FDA Recalls Implantable Infusion Pumps and Refill Kits
FDA issued a class I recall of Medtronic’s SynchroMed II and SynchroMed EL implantable infusion pumps and refill kits, used for the long-term infusion of pain, cancer treatment and anti-spasm medications. Class I recalls are the most serious type of recall and involve situations in which there is a reasonable probability that use of the recalled product will cause serious adverse health consequences or death. These pumps and refill kits have been recalled because pocket fills — the unintended injection of drugs or fluids into the tissue under the skin at the pump pocket site — have occurred and may result in patient harm, serious injury, and/or death due to drug overdose or underdose. The recalled products were manufactured from June 1998 to January 2003 and distributed from April 1999 to January 2011.
www.fda.gov/medwatch/report.htm
FDA Recalls Central Venous Catheter Trays
Because of leaks in the plunger luer detected during a routine syringe leak test, various types of Cook, Inc, central venous catheter trays are subject to a class I recall, FDA announced. “The potential exists for leakage and possible loss of sterility. This may lead to serious adverse health consequences and/or death,” according to an alert from MedWatch, the FDA’s safety information and adverse event reporting program. .
http://www.medscape.com/viewarticle/738433?src=mp&spon=46
Latest Johnson & Johnson Recall Involves Sterility-Risk Sutures
Johnson & Johnson, which has been plagued by repeated recalls of its consumer medicines and medical devices over the past year, has recalled 107 batches of surgical sutures in December due to potential sterility problems. The recall came to light on Wednesday after the United Kingdom Medicines and Healthcare products Regulatory Agency (MHRA) described the action on the agency’s website. The action stemmed from potentially faulty packaging seals on the individually wrapped sutures that raised a contamination risk, J&J said. The potential problem was caused by modifications of manufacturing equipment and has been corrected, said Barbara Montresor, a spokeswoman for J&J’s Ethicon surgical products division.
http://www.medscape.com/viewarticle/738340?src=mp&spon=46
Morphine/Naltrexone Combo Temporarily Withdrawn
King Pharmaceuticals Inc, a wholly owned subsidiary of Pfizer, has voluntarily recalled all dosages of a combination morphine sulfate and naltrexone hydrochloride (Embeda Extended Release Capsules CII) from wholesalers and retailers in the United States because “a prespecified stability requirement was not met during routine testing,” a message on the product’s Website notes. Available data suggest that the issue is unlikely to pose a safety risk to patients using the product as prescribed, the company’s message to patients and prescribers notes.
http://www.medscape.com/viewarticle/739008?src=mp&spon=46
Ventilators for Mass Casualty Scenarios
St. Louis-based Allied Healthcare has released a line of ventilators designed for mass casualty situations. During natural or man-made disasters, hospitals may be overwhelmed by the sheer number of patients needing life support, and clinical staff typically not qualified to provide life support may be required to assist. Additionally, electric power may become unavailable. The Allied Mass Casualty Ventilators feature simple operation, long battery life, and are able to operate without external gas connections.
MedeGrip
MedeGrip, a simple foam device for working with small or breakable things in the clinic or on the floor, has received the European CE Mark of approval. It was less than a month ago that MedeGrip was successfully registered with the FDA as a Class 1 device and Access Scientific included it as part of the POWERWAND Maximum Barrier Kit. Developed by a PICC line nurse, the device helps take away the pain of handling things like Luer Loks and pin knots, and makes it a bit safer to work with glass ampules.
Masimo E1 Pulse Oximetry Ear Sensors
Masimo has received both US FDA and European CE clearance for its E1 single-patient-use pulse oximetry ear sensor. The ear is often used as an alternative site to measure oxygenation in situations where measurement at the fingertip is impractical, e.g. excessive patient movement or low perfusion states. While traditional fingerclip sensors are often applied to the earlobe, the E1 is attached more securely in the cavum conchae. It promises to give faster detection of oxygen saturation changes during low perfusion, and to avoid cross-contamination risks associated with reusable sensors.
SpectraShield Respirator
The FDA has cleared Nexera Medical‘s (Richmond, British Columbia) SpectraShield 9500 surgical respirator, a face mask the outside of which has been shown to kill three types of bacteria (Streptococcus pyogenes, Methicillin-resistant Staphylococcus aureus (MRSA), and Haemophilus influenzae). The device is also a certified N95 respirator, blocking at least 95% of dust particles.
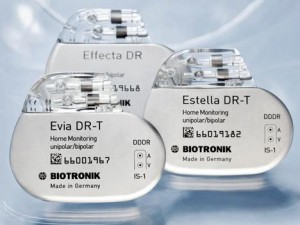
BIOTRONIK’s Latest Pacemaker Offerings
The FDA approved BIOTRONIK’s Estella and Effecta pacemakers, as well as Vp (ventricular pace) Suppression feature in the Evia line. Evia and Estella pacemakers are compatible with BIOTRONIK’s Home Monitoring technology for reporting device readings directly to the physician.
The Evia family combines the industry’s smallest wireless remote monitoring pacemakers with a decade of longevity. This advancement increases the intervals between necessary device replacement procedures, which could potentially reduce the risk associated with replacements—a risk demonstrated in the recently published REPLACE study .
The Evia family significantly expands BIOTRONIK’s spectrum of innovative treatment options to maximize intrinsic conduction, thus reducing unnecessary right ventricular pacing. Ventricular pace suppression, known as Vp Suppression, is a new, highly sophisticated algorithm that can promote innate conduction by enabling the pacemaker to stimulate the heart muscle only when appropriate.
Spirodoc

Rechargeable high capacity lithium battery.
Actigraph and step-counter accelerometer to determine patient position during sleep analysis and distance walked estimation for a 6 min. walk test.
Massive user-friendly database patient memory with powerful search, visualization and printing functions.
Innovative detachable flowmeter ensures a sturdy and durable connection.
External connectivity via Bluetooth® or USB cable to PC or printer.
Spirometry features
FVC, VC, IVC, MVV, PRE-POST BD.
Automatically records all trials.
Internal temperature sensor for automatic BTPS conversion.
Advanced spirometry test interpretation.
100% cross contamination free using MIR’s patented disposable turbine.
Reusable turbine for long-term use available on request.
Pulse Oximetry features
Simple and clear SpO2 and Pulse measurement with pletismographic curve.
Sleep disorder detection with events recording.
Six minute walk test with desaturation area index.
Parameters directly shown on the display (min, max, mean SpO2 and Pulse Rate, Δ Index, T90, T89, T88, T5, ODI, NOD, Desaturation Area etc.).