed at the DLPFC position of the dominant hemisphere based on the prior random group assignment, the cathode was positioned at the COF location. The intensity was 2 mA for 20 min, with a start/end ramp time of 20 sec (current density 0.057 mA/cm2, total daily charge 68.6 mC/cm2). Stimulation was administered continuously, five days per week, for two consecutive weeks.
All patients were then evaluated and recorded for treatment effectiveness as well as post-procedure conditions at various time points.
2.3. Outcome Measures
Baseline data, including patient characteristics, such as gender, age, duration of symptoms (months), injury level (cervical, thoracic or lumbar spine), causes (trauma, spinal cord surgery or myelitis), grade of injury (complete or incomplete), were systematically collected.
DN4 and NRS scores are 2 primary outcome measures and were assessed at 2 weeks post-treatment and then monthly during 3 months follow-up period. A responder was defined as a reduction of ≥ 2 points on the NRS or a 50% reduction in pain intensity. Additionally, side effects were diligently reported after each tDCS session.
Throughout the study duration, patients received comprehensive care, health education and tailored rehabilitation treatments based on their individual conditions.
2.4. Statistical analysis
Descriptive statistics, including mean, standard deviation, median, interquartile range, frequency and percentage, were employed for data presentation. The association between patient characteristics and pain severity was assessed using Chi-squared tests, Fisher’s exact tests and Paired-Sample T-tests. All statistical tests were two-sided, with a significance level set at 0.05. For continuous data with a non-normal distribution before the operation, the Mann–Whitney U-test and Wilcoxon Signed Rank test were utilized. Data analysis was conducted using the SPSS 28.0 software (IBM Corp., Armonk, NY, USA).
3.1. Baseline characteristics
The study enrolled a total of 25 patients meeting the criteria for chronic NP after SCI. The mean age was 48.2 ± 11.8 years, males constituted 76% of the cohort, with 19 out of 25 patients. The cervical and thoracic cords were the predominant lesion locations, accounting for 56% and 36% of cases, respectively. Among these cases, 60% resulted from trauma, 24% from myelitis and the remaining 16% from surgery. The majority of cases involved incomplete damage, representing 64% of the total cohort (Table 1).
3.2. tDCS effects on NP
Before treatment, the mean DN4 score was 5.8 ± 2.5, indicating moderate to severe neuropathic pain. The VAS score was 5.9 ± 1.6, indicating a similar level of pain intensity. After treatment, significant improvement was observed, with the DN4 score decreasing to 3.3 ± 2.3 and the VAS score to 3.2 ± 1.2. This indicates an immediate effect of tDCS on the pain intensity (Table 2).
At 2-month follow-up, both scores increased slightly, while remaining statistically significant differences from the baseline values (P < 0.05) (DN4: 4.0 ± 1.4, VAS: 4.2 ± 1.3).
Three months after treatment: DN4 and VAS scores returned to pre-treatment levels and there was no significant difference from baseline (P = 0.4).
The proportion of patients showing a ≥ 2-point reduction in VAS or a 50% reduction in pain intensity was 72% immediately after treatment, 60% at 1-month follow-up, 52% at 2-month follow-up and 36% at 3-month follow-up (Table 2).
3.3. Side effects
The most common side effects were sleepiness and a transient tingling sensation on the scalp (40% and 32%, respectively). Other common side effects such as headache, dizziness and fatigue accounted for less than 20%. Almost, all side effects were mild and recovered on their own (Figure 1).
The mechanisms underlying the analgesic effects of tDCS are thought to involve modulation of cortical excitability and connectivity between the prefrontal cortex and other brain regions involved in pain perception, such as the thalamus and somatosensory cortex.5,6,7 Additionally, tDCS has been shown to influence the release of neurotransmitters and neurotrophic factors, which may contribute to its pain-relieving effects.
These findings suggest that tDCS not only provides symptomatic relief but also targets the underlying neural mechanisms of NP. The comparable efficacy observed between DLPFC and M1 stimulation in this study was particularly intriguing. While M1 has been a traditional target for pain modulation, the DLPFC's role in pain processing, possibly through the modulation of affective and cognitive dimensions of pain, warrants further exploration. A study utilizing positron emission tomography revealed that tDCS over the M1 led to metabolic changes in brain regions associated with pain processing, including the DLPFC.6 This interplay between cortical areas may explain the similar analgesic outcomes observed with DLPFC stimulation.
The findings of our study demonstrate that tDCS targeting the DLPFC significantly reduces NP in patients with SCI. The observed reduction in pain scores, as measured by the DN4 and NRS scores, aligns with previous research indicating the efficacy of tDCS in managing NP.9 Specifically, the DN4 scores decreased from 5.8 to 3.3, and the NRS scores from 5.9 to 3.2 after two weeks of treatment, with effects maintained for up to two months post-treatment.
Comparing these results with other international studies, our findings are consistent with those reported by Soler et al. (2010), who found that tDCS combined with visual illusion significantly reduced NP in SCI patients.1 Similarly, Fregni et al. (2006) demonstrated that tDCS targeting the motor cortex effectively alleviated central pain in traumatic SCI patients.3 These studies support the potential of tDCS as a non-invasive and effective treatment modality for NP.
However, the delayed onset of analgesia noted in some studies raises questions about the optimal tDCS protocol. For instance, research has shown that repeated tDCS sessions are necessary to achieve long-lasting pain reduction, with significant effects emerging after multiple sessions.10 This underscores the importance of treatment duration and frequency in clinical applications. Moreover, the combination of tDCS with adjunct therapies, such as exercise, has been explored to enhance treatment outcomes. A pilot trial investigating tDCS followed by upper body exercise in chronic SCI patients reported pain intensity reductions, though the combined approach did not significantly outperform exercise alone.11 These findings suggest that while tDCS holds promise, its integration into multimodal treatment strategies requires further investigation.
Our study also highlights the safety and tolerability of tDCS, with most side effects being mild and self-resolving. Common side effects included drowsiness and transient tingling sensations of the scalp, which were consistent with those reported in other studies. This reinforces the feasibility of tDCS as a safe intervention for NP management.
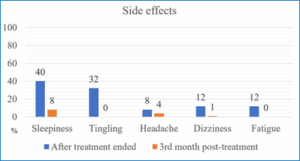
Figure 1: Side effects of tDCS treatment
Despite the promising results, our study has some limitations. The sample size was relatively small, and the follow-up period was limited to three months. Future research should focus on larger, randomized controlled trials with longer follow-up periods to better assess the long-term efficacy and safety of tDCS. Additionally, exploring different stimulation parameters and identifying predictors of treatment response could further optimize tDCS protocols for NP management.
In summary, tDCS was applied on the DLPFC using a 2 mA anodal current for 20 min daily, over 5 days per week for 2 consecutive weeks and demonstrates the potential for short-term reduction of NP symptoms following SCI within relatively safe parameters. Future investigations should focus on broader studies with increased sample sizes and diverse protocols to rigorously assess the safety and efficacy of tDCS, aiming for sustained medium- and longer-term relief from pain.
7. Ethical Considerations
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Science Council of University Medical Center, Ho Chi Minh City (Approval No. 67/2023/HĐ-ĐHYD), dated 30/03/2023.
8. Data availability
All data generated or analyzed during this study are included in this published article.
9. Funding
No funding was received for this research.
10. Competing interests
The authors declare that there was no competing interest in this study.
11. Authors contribution
AMN, PTD, VL: conceived the experiments, analyzed the results, wrote the main manuscript
DTHB: conducted the experiments, prepared figures and tables, drafted the manuscript
Author affiliations:
- Anh Minh Nguyen, Faculty of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam / Pain Management Unit, Department of Neurosurgery, University Medical Center Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam; Email: nm@umc.edu.vn, {ORCID:0000-0001-6451-2839}
- Phuoc Trong Do, Faculty of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam / Pain Management Unit, Department of Neurosurgery, University Medical Center Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam; Email: dt@umc.edu.vn, {ORCID:0000-0003-1831-1942}
- Dung Hoang Tuan Bui, Pain Management Unit, Department of Neurosurgery, University Medical Center Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam; Email: bht@umc.edu.vn, {ORCID:0009-0006-9753-6398}
- Viet-Thang Le, Faculty of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam / Pain Management Unit, Department of Neurosurgery, University Medical Center Ho Chi Minh City, Ho Chi Minh City, 700000, Vietnam; Email: tlv@umc.edu.vn, {ORCID:0000-0002-4702-7214}
ABSTRACT
Background & objective: Neuropathic pain (NP) is perhaps one of the most difficult to manage pain syndrome. Pharmacologic management is often not successful and various other pain management modalities have to be used. This study evaluated the effectiveness of transcranial direct current stimulation (tDCS) targeting the dorsolateral prefrontal cortex (DLPFC) for treating NP, which followed Spinal Cord Injury (SCI).
Methodology: We enrolled 25 patients, 18-80 years old, from both genders, with NP, which followed SCI, From March 2023 to July 2024, in this retrospective study. Single-pulse transcranial magnetic stimulation (TMS) was used to locate the primary motor cortex (M1) and DLPFC regions before treatment. Patients received tDCS with the anode over the DLPFC and the cathode over the contralateral orbitofrontal (COF) region, with 2 mA current for 20 min daily, five days a week, for two weeks. Pain levels were assessed using Douleur Neuropathique 4 (DN4) and Numeric Rating Scale (NRS) before treatment, immediately after treatment, and then on monthly basis for three months. A responder was defined as a reduction of ≥ 2 points on the NRS or a 50% reduction in pain intensity. Additionally, side effects were diligently noted after each tDCS session.
Results: DN4 scores decreased from 5.8 to 3.3 and VAS from 5.9 to 3.2 after two weeks (P < 0.05). This effect lasted for two months post-treatment. Common side effects included drowsiness and transient scalp tingling, while less frequent side effects like headache, dizziness, and fatigue were mild and self-resolving.
Conclusion: tDCS stimulation on the DLPFC region can effectively alleviate NP symptoms after SCI in the short term, with no severe side effects. Further research is needed to optimize treatment regimens.
Abbreviations: COF: contralateral orbitofrontal, DN4: Douleur Neuropathique 4, DLPFC: dorsolateral prefrontal cortex, NP: Neuropathic pain, SCI: Spinal Cord Injury, tDCS: transcranial direct current stimulation, TMS: transcranial magnetic stimulation
Keywords: Neuropathic Pain; Spinal Cord Injury; Transcranial Direct Current Stimulation; Dorsolateral Prefrontal Cortex
Citation: Nguyen AM, Do PT, Bui DHT, Le VT. Transcranial direct current stimulation in spinal cord-injured patients with neuropathic pain. Anaesth. pain intensive care 2025;29(3):480-484.
DOI: 10.35975/apic.v29i3.2761
Received: February 23, 2025; Revised: March 04, 2025; Accepted: March 05, 2025
1. INTRODUCTION
NP is a prevalent and debilitating condition affecting approximately 40% of patients following SCI1. The pathophysiology of NP involves complex mechanisms, including aberrant nociceptive signaling and central sensitization, which contribute to the chronic and often refractory nature of this pain.1 Traditional pharmacological treatments, such as opioids and anticonvulsants, often provide limited relief and are associated with significant side effects.2 Therefore, there is a growing interest in non-invasive neuromodulation techniques, such as tDCS, as potential therapeutic options for NP management.3-8
tDCS is a form of non-invasive brain stimulation that modulates cortical excitability through the application of a low-intensity direct current via electrodes placed on the scalp. The DLPFC has been identified as a key target for tDCS in the treatment of NP due to its role in pain modulation and cognitive-emotional processing of pain. Previous studies have demonstrated that tDCS targeting the DLPFC can significantly reduce pain intensity and improve quality of life in patients with chronic pain conditions, including NP following SCI.
Recent research has further elucidated the mechanisms underlying the analgesic effects of tDCS. It has been shown that tDCS can modulate the activity of the prefrontal cortex and its connectivity with other brain regions involved in pain perception, such as the thalamus and somatosensory cortex. Additionally, tDCS has been found to influence the release of neurotransmitters and neurotrophic factors, which may contribute to its pain-relieving effects. These findings suggest that tDCS not only provides symptomatic relief but also targets the underlying neural mechanisms of NP.
Despite the promising results, there is still a need for further research to optimize tDCS protocols and determine the long-term efficacy and safety of this intervention. Our study aims to build on previous findings by investigating the effects of tDCS targeting the DLPFC on NP in patients with SCI. Specifically, we aim to assess the impact of tDCS on pain intensity as well as to explore potential predictors of treatment response.
2. METHODOLOGY
2.1. Patient selection
It was a retrospective study. All patients were 18 to 80 years old, met the diagnostic criteria for SCI with NP status and were treated from March 2023 to July 2024. We included patients diagnosed with SCI at least 1 month prior; with an exact cause of SCI (trauma, myelitis or spinal cord surgery); clinical manifestations consistent with the definitions of quadriplegia and/or hemiplegia in the “International Standards for Neurological Classification of SCI” (2011); sensory and motor impairment at or below the level of injury and evidence of SCI on magnetic resonance imaging.
Patients had NP at or below the level of SCI with an average NRS score ≥4 and stable in the past week (Diagnostic classification criteria recommended by the International Association for the Study of Pain in 2008).
Exclusion criteria included patients with metal implants in the head (except dental implants); patients with epilepsy, cognitive disturbances, inability to cooperate, unstable vital signs, a history of unmanaged substance abuse or acute dermatological condition affecting the scalp.
2.2. tDCS Therapy Administration
Patients from the outpatient clinic, after being evaluated by pain specialists and determined to meet the above criteria for participating in the study, were explained the proceedings. In a designated treatment room, metallic items were removed from their head and face, donning a head-hugging fabric hat.
M1 region was identified as the site eliciting the strongest contralateral thumb interstitial muscle contraction with the lowest stimulation intensity of TMS machine.
DLPFC position was determined by 5 cm anterior shifting from the M1 position and parallel to the midline and marked on the hat using a non-fading pen.
Trained physicians administered tDCS using a programmable, battery-powered constant current generator (Neurostim, Neurosoft LLC®, 5, Voronin str., Ivanovo, 153032, Russia). Carbon rubber electrodes, embedded in porous bags (both 5 × 7 cm), soaked in isotonic saline and coated with conductive ultrasound gel (to minimize scalp/hair resistance <10 kOhms), delivered electrical stimulation.
All patients received stimulation from the positive (anode) electrode placed at the DLPFC position of the dominant hemisphere based on the prior random group assignment, the cathode was positioned at the COF location. The intensity was 2 mA for 20 min, with a start/end ramp time of 20 sec (current density 0.057 mA/cm2, total daily charge 68.6 mC/cm2). Stimulation was administered continuously, five days per week, for two consecutive weeks.
All patients were then evaluated and recorded for treatment effectiveness as well as post-procedure conditions at various time points.
2.3. Outcome Measures
Baseline data, including patient characteristics, such as gender, age, duration of symptoms (months), injury level (cervical, thoracic or lumbar spine), causes (trauma, spinal cord surgery or myelitis), grade of injury (complete or incomplete), were systematically collected.
DN4 and NRS scores are 2 primary outcome measures and were assessed at 2 weeks post-treatment and then monthly during 3 months follow-up period. A responder was defined as a reduction of ≥ 2 points on the NRS or a 50% reduction in pain intensity. Additionally, side effects were diligently reported after each tDCS session.
Throughout the study duration, patients received comprehensive care, health education and tailored rehabilitation treatments based on their individual conditions.
2.4. Statistical analysis
Descriptive statistics, including mean, standard deviation, median, interquartile range, frequency and percentage, were employed for data presentation. The association between patient characteristics and pain severity was assessed using Chi-squared tests, Fisher’s exact tests and Paired-Sample T-tests. All statistical tests were two-sided, with a significance level set at 0.05. For continuous data with a non-normal distribution before the operation, the Mann–Whitney U-test and Wilcoxon Signed Rank test were utilized. Data analysis was conducted using the SPSS 28.0 software (IBM Corp., Armonk, NY, USA).
3. RESULTS
3.1. Baseline characteristics
The study enrolled a total of 25 patients meeting the criteria for chronic NP after SCI. The mean age was 48.2 ± 11.8 years, males constituted 76% of the cohort, with 19 out of 25 patients. The cervical and thoracic cords were the predominant lesion locations, accounting for 56% and 36% of cases, respectively. Among these cases, 60% resulted from trauma, 24% from myelitis and the remaining 16% from surgery. The majority of cases involved incomplete damage, representing 64% of the total cohort (Table 1).
| Table 1: Characteristics of NP and patients with NP | |
| Variables | N = 25 |
| Age (Year) | 48.2 ± 11.8 |
| Gender
· Male · Female |
19 (76) 6 (24) |
| Duration of symptoms (Month) | 38.6 ± 10.7 |
| Injury level:
· Cervical · Thoracic · Lumbar |
14 (56) 9 (36) 2 (8) |
| Causes:
· Trauma · Myelitis · Surgery |
15 (60) 6 (24) 4 (16) |
| Grade of injury:
· Complete · Incomplete |
9 (36) 16 (64) |
| Data presented as mean ± SD or n (%) | |
3.2. tDCS effects on NP
Before treatment, the mean DN4 score was 5.8 ± 2.5, indicating moderate to severe neuropathic pain. The VAS score was 5.9 ± 1.6, indicating a similar level of pain intensity. After treatment, significant improvement was observed, with the DN4 score decreasing to 3.3 ± 2.3 and the VAS score to 3.2 ± 1.2. This indicates an immediate effect of tDCS on the pain intensity (Table 2).
At 2-month follow-up, both scores increased slightly, while remaining statistically significant differences from the baseline values (P < 0.05) (DN4: 4.0 ± 1.4, VAS: 4.2 ± 1.3).
Three months after treatment: DN4 and VAS scores returned to pre-treatment levels and there was no significant difference from baseline (P = 0.4).
The proportion of patients showing a ≥ 2-point reduction in VAS or a 50% reduction in pain intensity was 72% immediately after treatment, 60% at 1-month follow-up, 52% at 2-month follow-up and 36% at 3-month follow-up (Table 2).
| Table 2: Main outcomes and response rates after tDCS treatment. | |||||
| Variables | DN4 score | P-value | VAS score | P-value | Response Rate (%) |
| Before treatment | 5.8 ± 2.5 | 5.9 ± 1.6 | - | ||
| After treatment ended | 3.3 ± 2.3 | 0.001 | 3.2 ± 1.2 | 0.000 | 72 |
| 1st month post-treatment | 3.6 ± 1.9 | 0.001 | 3.6 ± 1.2 | 0.000 | 60 |
| 2nd month post-treatment | 4.0 ± 1.4 | 0.003 | 4.2 ± 1.3 | 0.000 | 52 |
| 3rd month post-treatment | 5.3 ± 1.6 | 0.4 | 5.5 ± 1.7 | 0.4 | 36 |
| Data presented as mean ± SD | |||||
3.3. Side effects
The most common side effects were sleepiness and a transient tingling sensation on the scalp (40% and 32%, respectively). Other common side effects such as headache, dizziness and fatigue accounted for less than 20%. Almost, all side effects were mild and recovered on their own (Figure 1).
4. DISCUSSION
The mechanisms underlying the analgesic effects of tDCS are thought to involve modulation of cortical excitability and connectivity between the prefrontal cortex and other brain regions involved in pain perception, such as the thalamus and somatosensory cortex.5,6,7 Additionally, tDCS has been shown to influence the release of neurotransmitters and neurotrophic factors, which may contribute to its pain-relieving effects.
These findings suggest that tDCS not only provides symptomatic relief but also targets the underlying neural mechanisms of NP. The comparable efficacy observed between DLPFC and M1 stimulation in this study was particularly intriguing. While M1 has been a traditional target for pain modulation, the DLPFC's role in pain processing, possibly through the modulation of affective and cognitive dimensions of pain, warrants further exploration. A study utilizing positron emission tomography revealed that tDCS over the M1 led to metabolic changes in brain regions associated with pain processing, including the DLPFC.6 This interplay between cortical areas may explain the similar analgesic outcomes observed with DLPFC stimulation.
The findings of our study demonstrate that tDCS targeting the DLPFC significantly reduces NP in patients with SCI. The observed reduction in pain scores, as measured by the DN4 and NRS scores, aligns with previous research indicating the efficacy of tDCS in managing NP.9 Specifically, the DN4 scores decreased from 5.8 to 3.3, and the NRS scores from 5.9 to 3.2 after two weeks of treatment, with effects maintained for up to two months post-treatment.
Comparing these results with other international studies, our findings are consistent with those reported by Soler et al. (2010), who found that tDCS combined with visual illusion significantly reduced NP in SCI patients.1 Similarly, Fregni et al. (2006) demonstrated that tDCS targeting the motor cortex effectively alleviated central pain in traumatic SCI patients.3 These studies support the potential of tDCS as a non-invasive and effective treatment modality for NP.
However, the delayed onset of analgesia noted in some studies raises questions about the optimal tDCS protocol. For instance, research has shown that repeated tDCS sessions are necessary to achieve long-lasting pain reduction, with significant effects emerging after multiple sessions.10 This underscores the importance of treatment duration and frequency in clinical applications. Moreover, the combination of tDCS with adjunct therapies, such as exercise, has been explored to enhance treatment outcomes. A pilot trial investigating tDCS followed by upper body exercise in chronic SCI patients reported pain intensity reductions, though the combined approach did not significantly outperform exercise alone.11 These findings suggest that while tDCS holds promise, its integration into multimodal treatment strategies requires further investigation.
Our study also highlights the safety and tolerability of tDCS, with most side effects being mild and self-resolving. Common side effects included drowsiness and transient tingling sensations of the scalp, which were consistent with those reported in other studies. This reinforces the feasibility of tDCS as a safe intervention for NP management.

Figure 1: Side effects of tDCS treatment
5. LIMITATIONS
Despite the promising results, our study has some limitations. The sample size was relatively small, and the follow-up period was limited to three months. Future research should focus on larger, randomized controlled trials with longer follow-up periods to better assess the long-term efficacy and safety of tDCS. Additionally, exploring different stimulation parameters and identifying predictors of treatment response could further optimize tDCS protocols for NP management.
6. CONCLUSION
In summary, tDCS was applied on the DLPFC using a 2 mA anodal current for 20 min daily, over 5 days per week for 2 consecutive weeks and demonstrates the potential for short-term reduction of NP symptoms following SCI within relatively safe parameters. Future investigations should focus on broader studies with increased sample sizes and diverse protocols to rigorously assess the safety and efficacy of tDCS, aiming for sustained medium- and longer-term relief from pain.
7. Ethical Considerations
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Science Council of University Medical Center, Ho Chi Minh City (Approval No. 67/2023/HĐ-ĐHYD), dated 30/03/2023.
8. Data availability
All data generated or analyzed during this study are included in this published article.
9. Funding
No funding was received for this research.
10. Competing interests
The authors declare that there was no competing interest in this study.
11. Authors contribution
AMN, PTD, VL: conceived the experiments, analyzed the results, wrote the main manuscript
DTHB: conducted the experiments, prepared figures and tables, drafted the manuscript
12. REFERENCES
- Soler MD, Kumru H, Pelayo R, Vidal J, Tormos JM, Fregni F, et al. Effectiveness of transcranial direct current stimulation and visual illusion on neuropathic pain in spinal cord injury. Brain. 2010;133(9):2565–77. PubMed DOI: 1093/brain/awq184
- Furlan JC, Sakakibara BM, Miller WC, Krassioukov AV. Global incidence and prevalence of traumatic spinal cord injury. Can J Neurol Sci. 2013;40(4):456–64. PubMed DOI: 1017/s0317167100014530
- Fregni F, Boggio PS, Lima MC, Ferreira MJ, Wagner T, Rigonatti SP, et al. A sham-controlled, phase II trial of transcranial direct current stimulation for the treatment of central pain in traumatic spinal cord injury. Pain. 2006;122(1–2):197–209. PubMed DOI: 1016/j.pain.2006.02.023
- Nitsche MA, Paulus W. Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology. 2001;57:1899–901. PubMed DOI: 1212/wnl.57.10.1899
- Lorenz J, Minoshima S, Casey KL. Keeping pain out of mind: the role of the dorsolateral prefrontal cortex in pain modulation. Brain. 2003;126(5):1079–91. PubMed DOI: 1093/brain/awg102
- Yoon EJ, Kim YK, Kim HR, Kim SE, Lee Y, Shin HI. Transcranial direct current stimulation to lessen neuropathic pain after spinal cord injury: a mechanistic PET study. Neurorehabil Neural Repair. 2014;28:250–9. PubMed DOI: 1177/1545968313507632
- Vaseghi B, Zoghi M, Jaberzadeh S. Differential effects of cathodal transcranial direct current stimulation of prefrontal, motor and somatosensory cortices on cortical excitability and pain perception: a double-blind randomised sham-controlled study. Eur J Neurosci. 2015;42:2426–37. PubMed DOI: 1111/ejn.13043
- DosSantos MF, Love TM, Martikainen IK, Nascimento TD, Fregni F, Cummiford C, et al. Immediate effects of tDCS on the μ-opioid system of a chronic pain patient. Front Psychiatry. 2012;3:93. PubMed DOI: 3389/fpsyt.2012.00093
- Soler MD, Kumru H, Pelayo R, Vidal J, Tormos JM, Fregni F, et al. Effectiveness of transcranial direct current stimulation and visual illusion on neuropathic pain in spinal cord injury. Brain. 2010;133(9):2565–77. PubMed DOI: 1093/brain/awq184
- Thibaut A, Carvalho S, Morse LR, Zafonte R, Fregni F. Delayed pain decrease following M1 tDCS in spinal cord injury: a randomized controlled clinical trial. Neurosci Lett. 2017;658:19–26. PubMed DOI: 1016/j.neulet.2017.08.024
- Yeh NC, Yang YR, Huang SF, Ku PH, Wang RY. Effects of transcranial direct current stimulation followed by exercise on neuropathic pain in chronic spinal cord injury: a double-blinded randomized controlled pilot trial. Spinal Cord. 2021;59(6):684–92. PubMed DOI: 1038/s41393-020-00560-x