Huda A. Owaid 1, Abdulkareem M. Jewad 2
Authors affiliations:
Background & objectives: Breast cancer is a major worldwide health issue. A delayed and often inadequate diagnosis results in the death of many women. As a result, it is beneficial to conduct research and analyze new markers for an early diagnosis and surveillance of the breast cancer. This study aimed to examine the following novel diagnostic biomarkers in the serum of women with breast cancer: Trefoil Factor 1 (TFF1), Cytokeratin 21-1 (CYRA21-1), and matrix metalloproteinase-9 (MMP-9) to establish any relationship in between.
Methodology: This case-control study comprised of 72 women diagnosed with breast cancer. The participants were distributed as follows: pretreatment group (n = 15) and a post-treatment group with chemotherapy composed of anthracycline, cyclophosphamide, and docetaxel (ACD) (n = 57). A total of 15 healthy women were included in the control group. Concentrations of TFF1, CYRA21-1, and MPP-9 were measured in all study groups.
Results: The results revealed significant increases in TFF1, CYRA21-1, and MPP-9 levels (P< 0.000) in all patient groups before and after chemotherapy compared to the control group. TFF1 levels also showed a significant positive correlation with MPP-9 levels.
Conclusion: This study identified TFF1, CYFRA 21-1, and MMP-9 as valuable diagnostic biomarkers for the diagnosis of breast cancer with differential response to chemotherapy. While CYFRA 21-1 and MMP-9 showed consistent diagnostic potential, TFF1 levels were partially reduced post-treatment.
Keywords: Biomarkers; Breast cancer; Chemotherapy; TFF1; CYRA21-1; MPP-9.
Citation: Owaid HA, Jewad AM. Assessment of trefoil factor 1, cytokeratin 21-1, and human matrix metalloproteinase 9 as novel diagnostic biomarkers for breast cancer. Anaesth. pain intensive care 2025;29(3):436-443. DOI: 10.35975/apic.v29i3.2685
Received: February 17, 2025; Revised: February 22, 2025; Accepted: March 18, 2025
Breast cancer is a major health issue worldwide. It is the most frequently diagnosed carcinoma globally and a primary cause of cancer-related deaths among women. In 2020, over 50 percent of breast cancer diagnoses and two-thirds of breast cancer-related deaths occurred in less developed regions, despite the disease's prior association with affluent nations.1–3 Therefore, diagnostic and prognostic assessments remain the primary preventive and treatment approaches for the spread of breast cancer. In the present study, we aimed to assess the diagnostic value of newly discovered biomarkers (TFF1, Cytokeratin 21-1, MMP9) to detect BC in the preliminary phase. Previous studies have shown their significance in the diagnosis of various types of cancer. Trefoil Factors are components of the mucus barrier and are found in exocrine bodily fluids, such as gastric juice, saliva, and breast milk. Urine and tears.4 Trefoil Factor-1 (TFF1) was first identified in the MCF-7 breast cancer cell line and is commonly found in gastric carcinomas and gastroduodenal mucosa. It also interacts with mucins and other members of the ECM, enhancing its protective role and playing a role in repair. Trefoil Factor-1 plays a crucial role in maintaining the structural integrity of epithelia, with anti-damage and repair-promoting effects on the tissue. They strengthen the mucosa and contribute to cell growth and movement, which are crucial for tissue repair.5 It has been identified as a biomarker of metastatic colon cancer.6 TFF1 is encoded by the Trefoil Factor1 gene, which is located on chromosome 21 in humans. TFF1 is predominantly expressed in the gastrointestinal tract, including the stomach, duodenum, and mammary gland. Nonetheless, TFF1 in triple-negative breast cancer has been investigated relatively rarely to date.7 TFF1 has also been implicated in gastric cancer. Under conditions of mucosal damage and inflammation, TFF1 can be induced. Thus, the function of TFF1 may be context-sensitive, depending on the type and stage of cancer.8
Cytokeratin 21-1 (CK 21-1) is a proteolytic degradation product of cytokeratin 19 (CK19), a structural component of epithelial cell intermediate filaments. Specifically, CK 21-1 represents soluble fragments of CK19 released into the systemic circulation during pathological processes,9 an intermediate filament protein of epithelial cells. This fragment is mainly employed in oncology to evaluate the overall progression of the disease as well as the response to therapy, especially in cancers that sample strongly for cytokeratin 19,10 Elevated levels of CYRA21-1 in the serum or blood can result from the breakdown of cytokeratin 19 in response to cellular damage or cancerous processes. Cytokeratin 21-1 serves as a marker for cancer diagnosis and monitoring. It is particularly useful for detecting the presence of tumor cells and tracking disease progression because of its release from epithelial cells during tumor invasion and metastasis. It is elevated in lung cancer,11 but is not a primary marker of prostate cancer.12 Cytokeratin 21-1 is useful in identifying epithelial cancers on a general basis because it originates from cytokeratin 19. However, their utility is cancer type- and stage-dependent. Analogously, according to the available studies, the utility of CYRA21-1 as a biomarker in gastric and colorectal cancers is still being assessed and is not as established as in breast and lung cancers.12,13
Human matrix metalloproteinase 9 (MMP9), also known as Gelatinase B, belongs to the type IV collagenase family. It is distinguished by its zinc content and participates in pathological processes that result in massive tissue dissolution, as well as physiological functions that involve the degradation of the tissue matrix. Because of the function of the enzyme, it is a particularly promising target for studying the mechanisms by which specific diseases and therapies drive tissue disintegration.14 Human matrix metalloproteinase 9 is encoded by the MMP9 gene, which is located on chromosome 20 in humans. This protein is synthesized as an inactive proenzyme (proMMP-9), which requires activation to become functionally active. Human matrix metalloproteinase 9 s produced primarily by neutrophils but also by other cell types, such as macrophages, fibroblasts, and cancer cells. It is released into the extracellular environment in response to stimuli, such as inflammation and tissue damage. Matrix metalloproteinase 9 isolated from humans has been implicated in the final step of tissue remodeling through the cleavage of type IV and type V collagen, which forms the basis of the basement membrane and Extracellular Matrix (ECM). This activity is crucial for tissue remodeling and repair. The exception is breaking down ECM boundaries, in which MMP-9 also promotes cell migration and invasion, which is essential for wound healing, but also for pathological conditions, such as cancer metastases.15 Elevated levels of MMP-9 are often linked to aggressive cancer behavior and poor prognosis in various cancers, including lung and prostate cancers.16,17 CYFRA 21-1 is mainly a detection marker indicative of tumor-induced degradation,9 whereas TFF1 is context-dependent and capable of suppressing and supporting tumorigenesis7. MMP-9 is an essential factor involved in cancer progression via several mechanisms.18 These biomarkers offer complementary insights into cancer biology, making them promising components of multi-marker panels for cancer diagnosis, prognostic evaluation, and therapeutic monitoring.7,9,18 Treatment with chemotherapy in women with breast cancer changed some measured biomarkers, such as oxidative stress-related biomarkers.19 We believe that it is worthwhile to study the above markers before and after chemotherapy to determine whether they affect the diagnostic ability of these novel biomarkers. The current study aimed to examine Trefoil Factor 1 (TFF1), Cytokeratin 21-1 (CYRA21-1), and matrix metalloproteinase-9 (MMP-9) as novel diagnostic biomarkers in the serum of women with breast cancer and a healthy control group.
This was a case-control study. This study included patients who visited the Oncology and Hematology Center/Tumor LAB Department of Al-Sadder Teaching Hospital in Basrah governorate; the sample was taken in the duration of December 2023 to May 2024. Diagnosis of breast cancer is based on The TNM stage (Tumor, Node, Metastasis) system, a globally standardized framework that categorizes malignancies based on their anatomical extent. This system classifies primary tumor size and invasiveness (T), regional lymph node involvement (N), and distant metastasis (M). The American Joint Committee on Cancer (AJCC) delineates breast cancer staging into two categories: anatomic staging, which relies solely on TNM criteria, and prognostic staging, which incorporates TNM alongside histopathological tumor grade and biomarker profiles (e.g., hormone receptor and HER2 status). The study population comprised 72 women with breast cancer divided into two categories: 15 before therapy (group 2) and 57 post treated with chemotherapy, which included 18 women at stage 1 (group 3), 32 at stage 2 (group 4), and 7 at stage 3 (group 5). Fifteen apparently healthy women served as the control group (Group 1).
Inclusion and Exclusion criteria
Patients with other chronic diseases (cardiovascular diseases, infections, and endocrine disorders) were excluded from the study.
Sample collection
Both patients and controls were dispensed in a gel tube and allowed to coagulate at room temperature for 10 min. Blood samples were centrifuged at 3000 rpm for 5 min. Subsequently, the sera were precipitated and stored at − 20 °C until further use. Serum evaluations of the three biomarkers were conducted utilizing ELISA (Enzyme-Linked Immunosorbent Assay (ELISA) with the following specific commercial kits: (TFF1, CYRA21-1, and MMP-9) (Elabscience, USA). All ELISA were performed according to the manufacturer's instructions.
Statistical analysis
T he collected data were entered into SPSS version 26 for tabulation and analyses. Variables were coded and defined such that categorical variables were presented as frequencies and percentages, whereas continuous variables were presented as means and standard deviations. The Kolmogorov test was used to examine the normality of the continuous variables. The Kruskal–Wallis test was used to investigate the differences in the variables across the five study groups. Pairwise comparisons were made for variables that showed significant statistical differences. Corresponding post-hoc tests were used to examine between-group differences. The level of significance was set at p < 0.05.
The present case-control study population comprised 72 women with breast cancer, divided into two categories: 15 before therapy (group 2) and 57 post-treated with chemotherapy, which comprised 18 women at stage 1 (group 3), 32 women at stage 2 (group 4), and 7 women at stage 3 (group 5). Fifteen healthy women were included in the control group (Group 1). Table 1 shows that There were no significant differences in age between the study groups (Table 1). TFF1 levels increased significantly in all patient groups post- and pre-treatment in comparison to the experimental control group (P < 0.0001), and the post-treatment group demonstrated significant decreases in comparison to the pre-treatment group, as shown in Table 1. CYFRA 21-1 levels in the same table were also much higher in all the patient groups, post- and pre-treatment groups, than in the control experimental group. However, the data presented in Table 1 demonstrate that MMP-9 levels were significantly elevated across all patient groups, including both the pre-treatment and post-treatment cohorts, when compared to the control or healthy group. However, a slight reduction in MMP-9 levels was observed, particularly in the stage 3 patient group, although these levels remained significantly higher than those in the control group. In summary, TFF1, CYFRA 21-1, and MMP-9 levels changed
significantly with disease progression and treatment (Table 1). (1), (2), and (3)
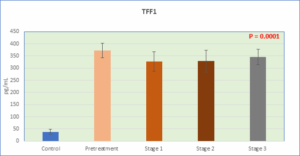
Figure 1: Level of TFF1 in serum in the study groups
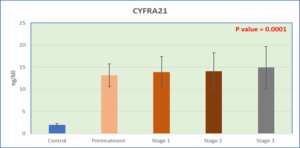
Figure 2: Level of CYRA21-1 in serum in the study groups
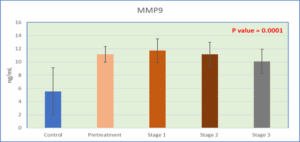
Figure 3: Level of MMP-9 in serum in the study groups correlation among the studied biomarkers:
Table 3 shows a moderate positive correlation between the TFF1 and MMP-9 levels (R= 0.412, P = 0.019).
The present study demonstrated a significant elevation in TFF1 levels across all patient cohorts following treatment compared to the experimental control group. Concurrently, a marked reduction in TFF1 levels was observed in the post-treatment groups, consistent with recent findings of elevated TFF1 expression in patients with BC. For instance, Lang et al. (2021) documented that TFF1 is overexpressed in BC tissues and that its expression levels are positively correlated with tumor grade and stage. These results confirm the initial hypothesis that TFF1 might be a candidate biomarker for BC progression. Moreover, their results also validated that a high level of TFF1 was generally associated with an increased cancer grade and more processive stage of BC tissues in comparison with adjacent normal tissue,20 which provides a rationale for exploring the prognostic implications of TFF1 in BC. A subsequent study by Yi et al. (2020) also identified TFF1 as an early cancer biomarker. TFF1 levels were significantly higher in patients with early-stage BC than in healthy control subjects. This indicates that TFF1 could be a promising biomarker for the diagnosis of early stage cancer and is helpful for cancer multiplexing.21
TFF1 is widely recognized for its critical role in mucosal protection and repair. In cancer biology, TFF1 upregulation could be linked to tumor adaptive mechanisms used to protect it against the surrounding microenvironment or to form a specific niche that favors cancer progression. Interestingly, new data suggest that TFF1 is involved in the epithelial-mesenchymal transition (EMT), a process directly correlated with cancer metastasis, thereby strengthening this working hypothesis. High expression levels of TFF1 could reflect the tumor’s attempt to create a protective microenvironment, thus facilitating its development. The repeated finding that TFF1 is upregulated in a range of tumors points to its possible application as a diagnostic or prognostic biomarker in cancer.22
However, some studies have reported findings inconsistent with the current results, which demonstrated elevated TFF1 levels across all tested cases. These studies found no significant difference in TFF1 levels between cancer patients and normal controls, suggesting that TFF1 may not serve as a reliable biomarker for all molecular subtypes of breast cancer. Furthermore, it has been proposed that TFF1 expression may be influenced by disease stage of the disease.23 Additionally, different authors have reported discrepancies in circulating TFF1 levels based on factors including both the hormone receptor status of their patients and the treatments administered to them. Wang et al. Shunyi et al. (2022) suggested that TFF1 expression is modulated by estrogen receptor-positive status, which complicates its further application as a consistent biomarker in BC.24
The study showed that CYFRA 21-1 levels were significantly higher in all patient groups than in the control group, both pre- and post-treatment. These results confirm those of previous studies on the importance of CYFRA 21–1 in BC, all of which found elevated levels in patients with more advanced forms of the disease. These data suggest that CYFRA 21-1 is a potential biomarker of disease progression and treatment response.25 Moreover, another study showed that the levels of CYFRA 21-1 were markedly elevated in patients with breast cancer compared to those in healthy individuals and correlated positively with tumor stage. They stated that although cytokeratin fragments 21-1 might not be an evolutionary biomarker, when used simultaneously with different biomarkers, it holds huge diagnostic worth.26 Additionally, previous research has highlighted notable differences in CYFRA 21-1 levels between cancer patients and controls, particularly in individuals with advanced disease, underscoring its potential utility in monitoring disease status and progression.27
CYFRA 21-1 is distinguished by its epitope, which is a polypeptide typically released following cellular death. Elevated serum titers of CYFRA 21-1 have been detected in various malignancies, particularly lung cancer; however, healthy individuals rarely exhibit abnormal serum levels of this marker.28 In breast cancer patients, elevated CYFRA 21-1 levels may result from increased cellular turnover and cytokeratin breakdown, a phenomenon also observed in other cancers. Higher CYFRA 21-1 levels in advanced breast cancer stages may reflect a greater tumor burden and more aggressive disease, consistent with its role in other malignancies, where elevated levels are associated with advanced disease progression.26
These findings contrast with those of previous research, suggesting that CYFRA 21-1 has not yet been established as a standard biomarker for breast cancer.29 For instance, Filella et al. (2023) reported that while some breast cancer patients exhibited elevated CYFRA 21-1 levels, variability in sensitivity and specificity limited its clinical utility. The study concluded that although CYFRA 21-1 may provide supplementary diagnostic information, it should not replace established biomarkers.30
This study demonstrated elevated MMP-9 levels across all patient groups, with a modest reduction observed in stage 3 patients. Despite this decrease, the MMP-9 levels remained significantly higher than those in the control group. These findings align with those of prior studies, indicating that elevated MMP-9 levels are associated with the early stages of BC and may serve as a potential biomarker for disease progression. Increased MMP-9 levels may reflect active remodeling of the extracellular matrix (ECM) during the initial phases of tumorigenesis.31 Joseph et al. MMP-9 also plays a critical role in the late stage of breast cancer progression, and high plasma MMP-9 levels are correlated with the initial stage of breast cancer and have been associated with poor prognosis.32 These findings are also supported by previous reports, which confirmed that upregulated levels of MMP-9 assure a higher level of cellular proliferative activity. Together, these results indicate that MMP-9, which is expressed in both stromal and tumor cells, contributes to BC progression. MMP-9 plays a decisive role in ECM remodeling, which is especially active during the early stages of tumorigenesis when the tumor adapts to invade the surrounding tissue. Cumulatively, high serum MMP-9 levels may also indicate the pronounced ECM restructuring required for tumor progression and invasion. Therefore, MMP-9 levels in early breast cancer may be an important tool for early detection and prognosis as well as for the stratification of patients according to their risk profile.33
Jiang and Li (2021) found a decrease in MMP-9 levels in certain subtypes of advanced-stage breast cancer. They suggested that this reduction could be a consequence of modifications in the tumor microenvironment or of adaptive behaviors adopted by cancer cells to counteract excessive ECM degradation, leading to a different protein degradation profile in more advanced stages.34 It is hypothesized that during advanced tumor development, tumors may alter the proteolytic/anti-proteolytic balance. The observed decrease in MMP-9 levels could also be attributed to compensatory mechanisms or changes in other components of the tissue microenvironment, which may not directly reflect a reduction in proteolytic activity but rather a shift in enzyme functionality. Breast cancer is a highly heterogeneous disease, and different subtypes of breast cancer may exhibit varying MMP-9 concentrations.31
The findings of this study are consistent with prior research that demonstrated a significant association between TFF1 and MMP-9 expression levels, aggressive breast cancer phenotypes, and poor clinical outcomes. Specifically, a significant p-value was observed for both biomarkers, indicating their potential role in tumor progression. These results align with earlier studies that reported a significant correlation between TFF1 and MMP-9 levels, suggesting a possible interplay wherein TFF1 may modulate MMP-9 activity, or vice versa, thereby contributing to cancer progression.35 The results are also consistent with the study by Manfioletti and Fedele (2023), who identified a good association between TFF1 with MMP-9 positive breast cancer tissues, especially in those with more aggressive and metastatic phenotypes. This supports the hypothesis that TFF1 is mechanistically related to MMP-9 in promoting aggressive cancer behavior.36
However, this study contradicts previous research that showed a negative correlation between TFF1 and MMP-9. For example, some studies have found that MMP-9 is a strong predictor of poor prognosis, but that TFF1 and MMP-9 are not significantly correlated. These studies suggest that the involvement of TFF1 in breast cancer may be modulated through interactions with other biomarkers or signaling pathways, rather than through direct interaction with MMP-9.37 Similarly, another study found no significant relationship between TFF1 and MMP-9, attributing this discrepancy to potential variations in biomarker expression profiles or differences in the tumor microenvironment.38 The observed association between biomarkers and disease may be influenced by the disease subtype, stage, and underlying pathophysiology, as well as variations in biomarker expression across study populations. Tumor microenvironment factors, such as cytokines and ECM components, as well as patient-specific characteristics (e.g., age, treatment history, and disease stage), may further modulate biomarker levels and relationships. The findings of different studies may reflect the heterogeneity of the patient subgroups and microenvironmental conditions.
This study has limitations, including the small sample size, focusing on chemotherapy-only treatment groups, and being cross-sectional, which limits the ability to imply causation or long-term effects. The biomarker levels may be confounded by age, menopausal status, and comorbidities. The role of combination therapies, which should be larger and longitudinal, should be explored in future studies. However, an independent validation study is needed to further assess the reproducibility and generalizability of the findings. These novel interaction details would aid in a context-dependent understanding of the oncogenic roles of such markers while also highlighting the need for further characterization of the interactions between MMP-9 and TFF1 to enhance their clinical performance and advocate for their inclusion in a multi-parametric diagnostic panel.
Based on the findings of this study, biomarkers TFF1, CYFRA 21-1, and MMP-9 demonstrated significant potential as diagnostic indicators for breast cancer in women, both before and after chemotherapy. Notably, chemotherapy did not significantly alter elevated levels of CYFRA 21-1 and MMP-9 in patients with BC. However, TFF1 exhibited a marked reduction post-treatment, although its levels did not revert to those observed in the control group, suggesting a partial response to therapy. CYFRA 21-1 is a promising biomarker for advanced breast cancer stages; however, further validation and integration with complementary biomarkers are needed to improve the diagnostic and prognostic precision. MMP-9, a complex biomarker, has potential for early detection and prognosis; however, its functional dynamics require further research. Variations in TFF1 and MMP-9 interactions highlight their context-dependent roles in breast cancer progression.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
This work was completed on our own initiative without institutional funding or scholarships. We sincerely appreciate the management and personnel of the Oncology Center at Al-Sadr Teaching Hospital in the Basrah Governorate for helping with the patient diagnosis and blood sample collection. We also extend our sincere thanks to the Deanship of the College of Medical and Health Technologies, Southern Technical University, and to the staff of the biotechnology laboratory in it for the facilities they provided to us for conducting the research analyses.
9. Conflict of interest
The authors declare no conflict of interest.
10. Authors’ contribution
Both authors took equal part in the conduct of this study and the manuscript preparation.
Authors affiliations:
- Huda A. Owaid, College of Health and Medical Techniques/Al-Basrah, Southern Technical University, Ministry of Higher Education and Scientific Research, Iraq: Email: Huda.A.Owayd@fgs.stu.edu.iq, {ORCID: 0009-0002-4286-9102}
- Abdulkareem M. Jewad, College of Health and Medical Techniques/Al-Basrah, Southern Technical University, Ministry of Higher Education and Scientific Research, Iraq: Email: drkreem.mohammed@stu.edu.iq, {ORCID: 0000-0003-0608-4279}
ABSTRACT
Background & objectives: Breast cancer is a major worldwide health issue. A delayed and often inadequate diagnosis results in the death of many women. As a result, it is beneficial to conduct research and analyze new markers for an early diagnosis and surveillance of the breast cancer. This study aimed to examine the following novel diagnostic biomarkers in the serum of women with breast cancer: Trefoil Factor 1 (TFF1), Cytokeratin 21-1 (CYRA21-1), and matrix metalloproteinase-9 (MMP-9) to establish any relationship in between.
Methodology: This case-control study comprised of 72 women diagnosed with breast cancer. The participants were distributed as follows: pretreatment group (n = 15) and a post-treatment group with chemotherapy composed of anthracycline, cyclophosphamide, and docetaxel (ACD) (n = 57). A total of 15 healthy women were included in the control group. Concentrations of TFF1, CYRA21-1, and MPP-9 were measured in all study groups.
Results: The results revealed significant increases in TFF1, CYRA21-1, and MPP-9 levels (P< 0.000) in all patient groups before and after chemotherapy compared to the control group. TFF1 levels also showed a significant positive correlation with MPP-9 levels.
Conclusion: This study identified TFF1, CYFRA 21-1, and MMP-9 as valuable diagnostic biomarkers for the diagnosis of breast cancer with differential response to chemotherapy. While CYFRA 21-1 and MMP-9 showed consistent diagnostic potential, TFF1 levels were partially reduced post-treatment.
Keywords: Biomarkers; Breast cancer; Chemotherapy; TFF1; CYRA21-1; MPP-9.
Citation: Owaid HA, Jewad AM. Assessment of trefoil factor 1, cytokeratin 21-1, and human matrix metalloproteinase 9 as novel diagnostic biomarkers for breast cancer. Anaesth. pain intensive care 2025;29(3):436-443. DOI: 10.35975/apic.v29i3.2685
Received: February 17, 2025; Revised: February 22, 2025; Accepted: March 18, 2025
1. INTRODUCTION
Breast cancer is a major health issue worldwide. It is the most frequently diagnosed carcinoma globally and a primary cause of cancer-related deaths among women. In 2020, over 50 percent of breast cancer diagnoses and two-thirds of breast cancer-related deaths occurred in less developed regions, despite the disease's prior association with affluent nations.1–3 Therefore, diagnostic and prognostic assessments remain the primary preventive and treatment approaches for the spread of breast cancer. In the present study, we aimed to assess the diagnostic value of newly discovered biomarkers (TFF1, Cytokeratin 21-1, MMP9) to detect BC in the preliminary phase. Previous studies have shown their significance in the diagnosis of various types of cancer. Trefoil Factors are components of the mucus barrier and are found in exocrine bodily fluids, such as gastric juice, saliva, and breast milk. Urine and tears.4 Trefoil Factor-1 (TFF1) was first identified in the MCF-7 breast cancer cell line and is commonly found in gastric carcinomas and gastroduodenal mucosa. It also interacts with mucins and other members of the ECM, enhancing its protective role and playing a role in repair. Trefoil Factor-1 plays a crucial role in maintaining the structural integrity of epithelia, with anti-damage and repair-promoting effects on the tissue. They strengthen the mucosa and contribute to cell growth and movement, which are crucial for tissue repair.5 It has been identified as a biomarker of metastatic colon cancer.6 TFF1 is encoded by the Trefoil Factor1 gene, which is located on chromosome 21 in humans. TFF1 is predominantly expressed in the gastrointestinal tract, including the stomach, duodenum, and mammary gland. Nonetheless, TFF1 in triple-negative breast cancer has been investigated relatively rarely to date.7 TFF1 has also been implicated in gastric cancer. Under conditions of mucosal damage and inflammation, TFF1 can be induced. Thus, the function of TFF1 may be context-sensitive, depending on the type and stage of cancer.8
Cytokeratin 21-1 (CK 21-1) is a proteolytic degradation product of cytokeratin 19 (CK19), a structural component of epithelial cell intermediate filaments. Specifically, CK 21-1 represents soluble fragments of CK19 released into the systemic circulation during pathological processes,9 an intermediate filament protein of epithelial cells. This fragment is mainly employed in oncology to evaluate the overall progression of the disease as well as the response to therapy, especially in cancers that sample strongly for cytokeratin 19,10 Elevated levels of CYRA21-1 in the serum or blood can result from the breakdown of cytokeratin 19 in response to cellular damage or cancerous processes. Cytokeratin 21-1 serves as a marker for cancer diagnosis and monitoring. It is particularly useful for detecting the presence of tumor cells and tracking disease progression because of its release from epithelial cells during tumor invasion and metastasis. It is elevated in lung cancer,11 but is not a primary marker of prostate cancer.12 Cytokeratin 21-1 is useful in identifying epithelial cancers on a general basis because it originates from cytokeratin 19. However, their utility is cancer type- and stage-dependent. Analogously, according to the available studies, the utility of CYRA21-1 as a biomarker in gastric and colorectal cancers is still being assessed and is not as established as in breast and lung cancers.12,13
Human matrix metalloproteinase 9 (MMP9), also known as Gelatinase B, belongs to the type IV collagenase family. It is distinguished by its zinc content and participates in pathological processes that result in massive tissue dissolution, as well as physiological functions that involve the degradation of the tissue matrix. Because of the function of the enzyme, it is a particularly promising target for studying the mechanisms by which specific diseases and therapies drive tissue disintegration.14 Human matrix metalloproteinase 9 is encoded by the MMP9 gene, which is located on chromosome 20 in humans. This protein is synthesized as an inactive proenzyme (proMMP-9), which requires activation to become functionally active. Human matrix metalloproteinase 9 s produced primarily by neutrophils but also by other cell types, such as macrophages, fibroblasts, and cancer cells. It is released into the extracellular environment in response to stimuli, such as inflammation and tissue damage. Matrix metalloproteinase 9 isolated from humans has been implicated in the final step of tissue remodeling through the cleavage of type IV and type V collagen, which forms the basis of the basement membrane and Extracellular Matrix (ECM). This activity is crucial for tissue remodeling and repair. The exception is breaking down ECM boundaries, in which MMP-9 also promotes cell migration and invasion, which is essential for wound healing, but also for pathological conditions, such as cancer metastases.15 Elevated levels of MMP-9 are often linked to aggressive cancer behavior and poor prognosis in various cancers, including lung and prostate cancers.16,17 CYFRA 21-1 is mainly a detection marker indicative of tumor-induced degradation,9 whereas TFF1 is context-dependent and capable of suppressing and supporting tumorigenesis7. MMP-9 is an essential factor involved in cancer progression via several mechanisms.18 These biomarkers offer complementary insights into cancer biology, making them promising components of multi-marker panels for cancer diagnosis, prognostic evaluation, and therapeutic monitoring.7,9,18 Treatment with chemotherapy in women with breast cancer changed some measured biomarkers, such as oxidative stress-related biomarkers.19 We believe that it is worthwhile to study the above markers before and after chemotherapy to determine whether they affect the diagnostic ability of these novel biomarkers. The current study aimed to examine Trefoil Factor 1 (TFF1), Cytokeratin 21-1 (CYRA21-1), and matrix metalloproteinase-9 (MMP-9) as novel diagnostic biomarkers in the serum of women with breast cancer and a healthy control group.
2. METHODOLOGY
This was a case-control study. This study included patients who visited the Oncology and Hematology Center/Tumor LAB Department of Al-Sadder Teaching Hospital in Basrah governorate; the sample was taken in the duration of December 2023 to May 2024. Diagnosis of breast cancer is based on The TNM stage (Tumor, Node, Metastasis) system, a globally standardized framework that categorizes malignancies based on their anatomical extent. This system classifies primary tumor size and invasiveness (T), regional lymph node involvement (N), and distant metastasis (M). The American Joint Committee on Cancer (AJCC) delineates breast cancer staging into two categories: anatomic staging, which relies solely on TNM criteria, and prognostic staging, which incorporates TNM alongside histopathological tumor grade and biomarker profiles (e.g., hormone receptor and HER2 status). The study population comprised 72 women with breast cancer divided into two categories: 15 before therapy (group 2) and 57 post treated with chemotherapy, which included 18 women at stage 1 (group 3), 32 at stage 2 (group 4), and 7 at stage 3 (group 5). Fifteen apparently healthy women served as the control group (Group 1).
Inclusion and Exclusion criteria
Patients with other chronic diseases (cardiovascular diseases, infections, and endocrine disorders) were excluded from the study.
Sample collection
Both patients and controls were dispensed in a gel tube and allowed to coagulate at room temperature for 10 min. Blood samples were centrifuged at 3000 rpm for 5 min. Subsequently, the sera were precipitated and stored at − 20 °C until further use. Serum evaluations of the three biomarkers were conducted utilizing ELISA (Enzyme-Linked Immunosorbent Assay (ELISA) with the following specific commercial kits: (TFF1, CYRA21-1, and MMP-9) (Elabscience, USA). All ELISA were performed according to the manufacturer's instructions.
Statistical analysis
T he collected data were entered into SPSS version 26 for tabulation and analyses. Variables were coded and defined such that categorical variables were presented as frequencies and percentages, whereas continuous variables were presented as means and standard deviations. The Kolmogorov test was used to examine the normality of the continuous variables. The Kruskal–Wallis test was used to investigate the differences in the variables across the five study groups. Pairwise comparisons were made for variables that showed significant statistical differences. Corresponding post-hoc tests were used to examine between-group differences. The level of significance was set at p < 0.05.
3. RESULTS
The present case-control study population comprised 72 women with breast cancer, divided into two categories: 15 before therapy (group 2) and 57 post-treated with chemotherapy, which comprised 18 women at stage 1 (group 3), 32 women at stage 2 (group 4), and 7 women at stage 3 (group 5). Fifteen healthy women were included in the control group (Group 1). Table 1 shows that There were no significant differences in age between the study groups (Table 1). TFF1 levels increased significantly in all patient groups post- and pre-treatment in comparison to the experimental control group (P < 0.0001), and the post-treatment group demonstrated significant decreases in comparison to the pre-treatment group, as shown in Table 1. CYFRA 21-1 levels in the same table were also much higher in all the patient groups, post- and pre-treatment groups, than in the control experimental group. However, the data presented in Table 1 demonstrate that MMP-9 levels were significantly elevated across all patient groups, including both the pre-treatment and post-treatment cohorts, when compared to the control or healthy group. However, a slight reduction in MMP-9 levels was observed, particularly in the stage 3 patient group, although these levels remained significantly higher than those in the control group. In summary, TFF1, CYFRA 21-1, and MMP-9 levels changed
significantly with disease progression and treatment (Table 1). (1), (2), and (3)
| Table 1: A Comparison between the studied biomarkers in all of the five groups | ||||||
| Parameter | Control
(n = 15) |
Pretreatment
(n = 15) |
Stage 1
(n = 18) |
Stage 2
(n = 32) |
Stage 3
(n = 7) |
P value |
| Age (years) | 44.60 ±13.94A | 49.27 ± 6.2A
|
49.28± 7.48A | 48.75 ± 7.02A
|
51.14 ± 5.01A
|
0.682 |
| TFF1
(Pg/ml) |
38.49 ± 10.7A | 371.86 ± 29.33B | 327.33 ± 40.23C | 328.62±44.7C | 345.57±31.41C | 0.0001** |
| CYFRA21-1
(ng/ml) |
1.94 ± 0.37A | 13.17 ± 2.57B
|
13.86 ± 3.57B
|
14.09±4.187B
|
14.96 ± 4.71B
|
0.0001** |
| MMP9
(ng/ml) |
5.53 ± 3.6A | 11.15 ± 1.2B | 11.71 ± 1.81B | 11.15 ± 1.83B | 10.1 ± 1.85B | 0.0001** |
| *Kruskal Wallis test **Significant at 0.05 level. Capital letters A, B, and C indicate the level of significance following Tukey's multiple comparisons test; similar letters indicate no significant differences, whereas different letters indicate significant differences. | ||||||

Figure 1: Level of TFF1 in serum in the study groups

Figure 2: Level of CYRA21-1 in serum in the study groups

Figure 3: Level of MMP-9 in serum in the study groups correlation among the studied biomarkers:
Table 3 shows a moderate positive correlation between the TFF1 and MMP-9 levels (R= 0.412, P = 0.019).
| Table 2: A correlation among the studied biomarkers. | ||||
| Biomarker | CYFRA21_1 | TFF1 | MM_9 | |
| CYFRA21_1 | r | 1.000 | -0.085 | -0.053 |
| P value | 0.645 | 0.773 | ||
| TFF1 | r | -0.085 | 1.000 | 0.412 |
| P value | 0.645 | 0.019* | ||
| *Spearman correlation coefficient; P < 0.05 considered as significant | ||||
4. DISCUSSION
The present study demonstrated a significant elevation in TFF1 levels across all patient cohorts following treatment compared to the experimental control group. Concurrently, a marked reduction in TFF1 levels was observed in the post-treatment groups, consistent with recent findings of elevated TFF1 expression in patients with BC. For instance, Lang et al. (2021) documented that TFF1 is overexpressed in BC tissues and that its expression levels are positively correlated with tumor grade and stage. These results confirm the initial hypothesis that TFF1 might be a candidate biomarker for BC progression. Moreover, their results also validated that a high level of TFF1 was generally associated with an increased cancer grade and more processive stage of BC tissues in comparison with adjacent normal tissue,20 which provides a rationale for exploring the prognostic implications of TFF1 in BC. A subsequent study by Yi et al. (2020) also identified TFF1 as an early cancer biomarker. TFF1 levels were significantly higher in patients with early-stage BC than in healthy control subjects. This indicates that TFF1 could be a promising biomarker for the diagnosis of early stage cancer and is helpful for cancer multiplexing.21
TFF1 is widely recognized for its critical role in mucosal protection and repair. In cancer biology, TFF1 upregulation could be linked to tumor adaptive mechanisms used to protect it against the surrounding microenvironment or to form a specific niche that favors cancer progression. Interestingly, new data suggest that TFF1 is involved in the epithelial-mesenchymal transition (EMT), a process directly correlated with cancer metastasis, thereby strengthening this working hypothesis. High expression levels of TFF1 could reflect the tumor’s attempt to create a protective microenvironment, thus facilitating its development. The repeated finding that TFF1 is upregulated in a range of tumors points to its possible application as a diagnostic or prognostic biomarker in cancer.22
However, some studies have reported findings inconsistent with the current results, which demonstrated elevated TFF1 levels across all tested cases. These studies found no significant difference in TFF1 levels between cancer patients and normal controls, suggesting that TFF1 may not serve as a reliable biomarker for all molecular subtypes of breast cancer. Furthermore, it has been proposed that TFF1 expression may be influenced by disease stage of the disease.23 Additionally, different authors have reported discrepancies in circulating TFF1 levels based on factors including both the hormone receptor status of their patients and the treatments administered to them. Wang et al. Shunyi et al. (2022) suggested that TFF1 expression is modulated by estrogen receptor-positive status, which complicates its further application as a consistent biomarker in BC.24
The study showed that CYFRA 21-1 levels were significantly higher in all patient groups than in the control group, both pre- and post-treatment. These results confirm those of previous studies on the importance of CYFRA 21–1 in BC, all of which found elevated levels in patients with more advanced forms of the disease. These data suggest that CYFRA 21-1 is a potential biomarker of disease progression and treatment response.25 Moreover, another study showed that the levels of CYFRA 21-1 were markedly elevated in patients with breast cancer compared to those in healthy individuals and correlated positively with tumor stage. They stated that although cytokeratin fragments 21-1 might not be an evolutionary biomarker, when used simultaneously with different biomarkers, it holds huge diagnostic worth.26 Additionally, previous research has highlighted notable differences in CYFRA 21-1 levels between cancer patients and controls, particularly in individuals with advanced disease, underscoring its potential utility in monitoring disease status and progression.27
CYFRA 21-1 is distinguished by its epitope, which is a polypeptide typically released following cellular death. Elevated serum titers of CYFRA 21-1 have been detected in various malignancies, particularly lung cancer; however, healthy individuals rarely exhibit abnormal serum levels of this marker.28 In breast cancer patients, elevated CYFRA 21-1 levels may result from increased cellular turnover and cytokeratin breakdown, a phenomenon also observed in other cancers. Higher CYFRA 21-1 levels in advanced breast cancer stages may reflect a greater tumor burden and more aggressive disease, consistent with its role in other malignancies, where elevated levels are associated with advanced disease progression.26
These findings contrast with those of previous research, suggesting that CYFRA 21-1 has not yet been established as a standard biomarker for breast cancer.29 For instance, Filella et al. (2023) reported that while some breast cancer patients exhibited elevated CYFRA 21-1 levels, variability in sensitivity and specificity limited its clinical utility. The study concluded that although CYFRA 21-1 may provide supplementary diagnostic information, it should not replace established biomarkers.30
This study demonstrated elevated MMP-9 levels across all patient groups, with a modest reduction observed in stage 3 patients. Despite this decrease, the MMP-9 levels remained significantly higher than those in the control group. These findings align with those of prior studies, indicating that elevated MMP-9 levels are associated with the early stages of BC and may serve as a potential biomarker for disease progression. Increased MMP-9 levels may reflect active remodeling of the extracellular matrix (ECM) during the initial phases of tumorigenesis.31 Joseph et al. MMP-9 also plays a critical role in the late stage of breast cancer progression, and high plasma MMP-9 levels are correlated with the initial stage of breast cancer and have been associated with poor prognosis.32 These findings are also supported by previous reports, which confirmed that upregulated levels of MMP-9 assure a higher level of cellular proliferative activity. Together, these results indicate that MMP-9, which is expressed in both stromal and tumor cells, contributes to BC progression. MMP-9 plays a decisive role in ECM remodeling, which is especially active during the early stages of tumorigenesis when the tumor adapts to invade the surrounding tissue. Cumulatively, high serum MMP-9 levels may also indicate the pronounced ECM restructuring required for tumor progression and invasion. Therefore, MMP-9 levels in early breast cancer may be an important tool for early detection and prognosis as well as for the stratification of patients according to their risk profile.33
Jiang and Li (2021) found a decrease in MMP-9 levels in certain subtypes of advanced-stage breast cancer. They suggested that this reduction could be a consequence of modifications in the tumor microenvironment or of adaptive behaviors adopted by cancer cells to counteract excessive ECM degradation, leading to a different protein degradation profile in more advanced stages.34 It is hypothesized that during advanced tumor development, tumors may alter the proteolytic/anti-proteolytic balance. The observed decrease in MMP-9 levels could also be attributed to compensatory mechanisms or changes in other components of the tissue microenvironment, which may not directly reflect a reduction in proteolytic activity but rather a shift in enzyme functionality. Breast cancer is a highly heterogeneous disease, and different subtypes of breast cancer may exhibit varying MMP-9 concentrations.31
The findings of this study are consistent with prior research that demonstrated a significant association between TFF1 and MMP-9 expression levels, aggressive breast cancer phenotypes, and poor clinical outcomes. Specifically, a significant p-value was observed for both biomarkers, indicating their potential role in tumor progression. These results align with earlier studies that reported a significant correlation between TFF1 and MMP-9 levels, suggesting a possible interplay wherein TFF1 may modulate MMP-9 activity, or vice versa, thereby contributing to cancer progression.35 The results are also consistent with the study by Manfioletti and Fedele (2023), who identified a good association between TFF1 with MMP-9 positive breast cancer tissues, especially in those with more aggressive and metastatic phenotypes. This supports the hypothesis that TFF1 is mechanistically related to MMP-9 in promoting aggressive cancer behavior.36
However, this study contradicts previous research that showed a negative correlation between TFF1 and MMP-9. For example, some studies have found that MMP-9 is a strong predictor of poor prognosis, but that TFF1 and MMP-9 are not significantly correlated. These studies suggest that the involvement of TFF1 in breast cancer may be modulated through interactions with other biomarkers or signaling pathways, rather than through direct interaction with MMP-9.37 Similarly, another study found no significant relationship between TFF1 and MMP-9, attributing this discrepancy to potential variations in biomarker expression profiles or differences in the tumor microenvironment.38 The observed association between biomarkers and disease may be influenced by the disease subtype, stage, and underlying pathophysiology, as well as variations in biomarker expression across study populations. Tumor microenvironment factors, such as cytokines and ECM components, as well as patient-specific characteristics (e.g., age, treatment history, and disease stage), may further modulate biomarker levels and relationships. The findings of different studies may reflect the heterogeneity of the patient subgroups and microenvironmental conditions.
5. LIMITATIONS
This study has limitations, including the small sample size, focusing on chemotherapy-only treatment groups, and being cross-sectional, which limits the ability to imply causation or long-term effects. The biomarker levels may be confounded by age, menopausal status, and comorbidities. The role of combination therapies, which should be larger and longitudinal, should be explored in future studies. However, an independent validation study is needed to further assess the reproducibility and generalizability of the findings. These novel interaction details would aid in a context-dependent understanding of the oncogenic roles of such markers while also highlighting the need for further characterization of the interactions between MMP-9 and TFF1 to enhance their clinical performance and advocate for their inclusion in a multi-parametric diagnostic panel.
6. CONCLUSION
Based on the findings of this study, biomarkers TFF1, CYFRA 21-1, and MMP-9 demonstrated significant potential as diagnostic indicators for breast cancer in women, both before and after chemotherapy. Notably, chemotherapy did not significantly alter elevated levels of CYFRA 21-1 and MMP-9 in patients with BC. However, TFF1 exhibited a marked reduction post-treatment, although its levels did not revert to those observed in the control group, suggesting a partial response to therapy. CYFRA 21-1 is a promising biomarker for advanced breast cancer stages; however, further validation and integration with complementary biomarkers are needed to improve the diagnostic and prognostic precision. MMP-9, a complex biomarker, has potential for early detection and prognosis; however, its functional dynamics require further research. Variations in TFF1 and MMP-9 interactions highlight their context-dependent roles in breast cancer progression.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
This work was completed on our own initiative without institutional funding or scholarships. We sincerely appreciate the management and personnel of the Oncology Center at Al-Sadr Teaching Hospital in the Basrah Governorate for helping with the patient diagnosis and blood sample collection. We also extend our sincere thanks to the Deanship of the College of Medical and Health Technologies, Southern Technical University, and to the staff of the biotechnology laboratory in it for the facilities they provided to us for conducting the research analyses.
9. Conflict of interest
The authors declare no conflict of interest.
10. Authors’ contribution
Both authors took equal part in the conduct of this study and the manuscript preparation.
11. REFERENCES
- Sung VYC, Knight JF, Johnson RM, Dey-Guha I, Kim J, Smith JL, et al. Co-dependency for MET and FGFR1 in basal triple-negative breast cancers. NPJ Breast Cancer. 2021;7(1):36. PubMed DOI: 1038/s41523-021-00238-4
- Usman A, Mehdi SR, Khan AA. Preventive analgesia with oral tramadol and pregabalin for post-operative pain in breast surgical patients. Anaesth Pain Intensive Care. 2024;28(1):81–4. DOI: 35975/apic.v28i1.2374
- Maqsood S, Rafiq M. Effect of circle therapy on breast cancer pain management. Anaesth Pain Intensive Care. 2019;23(1):63–7. DOI: 35975/apic.v23i1.1088
- Hoffmann W. Trefoil factor family (TFF) peptides and their links to inflammation: A re-evaluation and new medical perspectives. Int J Mol Sci. 2021;22(9):4909. PubMed DOI: 3390/ijms22094909
- Ishibashi Y, Ohtsu H, Ikemura M, Ishikawa T, Iwase H, Yamamoto H, et al. Serum TFF1 and TFF3 but not TFF2 are higher in women with breast cancer than in women without breast cancer. Sci Rep. 2017;7(1):4846. PubMed DOI: 1038/s41598-017-05129-y
- Minegishi K, Dobashi Y, Tsubochi H, Hagiwara K, Ishibashi Y, Nomura Y, et al. TFF-1 functions to suppress multiple phenotypes associated with lung cancer progression. Onco Targets Ther. 2021;14:4761–77. PubMed DOI: 2147/OTT.S322697
- Zhang Y, Liu Y, Wang L, Song H. The expression and role of trefoil factors in human tumors. Transl Cancer Res. 2019;8(4):1609–17. PubMed DOI: 21037/tcr.2019.07.48
- Qian Z, Jiang Y, Shou C, Yu J, Huang D, Xie H, et al. Validation of the DNA methylation landscape of TFF1/TFF2 in gastric cancer. Cancers (Basel). 2022;14(22):5474. PubMed DOI: 3390/cancers14225474
- Chen CJ. The roles of CYFRA 21-1 in cancer. Adv Eng Technol Res. 2024;12(1):788. DOI: 56028/aetr.12.1.788.2024
- Dall’Olio FG, Abbati F, Facchinetti F, Massucci M, Melotti B, Squadrilli A, et al. CEA and CYFRA 21-1 as prognostic biomarkers and as tools for treatment monitoring in advanced NSCLC treated with immune checkpoint inhibitors. Ther Adv Med Oncol. 2020;12:1–12. PubMed DOI: 1177/1758835920952994
- Liu L, Xie W, Xue P, Wei Z, Liang X, Chen N. Diagnostic accuracy and prognostic applications of CYFRA 21-1 in head and neck cancer: a systematic review and meta-analysis. PLoS One. 2019;14(5):e0216561. PubMed DOI: 1371/journal.pone.0216561
- Ajona D, Remirez A, Sainz C, et al. A model based on the quantification of complement C4c, CYFRA 21-1 and CRP exhibits high specificity for the early diagnosis of lung cancer. Transl Res. 2021;233:77-91. PubMed DOI: 1016/j.trsl.2021.02.009
- Bi H, Yin L, Fang W, Song S, Wu S, Shen J. Association of CEA, NSE, CYFRA 21-1, SCC-Ag, and ProGRP with clinicopathological characteristics and chemotherapeutic outcomes of lung cancer. Lab Med. 2023;54(4):372-9. PubMed DOI: 1093/labmed/lmac122
- Majeed S, Singh P, Sharma N, Sharma S. Role of matrix metalloproteinase-9 in progression of tuberculous meningitis: a pilot study in patients at different stages of the disease. BMC Infect Dis. 2016;16:1-7. PubMed DOI: 1186/s12879-016-1953-9
- Teng Z, Wang S, Yuan H, Wang H, Li J, Chang X, et al. MMP-9 gene polymorphisms on cancer risk: an updated systematic review and meta-analysis. Nucleosides Nucleotides Nucleic Acids. 2024;43(10):1090-113. PubMed DOI: 1080/15257770.2023.2299710
- Caimi G, Hopps E, Montana M, Urso C, Carollo C, Canino B, et al. The function of matrix metalloproteinase-9 (MMP-9) and its tissue inhibitor (TIMP-1) in several clinical conditions: results and analysis of our survey. Clin Hemorheol Microcirc. 2021;78(4):401-16. PubMed DOI: 3233/CH-201094
- Zhao Y, Yakufu M, Ma C, Wang B, Yang J, Hu J. Transcriptomics reveal a molecular signature in the progression of nonalcoholic steatohepatitis and identifies PAI-1 and MMP-9 as biomarkers in in vivo and in vitro studies. Mol Med Rep. 2023;29(1):15. PubMed DOI: 3892/mmr.2023.13138
- Kalali D. The role of the matrix metalloproteinase-9 gene in tumor development and metastasis: a narrative review. Glob Med Genet. 2023;10(2):48-53. PubMed DOI: 1055/s-0043-1768166
- Alimohammadi M, Rahimi A, Faramarzi F, Golpour M, Jafari-Shakib R, Alizadeh-Navaei R, et al. Effects of coenzyme Q10 supplementation on inflammation, angiogenesis, and oxidative stress in breast cancer patients: a systematic review and meta-analysis of randomized controlled trials. Inflammopharmacology. 2021;29(3):579-93. PubMed DOI: 1007/s10787-021-00817-8
- Lang DK, Sood A, Kaur R, Arora R, Behl T. Focus on the multimodal role of biomarkers in breast cancer. Curr Pharm Des. 2021;27(35):3686-701. PubMed DOI: 2174/1381612827999210111185213
- Yi J, Ren L, Li D, Wu J, Li W, Du G, et al. Trefoil factor 1 (TFF1) is a potential prognostic biomarker with functional significance in breast cancers. Biomed Pharmacother. 2020;124:109827. PubMed DOI: 1016/j.biopha.2020.109827
- Werner Rönnerman E, Pettersson D, Nemes S, Dahm-Kähler P, Kovács A, Karlsson P, et al. Trefoil factor family proteins as potential diagnostic markers for mucinous invasive ovarian carcinoma. Front Oncol. 2023;12:1112152. PubMed DOI: 3389/fonc.2022.1112152
- Djilali-Doula F, Mohammedi L, Senhadji R. Characterization of genotypic disorders in ERα+ breast cancers by evaluating ERα and TFF1 expression: is TFF1 a promising predictive biomarker for endocrine therapy? South Asian J Exp Biol. 2022;12(6). DOI: 38150/sajeb.12(6).p962-970
- Wang Q, Jiang Y, Du M, Yang L, Yuan Q. Association of functional genetic variants in TFF1 and nephrolithiasis risk in a Chinese population. BMC Urol. 2022;22(1):127. PubMed DOI: 1186/s12894-022-01081-w
- Ebied SA, Abdel-Rehim WME, El-Benhawy SA, El-Gawish MA, Hassan MAA, El-Settawy II. Serum CYFRA 21-1 in Egyptian women with breast cancer. Alex J Med. 2017;53(1):41-7. DOI: 1016/j.ajme.2016.02.006
- Garcia-Valdecasas Gayo S, Ruiz-Alvarez MJ, Gonzalez-Gay D, Ramos-Corral R, Marquez-Lietor E, Del Amo N, et al. CYFRA 21-1 in patients with suspected cancer: evaluation of an optimal cutoff to assess the diagnostic efficacy and prognostic value. Adv Lab Med Int J Lab Med. 2020;1(4):20200005. PubMed DOI: 1515/almed-2020-0005
- Varlotto JM, Cardarelli GA. Editorial: Recent advances in cervical cancer radiotherapy. Front Oncol. 2023;13:1144797 PubMed DOI: 3389/fonc.2023.1144797
- Sun A. Clinical role of serum tumor markers SCC, NSE, CA 125, CA 19-9, and CYFRA 21-1 in patients with lung cancer. Lab Med. 2023;54(6):638-45. PubMed DOI: 1093/labmed/lmad020
- Mehta A, Parkash A, Bhatia M. Cross-sectional study to establish the utility of serum tumor markers in the diagnosis of lung cancer. Asian Pac J Cancer Prev. 2021;22(8):2569. PubMed DOI: 31557/APJCP.2021.22.8.2569
- Filella X, Rodríguez-Garcia M, Fernández-Galán E. Clinical usefulness of circulating tumor markers. Clin Chem Lab Med. 2023;61(5):895-905. PubMed DOI: 1515/cclm-2022-1090
- Winkler J, Abisoye-Ogunniyan A, Metcalf KJ, Werb Z. Concepts of extracellular matrix remodelling in tumour progression and metastasis. Nat Commun. 2020;11(1):5120. PubMed DOI: 1038/s41467-020-18794-x
- Joseph C, Alsaleem M, Orah N, Narasimha PL, Miligy IM, Kurozumi S, et al. Elevated MMP9 expression in breast cancer is a predictor of shorter patient survival. Breast Cancer Res Treat. 2020;182:267-82. PubMed DOI: 1007/s10549-020-05670-x
- Khambri D, Suyuthie HD, Hilbertina N, Yetti H, Purwanto DJ. Matrix metalloproteinase-9 as prognostic factor for the treatment of HER-2 enriched breast cancer. Asian Pac J Cancer Prev. 2022;23(3):1013. PubMed DOI: 31557/APJCP.2022.23.3.1013
- Jiang H, Li H. Prognostic values of tumoral MMP2 and MMP9 overexpression in breast cancer: a systematic review and meta-analysis. BMC Cancer. 2021;21:149. PubMed DOI: 1186/s12885-021-07860-2
- Wu W, Warner M, Wang L, He WW, Zhao R, Guan X, et al. Drivers and suppressors of triple-negative breast cancer. Proc Natl Acad Sci U S A. 2021;118(33):e2104162118. PubMed DOI: 1073/pnas.2104162118
- Manfioletti G, Fedele M. Epithelial–mesenchymal transition (EMT). Int J Mol Sci. 2023;24(14):11386. PubMed DOI: 3390/ijms241411386
- Yu C, Wu Y, Hu K, Zhang S. Prominin 1 significantly correlated with bone metastasis of breast cancer and influenced the patient's prognosis. Biomed Res Int. 2022;2022(1):4123622. PubMed DOI: 1155/2022/4123622
- Jahan R, Shah A, Kisling SG, Macha MA, Thayer S, Batra SK, et al. Odyssey of trefoil factors in cancer: diagnostic and therapeutic implications. Biochim Biophys Acta Rev Cancer. 2020;1873(2):188362. PubMed DOI: 1016/j.bbcan.2020.188362