Samina Ismail, FCPS*, Arshad Taqi, FCPS**
*Associate Professor, Department of Anesthesia, Aga Khan University Hospital, Karachi (Pakistan)
**Consultant Anesthesiologist, Hameed Latif Hospital, Garden Town, Lahore (Pakistan)
Correspondence: Dr. Samina Ismail, Associate Professor, Department of Anaesthesia, Aga Khan University Hospital, Karachi (Pakistan); E-mail: samina.ismail@aku.edu
SUMMARY
Despite recent developments in the inventory management, introduction of electronic drug trolleys and cabinets, color coding of the filled syringes and many more interventions, medication errors could not be eliminated. The most common of these are syringe swap and human errors regarding wrong drug administration due to look-alike drug containers or sound-alike names of the drugs belonging to diverse groups. Many of the fatalities, that occur in third world countries due to these causes, go unnoticed and unregistered. This special article complements two special editorials on the same topic by Professor Joseph D. Tobias et al and Professor Robert Stoelting, a case report, a patient’s perspective and a ‘Cliniquiz’ being published in the current issue of the journal. It discusses salient features of this issue as well as preventive measures.
Key words: Medications; Medications errors; Adverse drug events; Look-alike drugs; Sound-alike drugs Tall man lettering; Medication Errors Reporting Program; Anesthesia Patient Safety Foundation
Citation: Ismail S and Taqi A. Medical errors related to look-alike and sound-alike drugs. Anaesth Pain & Intensive Care 2013;17(2):117-122
INTRODUCTION
Medication names that look similar or sound similar have been identified as a potential source of error in health care systems. Medications in which packaging is visually similar to another product or medication comes in the category of look-alikes. Medications for which generic or trade names of the product or medication sounds similar in the spoken or written words are categorized as sound-alike drugs. Look-alike and sound-alike (LASA) drug names can lead to the unintended interchange of drugs that can result in patient injury or death.
The existing medication-use system face problems because its safety depends on human perfection and systems that rely on human perfection are most likely to fail as no human can function perfectly all the time. Even the best-trained and most careful people occasionally make mistakes. Other hazardous industries also train and test for perfection, but they do not rely on it. Safety is achieved by designing systems, processes, and tasks that prevent mistakes.
Simplicity, standardization, differentiation, lack of duplication, and unambiguous communication are some of concepts that are relevant to the medication-use process. These principles have often been ignored in the naming, labeling, and packaging of medications. The consequences are predictable. Bad names, bad labels, and bad packages result in accidents waiting to happen.1
Medication errors are the most common cause of patient injuries in hospitals. Adverse drug events, about half of which are due to medication errors, accounted for 19% of all injuries identified by the population based Harvard Medical Practice Study.2 An estimated 2-7% of patients admitted to hospitals experience a serious medication error (one that has the potential to cause injury).2 One study at a teaching hospital yielded an estimate that, on average, each preventable adverse drug event (ADE) resulted in an additional stay of 4.6 days and a cost of $4685.3
The purpose of this article is to highlight medication errors related to LA-SA medications in order to create awareness among medical practitioners The intent is to encourage thought to formulate a system to prevent this preventable cause of morbidity and mortality in our patients. This article will discuss medication errors and the key elements that affect the medication error, bench marking for medication error, preventable measures like “Tall man lettering” to differentiate between look-alike drugs, nature and causes of look-alike drugs, potential solution to prevent this error and barrier in the implementation of these problem solving measures. In the end we will discuss the way forward and recommendations for prevention of medical errors related to LA-SA medications and similar packages.
DEFINITIONS
A medication error is “any error occurring in the medication use process”.4 The National Coordinating Council for Medication Error and Prevention has approved the following as its working definition of medication error: “… any preventable event that may cause or lead to inappropriate medication use or patient harm, while the medication is in the control of the health care professional, patient, or consumer. Such events may be related to professional practice, health care products, procedures, and systems including: prescribing; order communication; product labeling, packaging and nomenclature; compounding; dispensing; distribution; administration; education; monitoring; and use.”5
MEDICATION PROCESS
There are a number of key elements that affect the medication-use process including;
- Patient information
- Drug information
- Communication of drug information
- Drug labeling, packaging and nomenclature
- Drug storage, stock, standardization, and distribution
- Drug device acquisition, use and monitoring
- Environmental factors
- Staff competency and education:
- Patient education
- Quality processes and risk management:
As mentioned in the list, drug labeling, packaging and nomenclature is one of the key elements. Drug names that look-alike or sound-alike, as well as products that have confusing drug labeling and non-distinct drug packaging significantly contribute to medication errors. The incidence of medication errors is reduced with the use of proper labeling and the use of unit dose systems within hospitals. It is the interrelationships among all these key elements that form the structure within which medications are used.
National medication error rate and benchmarking for medication error:
It is difficult to have a national medication error rate, as the working of each organization and hospital in any country is different. Therefore true or universal bench marking is generally not possible. Even for individual organizations, it is difficult to have a true rate as most systems rely on voluntary reporting of actual events or near miss events. The problem with voluntary reporting lies in the fact that the actual number of events could be much higher than the reported event. In addition, organizations having good reporting systems have a high number of events and can be safer as compared to organizations having fewer reported incidents which could be due to poor reporting system.
Like all errors in health care, those that result from confusing names or packages are rarely reported unless they are known to result in death or serious injury. There have been no rigorous studies. A 2001 report by United State Pharmacopeia (USP) convention indicated that confusion over drug names accounted for 15% of errors reported to the USP’s Medication Errors Reporting Program (MERP) from January 1996 through December 2000. The same report lists 749 drug names reported to MERP because they sound alike when communicated orally or look alike in print or when written.1The increasing potential for LASA medication errors was highlighted in the Joint Commission’s Sentinel Event Alert6 in the United States of America and was incorporated into the Joint Commission’s National Patient Safety Goals.7
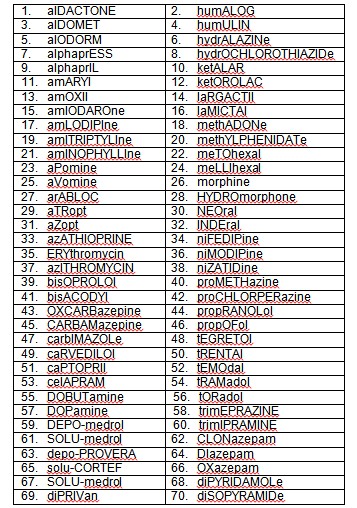
Tall Man Lettering
Tall Man lettering involves highlighting the dissimilar letters in two names to aid in distinguishing between the two for example HumaLOG and HumuLIN, oxyCODONE and OxyCONTIN; ceFAZolin and cefTRIAXONE; and FLUoxetine and DULOXetine; clonazePAM and LORazePAM.
In addition to the institution for safe medical process (ISMP), several studies have shown that highlighting sections of drug names using tall man (mixed case) letters can help distinguish similar drug names, making them less prone to mix-ups.8-13 ISMP, FDA, the Joint Commission, and other safety conscious organizations have promoted the use of tall man letters as one means of reducing confusion between similar drug names. To promote standardization, ISMP has created a list of Look-Alike Drug Name Sets with Recommended Tall Man Letters.14 A list of some drugs to which anesthesiologists are usually familiar is given in Table 1.
Table 1: Tall Man lettering for some drugs
Look-Alike Ampoules
Look-alike ampoules and vials are a reality; simply because there are so few designs and colors to choose from; so many injectable drugs to dispense; and the issue is not exactly at the forefront of stakeholders’ agenda. A few investigators have gone on to advocate uniform ampoules for all drugs in order to force the users to read their labels carefully before drawing up the medication.15 This view is, however, not supported by the majority who advocate distinct labeling to facilitate identification at the time of drawing the drug.16,17 Whereas drug regulatory authorities mandate the pharmaceutical companies to display requisite information on each ampoule/vial of the drug; the instructions on distinct labeling are more explicit for the packages while they are less binding for components within the package. Medicine and Healthcare Regulatory Agency of UK suggest an “Innovative pack design that may incorporate the judicious use of color is to be encouraged to ensure accurate identification of the medicine”.18 However, similar instructions for individual ampoules are missing. The medication safety conference hosted by the Anesthesia Patient Safety Foundation (APSF), while recognising the role of look-alike vials in drug errors, did not include a demand for the pharmaceutical industry to develop distinct looking vials/ampoules in its recommendations.19
Role of pharmaceutical companies
The factors for the medication-use process needs to be simple and should follow the principle of standardization, differentiation and lack of duplication. Unfortunately, these principles are not followed in drug naming, labeling, and packaging. Instead, current methods are based on long-standing commercial considerations and bureaucratic procedures. Drug companies have to undergo a lengthy and complex process for naming a marketable drug that involves submission of a new chemical entity and patent application, generic naming, brand naming, FDA review, and final approval. Pharmaceutical companies seek the fastest possible approval and may believe that the incremental benefit of evaluating human factors is small. “Trade dress” is the concept that underlies labeling and packaging issues for the drug industry. Even FDA standards do not require application of human factor principles, therefore drug names, labels, and packages are selected and designed in accordance with the marketplace rather than for practice conditions.
There are well-known, effective methods for minimizing confusion and making it more difficult to commit medical errors. However, pharmaceutical companies tend to resist implementing these methods because of the complexity, cost, concern about increasing already cumbersome regulations, fear of liability exposure, and loss of competitive advantage. Therefore, some new approach or combination of approaches is required to help us discover and continually define methods for improving safety.
Pharmaceutical companies on the other hand do not seem to own the problem of safety associated with look-alike drugs; they are satisfied with meeting minimum regulatory requirements for labeling and packaging.20 Drug labeling and packing rules (1986) of the Government of Pakistan state “Manner of Labeling: The following particulars shall appear either in print or in writing in inedible ink in a conspicuous manner on a label of the innermost container of drug and also on the package in which such container is packed namely”.21 Recent changes in the way the ampoules of injectable drugs are labeled in Pakistan have increased the number of look-alike drugs, which is likely to increase the risk of drug errors. Drug labels were largely printed as stickers on the ampoules; number of distinct combinations of background, color and print for individual drugs or groups of drugs with paper stickers facilitated identification of drugs during high use or crisis situations. More recently, names of the drugs and other information is imprinted on the ampoule. With a choice between transparent and dark brown glass and four ink colors, the number of combinations is small. Reading the small print particularly in yellow or white colors against a glass background is a test for an anesthesiologist’s vision.
Interestingly, healthcare professionals have either not noticed this change or accepted it as inevitable. It appears we have to live in a world of “look alike” drug containers and devise strategies to minimise errors in the face of this menace. One solution is to keep the vials/ampoules in their packages, only removing them at when they are ready to be drawn. Packages are easier to identify than ampoules or vials. However, this may be cumbersome if the medications are kept in a drug tray as the packages are generally much larger than the vials. Individual packaging may also significantly increase the cost of medications when compared to bulk packaging of vials.
FINDING THE SOLUTIONS
Prevention of errors was traditionally ascribed to individuals responsible for patient care; based on two myths, which have been challenged more recently:22
- If people try hard enough, they will not make any errors.
- If we punish people when they make errors, they will make fewer of them.
The anesthesia practitioner is responsible for prescribing, preparing, administering and recording drugs; working long hours, with distractions and often in crisis-like situations. Fraind and colleagues demonstrated multiple flaws in preparation, handling and administration of intravenous drugs and infusions by experienced anesthesia personnel in University Hospitals,23 emphasising the need to improve the systems. A comprehensive approach to prevent drug errors in anesthesia practice would involve standard prescribing, storing, dispensing, drawing, and drug administration practices. Experience with the airline industry has demonstrated that the frequency of errors can be reduced by breaking down the task into components and repeated drills.
A seven step approach to patient safety advocated by the National Patient Safety Foundation of the UK stresses the need to build a culture of safety; provide support to the staff; promote reporting; learn and share safety lessons; and implement solutions to prevent harm among the key features of safe practice.24 Organisational culture that fosters a culture of safety encourages shared responsibility instead of blame; where employees are encouraged to voice their safety concerns and the emphasis is on evolving systems based on safe practice instead of expecting humans not to make mistakes. The implication of such processes has been shown to have decrease the number of critical incidents and near misses.25
Incident reporting is central to improving quality of care and minimising errors; the incidence of errors is lower in organisations with a strong system of reporting. The purpose of these reports is not to assign responsibility, but to take corrective measures for the future. Reports, however, do have their limitations; all of the incidents are not reported and it is not agreed on what is the appropriate denominator; total number of anesthetics or total number of drug administrations. Under-reporting was highlighted in two ethnographic studies that looked into the practice of intravenous drug administration. This study identified a 49% and 48% error rate respectively during preparation and administration of drugs in UK and German Hospitals. These errors were severe in a small number of cases and were identified and corrected by another person in the majority of cases.
The reported frequency of drug errors is, however, very low, which makes it difficult to generate class one evidence to prove the efficacy of an intervention or a group of interventions. Recommendations are thus based largely on data extracted from incident reports and hospital information systems based on these reports.
RECOMMENDATIONS
Jensen et al analysed common practices advocated to prevent drug errors. A panel of experts was invited to rate the strength of recommendation, based on their expert opinion in the absence of direct evidence. This was followed by a validation study. The recommendations were analysed and weighted according to expert opinion; grade of recommendation depended on the agreement between the panel of experts.26
- The label on any drug ampoule or syringe should be read carefully before a drug is drawn or injected. Strongly recommended.
- The contents of the label on the ampoule should be optimised according to agreed standard. Strongly recommended.
- Syringes should be labeled (always or almost always). Strongly recommended.
- Formal organisation of drug drawers and workplace. Strongly recommended.
- Labels should be checked specifically by a second person or device. Recommended.
- Errors in intravenous drug administration during anesthesia should be reported or reviewed. Recommended.
- Management of inventory should focus on minimising the risk of drug error by appointing a safety officer or pharmacist in the OR. Recommended.
- Similar packaging and presentation of drugs contribute to drug error and should be avoided where possible. Recommended.
- Drugs should be presented in prefilled syringes (where possible) rather than ampoules. Possibly recommended.
- Drugs should be drawn up by the anesthetist who should label it. Possibly recommended.
- Color coding of drug class according to an agreed national or international standard should be used. Possibly recommended.
- Coding by syringe position or size or needle on the syringe should be used. Possibly recommended.
Experts disagreed on whether drugs should be prefilled or drawn at the time of administration; both were recommended in view of difference of opinion.
There were 81 error reports and 40 pre-error reports, which were analyzed by reviewers to validate these recommendations. In their opinion, strategies number 6 and 12 were ineffective in preventing these errors, whereas, recommendations 1, 5 and 9 were most effective in preventing them. Some of the incidents could not have been prevented with any of the strategies. These mainly involved human factors that included inattention, haste, distraction or fatigue.
Color coding has been a subject of debate. A study looking into the impact of color coded syringe labels on the frequency of drug errors failed to show a benefit, but this could be explained due to a 0.11% reported drug error rate.27 The Council for Scientific Affairs (CSA) of American Medical Association has expressed their reservations on the efficacy of color coding on the grounds that there are a limited number of discernible colors; colors may fade; and there is a high prevalence of color blindness among anaesthetists.28 Merry and colleagues describe this as error management rather than prevention. In the case of an error in reading the name, color coding would ensure that a drug of same class was administered, thus reducing the potential for harm through interclass error.29
Independent double check is not included in the Jensen et al. list, but it has been shown to prevent errors of drug administration.30 It is desirable that drugs should be checked by two persons independently and ideally verbally verified before drawing and administration.
What can anesthesia departments do to prevent/manage drug errors?
- Develop departmental protocols for prescription, storage, drawing, administration and documentation of intravenous drugs in the OR and PACU.
- Promote a culture of reporting drug related errors, no matter how trivial the error and whatever the outcome.
- Each report should initiate a full cycle of analysis → Solution development → Implementation → Audit and monitoring → Feedback → Reporting
- Accept that human beings make errors, but do not tolerate deliberate ignorance/breach of protocol
What can anesthesia consultants do to prevent/manage drug errors?
- Let everyone be aware that drug errors are a universal reality
- Provide a leadership role by developing a culture of safety; reporting errors and encouraging others to do so.
SUMMARY
Problems with the naming, labeling, and packaging of drugs contribute to errors that cause patient injuries and deaths. Names, labels, and packages are not selected and designed in accordance with human factors principles. FDA standards do not require application of these principles, the pharmaceutical industry has resisted change, and private sector initiatives have had only limited success.
When we say drug errors are uncommon, it is mostly because they are underreported. Safety should not rely on human perfection, but rather focus on designing systems, processes, and tasks that make it difficult for people to make mistakes at all. System based strategies; an institutional culture that fosters reporting of errors and encourages everyone to identify potential sources of errors are means to prevent these incidents.
REFERENCES
- Kenagy JW, and Stein GC. Naming, labeling, and packaging of pharmaceuticals. Am J Health Syst Pharm. 2001 Nov 1;58(21):2033-41. [PubMed]
- Bates DW, Cullen D, Laird N, Petersen LA, Small SD, Servi D, et al. Incidence of adverse drug events and potential adverse drug events: implications for prevention. JAMA. 1995;274(1):29-34. [PubMed] [Free Full Text]
- Bates DW, Spell N, Cullen DJ, Burdick E, Laird N, Petersen LA, et al. The costs of adverse drug events in hospitalized patients. JAMA. 1997;277(4):307-11. [PubMed]
- Bates DW, Boyle DL, Vander Vliet MB, Schneider J, Leape L. Relationship between medication errors and adverse drug events. J Gen Intern Med. 1995;10(4):100–205. [PubMed]
- National coordinating council for medication error reporting and prevention. Accessed on July 13, 2013. http://www.nccmerp.org/aboutMedErrors.html
- Look-alike, sound-alike drug names. Sentinel Event Alert, Issue 19, May 2001. Joint Commission.http://www.jointcommission.org/SentinelEvents/SentinelEventAlert/sea_19.htm.
- 2006 National Patient Safety Goals. Oakbrook Terrace, IL, The Joint Commission. Accessed 11 June 2006. http://www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/06_npsg_cah.htm?
- Filik R, Purdy K, Gale A, Gerrett D. Drug name confusion: evaluating the effectiveness of capital (“Tall Man”) letters using eye movement data. Soc Sci Med. 2004;59(12):2597-2601 [PubMed]
- Filik R, Purdy K, Gale A, Gerrett D. Labeling of medicines and patient safety: evaluating methods of reducing drug name confusion. Hum Factors 2006;48(1):39-47 [PubMed]
- Grasha A. Cognitive systems perspective on human performance in the pharmacy: implications for accuracy, effectiveness, and job satisfaction. Alexandria (VA): NACDS; 2000 Report No. 062100
- FDA. Name differentiation project. Center for Drug Evaluation and Research. 2002; Accessed on 18 July 2013. www.fda.gov/CDER/Drug/MedErrors/nameDiff.htm
- The Joint Commission. NPSG: Identify and, at a minimum, annually review a list of look-alike/sound-alike drugs used in the organization, and take action to prevent errors involving the interchange of these drugs; Accessed on 18 July 2013. www.jointcommission.org/NR/rdonlyres/C92AAB3F-A9BD-431C-8628-11DD2D1D53CC/0/LASA.pdf.
- National Association of Boards of Pharmacy. “TALL MAN” letter utilization for look-alike drug names. 2008; Accessed on 18 July 2013. www.nabp.net/ftpfiles/AM/104/104thAMResolutions/(1)%2.
- Institute for Safe Medication Practices. Lists of Look-Alike Drug Names with Recommended Tall Man Letters. Accessed on 15 July 2013. http://www.ismp.org/tools/tallmanletters.pdf.
- Manger D, Miguel R, Villarreal JR. Reporting every look-alike is no longer novel: similarities between labels are a fact of life. Journal of Clinical Anesthesiology 1992;4:347-8
- Nunn DS. Avoiding drug errors. Read the label. BMJ. 1995 Nov 18;311(7016):1367. [PubMed]
- Uncles DR. The importance of checking ampoule labels. Anesthesia 1993;48(2):180-1. [PubMed]
- Medicines and Healthcare products Regulatory Agency. Best Practice Guidance on the labeling and packaging of medicines. MHRA Guidance Note No. 25. June 2003. Accessed on July 20, 2013. http://www.mhra.gov.uk/home/groups/commsic/documents/publication/con007554.pdf.
- APSF Newsletter. Volume 25 Spring 2010. Accessed on 21 July 2013. http://www.apsf.org/downloads/Medication Safety Report.pdf.
- Ostini R, Roughhead E, Kirkpatrick CMJ, Monteith GR, Tell SE.Quality use of Medicines-medication safety isssues in naming; look-alike, sound-alike medicine names. Int J Pharm Prac 2012;20(6):349-357. [PubMed] doi: 10.1111/j.2042-7174.2012.00210.x.
- The Drugs (Labeling and Packing) Rules, 1986. Accessed on 21 July 2013. http://www.healthkp.gov.pk/downloads/Druglabeling.pdf.
- Leape LL. Striving for perfection. Clin Chem 2002;48(11):1871-2. [PubMed]
- Fraind D, Jason S, Victor T, Samuel H, Matthew W. Reengineering Intravenous Drug and Fluid Administrations in the Operating Room: Step One: Task Analysis of Existing Practice. Anesthesiology 2002:97(1):139-47. [PubMed]
- Seven Steps to Patient Safety. NHS. Accessed on 21 July 2013. http://www.nrls.npsa.nhs.uk/resources/collections/seven-steps-to-patient-safety/
- Glavin RJ. Drug errors: consequences, mechanism, and avoidance. Br J Anaesth 2010;105(1):76-82. [PubMed] doi: 10.1093/bja/aeq131.
- Jensen LS, Merry AF, Webster CS, Weller J, Larrson L. Evidence-based strategies for preventing drug administration errors during anesthesia. Anesthesia 2004;59:493-504. [PubMed]
- Fasting S, Gisvold SE. Adverse drug errors in anesthesia and the impact of color coded syringes. Can J Anaesth 2000;47(11):1060-1067. [PubMed]
- J. Chris Hawk, III, MD. The Role of Color Coding in Medication Error Reduction. Report of the council on scientific affairs. CSA Report 5-A-04. Accessed on 21 July 2013. http://www.ama-assn.org/resources/doc/csaph/a04csa5-fulltext.pdf
- Merry AF, Shipp DH, Lowinger JS. The contribution of labelling to safe administration of medication in anaesthetic practice. Best Pract Res Clin Anaesthesiol. 2011;25:145–159. [PubMed] doi: 10.1016/j.bpa.2011.02.009.
- Remote electronic prescribing can lead to wrong patient errors. Nurse Advise-ERR. December 2008 Volume 6 Issue 12. Accessed on 21 July 2013. http://www.ismp.org/Newsletters/nursing/Issues/NurseAdviseERR200812.pdf.