Tariq Hayat Khan, DA, MCPS, FCPS*
*Consultant Anesthesiologist & Pain Specialist, KRLHospital, Islamabad (Pakistan)
Correspondence: Tariq Hayat Khan, Consultant Anesthesiologist & Pain Specialist, KRLHospital, G-9/1, Mauve Area, Islamabad (Pakistan). Cell: +92 321 5149 709; e-mail: tariqhayatkhan@hotmail.com
SUMMARY
Since its introduction into clinical practice in 1921, millions of epidurals are performed daily around the world. Anesthesiologists were quick to grasp its use for prolonged or continuous analgesia and routinely use it alone or in combination with general anesthesia. It has been found to be associated with less complications as compared to spinal analgesia and has taken a major chunk out of spinal practice. The success of epidural depends upon successful location and deposition of drugs into the epidural space. Blind identification of epidural space by loss of resistance or negative epidural space pressure has resulted in mixed results. The figures vary among the researchers but in about 25-30% cases, drugs are deposited at wrong place (outside epidural space). Epidurography offers the best method of confirming the needle or catheter tip location in the space, and has evolved itself a standard practice now. Although very rarely adverse reactions to the technique or the contrast media have been reported, the method is highly recommendable and the pain practitioners and anesthesiologists are encouraged to use it more judiciously.
Key Words: Epidural analgesia; Epidural space; Epidurography; Epidural steroid injections; Contrast medium
Citation: Khna TH. Epidurography. Anaesth Pain & Intensive Care 2009;13(1):31-44
DEFINITION
Epidurography /epi·du·rog·ra·phy/ (-dūr-og´rah-fe) has been defined as radiography of the spine after a radiopaque medium has been injected into the epidural space.1,2
HISTORY
The epidural analgesia owes its birth to some notable surgeons. The first approach to the epidural space was the caudal approach, reported by Sicard and Cathelin (Paris, France) in 1901. The lumbar approach to the epidural space was developed 20 years after the caudal approach was discovered by Sicard and Cathelin in France. Pages in Spain and Dogliotti in Italy appear to have independently developed lumbar epidural anesthesia (EA). In 1921, Fidel Pages (Madrid, Spain, 1886-1923)) reported his work with lumbar EA. He is generally regarded as to be the father of modern EA. In “Anestesia Metamerica”3, he reported 43 cases performed under EA. Cases included inguinal hernia repair, lysis of adhesions, and lower extremity cases. He reported 3 failures and 1 total spinal. Unfortunately, EA was only a minor part of Pages’ practice, his experience with EA was not disseminated to others, and his work was not translated into other languages. He was killed in 1923 in an automobile accident.
A.M. Dogliotti (Turin, Italy, 1897-1966), also a surgeon, first utilized EA in 1931. His book Anesthesia Narcosis Local Regional Spinal 4 was one of the first foreign books on anesthesia to be translated into the English language. He devoted an entire chapter of his book to EA. He advocated the use of EA for a wide variety of procedures. Dogliotti first described the modern loss of resistance technique and classic grip of needle and syringe. Although a surgeon by training, he took a profound interest in anesthesia. Dogliotti was guest lecturer at the ASA Annual Meeting in Seatte, WA (1953) and taught 2 refresher course lectures. In the mean while, in 1932,
Vincent Ruiz and Alberto Gutierrez (Buenos Aires, Argentina) began their practice of EA. They published in the United States in 1939.5 Ruiz and Gutierrez discussed the history of EA (citing Pages, Dogliotti and early practitioners of caudal anesthesia), their technique (“the drop sign”) and most notably, their results with EA. By 1933, 81.5% of all their cases were being done under EA; 12.5% under local; 4% under general; 0.25% under spinal; 1.75% under misc. They used 2% procaine in volumes ranging from 25 to 50 ml. Their reported failure rate was 1%
After the discovery of epidural injections the technique has been refined over a period of time. Epidural injections offered many advantages over more conventional spinal analgesia, e.g. slow onset of hypotension, which gave anesthesiologists time to fill the patients veins with appropriate fluid, lesser incidence of spinal headache and the liberty to approach from cervical to sacral epidural space. The epidural catheters gave the operators an ability to offer analgesia for prolonged periods, which was further refined with the advent of syringe pumps. Touhy needle was presented with a bent tip, which was claimed to dent the dura and not to pierce it, thus minimizing the incidence of accidental dural puncture. It also facilitated threading the catheters in the desired direction. The conventional method of locating epidural space has been by either using the loss of resistance technique or the potential negative pressure within the space by hanging drop technique. Special low resistance syringes were developed to use for loss of resistance technique.
The initial views of the epidural space were accidentally obtained by Sicard and Forestier in 1921 using Lipiodol as the contrast agent6. In 1941, epidurography (EG) was performed with both air7 and Perabrodil 8, an ionic water-soluble contrast agent. Although several terms have been applied to the procedure, including peridurography9,10 and canalography 11, “EG” most accurately
describes the procedure anatomically. Several large European series of lumbar epidurograms
have been reported, including a group of 600 cases 12 and a second of 102 cases 10. The Japanese studied lumbar EG13 and an excellent comparison of lumbar EG, lumbar discography and lumbar venography was done in Australia14.
CONTRAST MEDIA
A medical contrast medium (CM) is a substance used to enhance the contrast of structures or fluids within the body in medical imaging. Iodine based contrast media are usually classified as ionic or non-ionic. Ionic contrast media have higher osmolarity and more side-effects, such as Urografin, Telebrix, Gastrografin, etc. Non-ionic contrast media have lower osmolarity and tend to have less side-effects, such as Omnipaque, Ultravist, Visipaque, etc.
All currently used CM are chemical modifications of a 2,4,6-tri-iodinated benzene ring.15 They are either high-osmolality (600 to 2100 mOsm/kg, versus 290 mOsm/kg for human plasma) or low-osmolality CM which are divided intothree categpries; (1) nonionic monomers, (2) ionic dimers, and (3) nonionic dimers.
In nonionic monomers, the tri-iodinated benzene ring is made water soluble by the addition of hydrophilic hydroxyl groups to organic side chains that are placed at the 1, 3, and 5 positions. At normally used concentrations, 25-76%, nonionic monomers have 290-860 mOsm/kg. Nonionic monomers are subclassified according to the number of milligrams of iodine in 1 mL of solution (eg, 240, 300, or 370 mg I/mL). Common nonionic monomers are iohexol (Omnipaque; GE Healthcare, Inc), iopamidol (Isovue; Bracco Diagnostics Inc), ioversol (Optiray; tyco Healthcare and Mallinckrodt Inc), and iopromide (Ultravist; Bayer HealthCare Pharmaceuticals Inc, Wayne, NJ). These are the contrast agents of choice. In addition to their nonionic nature and lower osmolalities, they are potentially less chemotoxic than the ionic monomers.
The compatibility of bupivacaine (0.25% and 0.125% wt/vol) with iohexol 300 mg I/ml was investigated. At room temperature bupivacaine does not decompose in these mixtures over a period of 24 h. pH Values (7.10 and 7.33), clarity, osmolality (370 and 379 mOsm/kg) and buffer capacity (0.035 ml and 0.010 ml 0.1000 mol/l NaOH per 10 ml) meet requirements for epidural injection. Both mixtures are suitable for EG16.
Verografin. Direct contrasting of the anterior epidural space with 9-10% verografin solution was conducted in 123 patients in 1983. “Spinal epilepsy” occurred in 5 cases, which regressed rapidly after a complex of measures was applied17.
Metrizamide: After iohexol, metrizamide – a water-soluble non-ionic contrast, is probably the most studied CM. Various researchers have studied its characteristics, spread, adverse reactions and compatibilities. Using it has made it possible to perform the examination on an outpatient basis. None of the patients suffer from headache nor any other complaint often associated with other diagnostic techniques which involve lumbar puncture18. In an experimental study of different contrast media, metrizamide has been found to be very safe for EG as no clinical, radiologic, or histologic changes after epidural injection were seen. Next in order was meglumine iothalmate. However, sodium containing salts like sodium acetrazoate and sodium iothalmate were found to be very irritating to the nerve roots and the spinal cord and, therefore, should not be used for epidurographic studies.19-21
Table 1: Commonly used iodinated contrast agents
|
Compound |
Name |
Type |
Iodine Content |
Osmolality |
Level |
| Non-Ionic | Iopamidol (Isovue 370) |
Non-ionic monomer |
370 |
796 |
Low Osmolar |
| Non-Ionic | Iohexol (Omnipaque 350) |
Non-ionic |
350 |
884 |
Low Osmolar |
| Non-Ionic | Ioxilan (Oxilan) |
Non-ionic |
Low Osmolar |
||
| Non-Ionic | Iopromide |
Non-ionic |
Low Osmolar |
||
| Non-Ionic | Iodixanol (Visipaque 320) |
Non-ionic dimer |
320 |
290 |
Iso Osmolar |
The use of epinephrine was claimed to enhances the value of EG procedure by delaying the absorption of metrizamide, allowing thorough plain radiograph evaluation and time to perform tomography22, but its routine use was never popular as the development of fluoroscopy allowed rapid interpretation of the EG.
Adverse reactions:
Adverse effects from the intravascular administration of CM are generally mild and self-limited; reactions that occur from the extravascular use of CM are rare.15 Nonetheless, severe or life-threatening reactions can occur with either route of administration. Radiologists and other physicians must be aware of the risk factors for reactions to contrast media, use strategies to minimize adverse events, and be prepared to promptly recognize and manage any reactions to the contrast media.23-25Adverse reactions to CM are classified as idiosyncratic and nonidiosyncratic.15,24,26,27 Idiosyncratic reactions typically begin within 20 minutes of the CM injection, independent of the dose that is administered. A severe idiosyncratic reaction can occur after an injection of less than 1 ml of a contrast agent.
Although reactions to CM have the same manifestations as anaphylactic reactions, these are not true hypersensitivity reactions. Immunoglobulin E (IgE) antibodies are not involved. In addition, previous sensitization is not required, nor do these reactions consistently recur in a given patient. For these reasons, idiosyncratic reactions to CM are called anaphylactic reactions. The symptoms of anaphylactic reaction can be classified as mild, moderate, and severe.
Mild symptoms; Mild symptoms include the following: scattered urticaria, which is the most commonly reported adverse reaction; pruritus; rhinorrhea; nausea, brief retching, and/or vomiting; diaphoresis; coughing; and dizziness. Patients with mild symptoms should be observed for the progression or evolution of a more severe reaction, which requires treatment.
Moderate symptoms; Moderate symptoms include the following: persistent vomiting; diffuse urticaria; headache; facial edema; laryngeal edema; mild bronchospasm or dyspnea; palpitations, tachycardia, or bradycardia; hypertension; and abdominal cramps.
Severe symptoms; Severe symptoms include the following: life-threatening arrhythmias (ie, ventricular tachycardia), hypotension, overt bronchospasm, laryngeal edema, pulmonary edema, seizures, syncope, and death.
EG after Inadvertent Dural Puncture:
If dura is accidentally punctured, EG should not be attempted, as although this will reveal passage of contrast medium from the epidural space into the subarachnoid space, with opacification of the caudal cul-de-sac, it may be harmful to the patient. Gentili and his colleagues reported a case of contrast entering into dura through a dural puncture and recommend that epidural be substituted with oral or other routes for analgesia, as high spinal may result.28
Routine EG was suggested by Michelangeli and colleagues in 1978 prior to continuous epidural analgesia, in order to ensure the diagnosis of dura mater perforation. In the case of the latter, it shows either a more or less typical appearance of radicolography only or, more rarely, a picture which combines opacification of the epidural space with the subarachnoid passage of the contrast medium. This “mixed” appearance, although rare, should be known since it makes it possible to prevent delayed total spinal anaesthesia.29,30
EG for Epidural Analgesia:
Effective bupivacaine therapy by epidural catheter injections constitutes no striking evidence for a correct epidural catheter position. If the catheter lies in the epidural space, the actual therapeutic effect of bupivacaine therapy is determined mainly by the distribution of the substance. The epidural position of the catheter–central or lateral in the epidural space–seems, however, not to be particularly important. If high-quality analgesia is to be achieved with bupivacaine alone, or in combination with an opioid, the catheter should usually be placed near to, or better into the centre of the segments to be treated. The results demonstrate that in the case of failure of epidural catheter therapy, epidurographic examinations are very helpful in ascertaining and evaluating the underlying reasons for this failure and in coming to a logical decision for changing the concept.31
Perioperative EA in elderly was strongly recommended by Michelangeli in a series of articles. He proved it to be highly beneficial and safe if applied in suitably selected patients.32-33
Even in experienced hands, blind epiduralsteroid injections result in inaccurate needle placement in25 to 30% of cases. Some researchers have placed this figure even at 40% with the use of LOR technique and without EG. The figure was 53% in case of cervical epidurals on first attempt.34* However not all investigators agree with such a high figure of wrong catheter placement. Wulf and his colleagues proved that in 99 of 110 patients they studied, the EG revealed a proper position of the catheter. Seven (6.3%) cases of partial displacement (e.g. paravertebral spread of radiopaque dye) and only two (1.8%) cases of complete misplacement were documented.35
Interventionists were quick to point out high ratio of erroneous placement of the needle tip as well as the epidural catheter (EC), and various methods were studied to rectify this problem. The epidural stimulation test to confirm EC position was described as being simple, fast, and reliable. It required a special EC capable of transmitting stimulation signals, but was regarded as inadequate due to several technical issues (e.g., need to flush catheter with saline in order to maintain adequate stimulation during >25% of all measurements) and interpretation problems (e.g., interference of respiratory activity made the implementation of the epidural stimulation test rather time consuming, both at the time of catheter placement and during epidural analgesia. In this study only epidurographically correct placement of EC ensured 100% effective analgesia in the patients. The use of fluoroscopy and radiologic contrastmaterial provided confirmation of accurate needle or catheter placementwithin the epidural space. 34,36,37
In 1992, Lewis and his colleagues described the ‘whoosh’ test which was previously unreported to confirm accurate needle placement in caudal epidurals. Of 26 patients undergoing caudal epidural, a high number of failures (27%) was noted; 19 (73%) patients had correct needle placement as determined by EG. All of these had a positive ‘whoosh’ test. The ‘whoosh’ test cannot be expected to be a replacement of EG, however.38
The incidence of erroneous placement of needle tip is higher in caudal epidurals. Price and colleagues investigated the accuracy of placement of epidural injections using the lumbar and caudal approaches. They observed that 93% of lumbar and 64% of caudal epidural injections were correctly placed (p< 0.001). 97% of lumbar and 85% of caudal epidural injections clinically thought to be correctly placed were confirmed radiographically. For epidural injections, where the clinical impression was “maybe”, 91% of lumbar injections, but only 45% of caudal injections were correctly placed. They suggested that in the non-obese patient, lumbar epidural injections can be accurately placed without x ray screening, but caudal epidural injections, to be placed accurately, require x ray screening no matter what the weight of the patient.39,40
Hence, EG is by far the only method available to document the correct placement of the epidural needle tip, distribution of the injectate, anddepiction of epidural space disease. EG in conjunction with epidural steroid injections provides for safe and accurate therapeutic injection and is associated with an exceedingly low frequency of untoward sequelae. It can be performed safely on an outpatient basis and does not require sedation or special monitoring.41
EG also provides objective monitoring of tip placement and trajectory of epidural catheters, advantages which argue in favor of more frequent application of this imaging technique42. An epidural block detected postoperatively after continuous psoas compartment block led to X-ray imaging after the injection of contrast medium, which showed a typical EG with the catheter tip projecting into the epidural space. After drawing back the catheter, another X-ray showed a correct placement of the catheter and a typical spreading of the contrast medium and the catheter could be used for pain relief.42,43
Various authors have presented non-consistent data of their experience of success in placing EN with EG. Johnson et al described their experience in a series of 5334 outpatient epidural steroid injections performed after EG with nonionic contrast to confirm needle tip position. Only four minor complications were encountered (0.07%), and the dura was never transgressed. The authors asserted that the subarachnoid injection of corticosteroids is a serious complication that can be avoided with the use of EG.44 In one study, one needle was subarachnoid, one was intra vascular, three were in tissue plains superficial to the epidural space and were apparent only with contrast injection.
EG helped diagnosing following malpositions of epidural catheters in a large group of patients: intravascular misplacement, paravertebral misplacement, paravertebral escape of radiopaque dye and correct distribution after the catheter had been withdrawn 2 cm, intrathecal misplacement, and simultaneous spread of dye in the subarachnoid and epidural space in a patient with preceding dural tap.45
EG AND EPIDURAL ANATOMY
The anatomy of the posterior lumbar epidural space (PLES) has been extensively studied. Besides the anatomists, surgeons, radiologists and anaesthetists have taken an interest in this. However, because each one has considered the PLES from his own specialist field, descriptions are not always concordant. In particular, the reality of a medial partition in the PLES has been suggested by EG and intraoperative observations. Lewit and Sereghy and Luyendijk opened the debate by reporting, on antero-posterior epidurographic films, a clear-cut, medial, vertical and narrow picture which partitioned the PLES. However, this was not constant. Savolaine et al. also recognized this partition on epidurographic CT scans. During laminectomies, Luyendijk has taken photographs of a medial fold of the dura mater which appeared to hold it to the posterior vertebral arch, being collapsed on either side of the midline. He named it “plica mediana dorsalis durae matris” (PMD). Several anaesthetists considered that this could explain why epidural analgesia sometimes acted on one side only. Blomberg tried to see this space by epiduroscopy in the cadaver. Unfortunately, for technical reasons, his photographs were of poor quality. He, however, reported having seen each time the PMD and a band of connective tissue fixing it to the vertebral arch in the midline. However, all these anatomical studies used methods which alter the natural structures. Their results are therefore questionable. The PLES is a virtual space. Histological studies have shown that it is filled with fatty tissue between the dura and the vertebral arch. It is therefore conceivable that any liquid injected into the PLES, such as contrast medium or local anaesthetic, must push back the dura, the only tissue which can move to give it any room. The fatty tissue could therefore be compressed and take any of the shapes which have been described on EG. On the other hand, should it be torn, it seems this fatty tissue could make up these haphazard fibrous tracts tensed between the dura and the vertebral arch, such as described in classical anatomy, as Bonica recalled. These can be clearly seen during surgical and anatomical dissections, and during endoscopies carried out on cadavers with sufficient optical means, as opposed to the medial fibrous band fixing the dura to the vertebral arch. These fibrous tissue strands and plica mediana dorsalis may be responsible for unilateral epidural analgesia, entrapment and coiling of epidural catheters.46-50
The application of current-generation computed tomography scanning to lumbar EG has resulted in a better appreciation of epidural anatomy and improved ease of interpretation of non-ionic contrast material within the epidural space. The development of extended window width has greatly improved imaging of the more variable levels of attenuation encountered in contrast material within the epidural space.51 Savolaine and colleagues used CT-EG to demonstrate the posterior epidural space to be divided by the plica mediana dorsalis and an additional transverse connective tissue plane not previously described. They were able to demonstrate a great amount of fatty tissue within the junctions of the posterior midline epidural connective tissue structures, producing a bulky triangular-shaped structure which might be an impediment to catheterization.46
SEDATION
The use of sedation to perform these procedures is unnecessary, and deep sedation may be dangerous when injections are performed in the vicinity of the spinal cord. Johnson has demonstrated wide experience, which includes clinical feedback and formal trials, and shows the technique to be performed safely without sedation in an outpatient setting, with a high success rate for alleviating pain symptoms. In one of his studies comprising of 5334 cases he used sedation only in a few very anxious and restless patients.41,52,53
DIAGNOSTIC VALUE
EG has been claimed to be useful to predict the dermatomal distribution of analgesic block because it is impossible to predict the cephalad and caudal spread of anesthetic, whether it will spread unilaterally and the general pattern of spread without EG.54 It is sufficiently accurate to be regarded as the examination of choice in the diagnosis of lumbar disc prolapse.55It can help in diagnosis of definite abnormalities. It was of particular value in the diagnosis of lateral canal stenosis56, and was used to determine the localization, severity and nature of pathological changes. Spinal arachnoiditis and epiduritis are associated with non-filling or non-uniform filling of the membranous spaces, their narrowing and deformation, non-filling and deformation of radicular pockets. The diagnosis is made by correlating clinical, laboratory and radiological findings.
EG was used to exclude disc prolapse in patients with nonhernial form of osteochondrosis and it permitted to determine the initial growth of the tumor, its size and relation to the dura mater.58
Lumbar EG serves as an important radiographic procedure in the evaluation of patients with equivocal myelography and/or confusing or nondiagnostic physical findings. It is particularly valuable in patients with a wide ventral epidural space secondary to previous surgery, arachnoiditis or congenital condition, most commonly at the L5-S1 disk space.
EG was useful for identifying the epidural space and determining the likely spread of an epidural blood patch in an unconscious patient with spontaneous intracranial hypotension.55 Epidural blood patch with blood alone, when used to treat postdural puncture headache, and examined with computed tomography in order to demonstrate the distribution of the injected blood, could not be identified, but adding 2 ml contrast agent Iohexol 180 mg J/ml (Omnipaque, Nycomed Imaging) to 18 ml blood gave an excellent demonstration of the distribution of the blood in the epidural space, both cranio-caudally (7-14 segments) and spatially in relation to the epidural septae. The blood-contrast media has a strong affinity to the dural sac.59
The review of repeated epidurograms and presented case reports support the use of epidurograms as a diagnostic tool for clinical practice. Abnormal epidurograms helped determine epidural space infection, tumor obstruction, space compression from vertebral compression fractures, and epidural fibrosis. The addition of a contrast CT after EG allows for more accurate diagnostic interpretation of epidural space pathology. The use of a standard epidurogram technique allows this procedure to be used in the differential diagnosis of suspected problems in the epidural space.60
EG might confirm epidural filling defects for contrast dye in the patients with epidural fibrosis. However, a better contrast dye spread, assuming scar lysis, after repeated injections of contrast dye, local anesthetic, corticosteroid, and hypertonic NaCl 10% does not guarantee a sustained pain relief. A more direct visualization of the resulting functional changes after adhesiolysis as with epiduroscopy might be useful.61 Abnormalities identified with EG correlate well with surgically confirmed lesions.62
Table 2: LESI procedures and initial needle tip position with loss of resistance as seen in the lateral projection
|
Initial Needle Tip Position |
# LESI Procedures |
% |
Adjacent to Spinal Canal |
Mid Spinous Process |
| Extracanal position |
19 |
26 |
15 |
4 |
| Epidural position |
55 |
74 |
||
| Total |
74 |
100 |
15 (20.3%) |
4 (5.4%) |
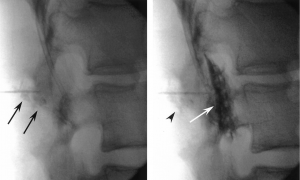
FIG 1. Lateral fluoroscopic images of during LESI procedure. A, Contrast medium injected at loss of resistance is seen overlying the deep paraspinal region just posterior to the ligamentum flavum and spinal canal (arrows). B, The needle advanced and the correct needle-tip location is confirmed with epidurogram (white arrow). Initial injection is also visible (black arrowhead).
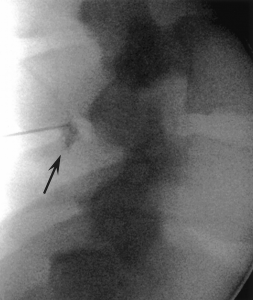
FIG 2. Lateral fluoroscopic images during LESI procedure. Contrast medium injected at point of loss of resistance is seen overlying the spinous process still within the midback soft tissues (arrow).
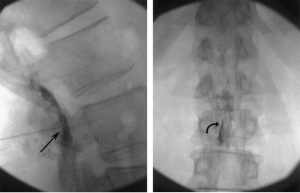
FIG 3. A and B, Lateral and anteroposterior fluoroscopic images of proper position of the needle tip after initial loss of resistance in the posterior epidural space in the lateral (arrow) and anteroposterior (curved arrow) projections.
A pure Seldinger technique with a caudal approach through the sacral notch and selective nerve root sheath injection affords the best technical results.
CONTRAST FLOW
There is great variability in the distribution of contrast medium according to the individual morphology of the epidural space and the way it is injected. Results of investigations of the anatomy of the spinal canal and epidural space performed with anatomic specimens, epiduroscopy, and conventional and computed tomographic (CT) EG are sometimes contradictory.63
Intralaminar approach; The spread of the injectate through EN has been a major issue for research. It was described that the drugs injected through intralaminar approach may remain accumulate in the dorsal ES and may not spread to the ventral aspect. The cephalad or caudal spread of the drugs was also extensively discussed. Injection of a contrast material with subsequent EG was found helpful.
Botwin and his colleagues, in a prospective case series of 25 (twenty-five) consecutive patients with either lumbar spinal stenosis (LSS) or herniated nucleus pulposus (HNP) used a total of 5 ml contrast media and obtained AP and lateral radiographs to study the contrast spread. They found that the dorsal contrast flow occurred in all 25 injections. Thirty-six percent (9 out of 25) resulted in ventral spread of contrast. Eighty-four percent (21 out of 25) of the injections had flow of contrast unilaterally and only 16% (4 out of 25) was bilateral. The mean number of levels of flow of contrast cephalad from the injection site was 1.28 and caudally 0.88. In their study there was a significant difference in more cephalad than caudal contrast flow (P = 0.004). 64
The effect on contrast spread in lumbar EG with multi-orifice and single orifice epidural catheters was studied by Magides and colleagues and they found that there was no significant difference in the total number of vertebral segments covered by dye or the total number of segments reached above or below the level of epidural insertion, spread was more even to the right and left of midline using the single orifice catheters. Sacral spread of dye was only observed when a single orifice catheter was used. They concluded that epidural catheter design affects the distribution of solutions in the epidural space, with single orifice epidural catheters producing more even distribution of dye with sacral extension.65
When small volumes (5 ml) of iopamidol were injected slowly in thoracic epidural catheters, a railroad-track phenomenon was observed regularly in the AP images of conventional epidurographies. After rapid injection of larger volumes (10-15 ml), the spread was more homogeneous and sometimes outline a lighter zone of contrast distribution in the midline. CT EG in these cases revealed a plica mediana dorsalis. In the majority of the CT scans of the thoracic epidural space its anterior (ventral) compartment was unfilled by contrast medium. According to the findings of several investigators, dura mater and ligamentum longitudinal posterius have grown together, so that an epidural space does not exist in ventral thoracic segments of the spinal canal. In lower thoracic and lumbar segments both structures are separate, and a wide anterior epidural space was present and filled with contrast medium. In other cases the dorsal and dorsolateral epidural space was completely filled, but the contrast medium stopped behind the spinal nerves and surrounding dural sheaths, as if these structures, together with the connective tissue strands between them, formed a membrane that spread in a frontal plane. A railroad-track phenomenon is interpreted in the literature as the X-ray correlate of contrast medium spread in the subdural space, indicating an incorrect catheter position. According to conventional epidurographies and confirmed by CT scans, this feature is also seen regularly in cases with correct catheter position. Small volumes of iopamidol spread preferentially into the wider parts of the epidural space between the gaps of the vertebral arcs, whereas the dura mater and periosteal layers touch at the inner side of the pedicules and laminae, allowing the existence of only a potential epidural space. This rhythmic widening and narrowing of the epidural space is the explanation of the “railroad track” in AP contrast epidurographies.66
In cervical epidurals, Stojanovic et al found unilateral epidural contrast spread in 51% and ventral epidural spread in 28% of cases. The average number of cervical vertebral levels covered with 2 mL of contrast was 3.14, with significantly wider spread noted in those patients who had not undergone previous cervical laminectomy15 Two milliliters of contrast solution in fluoroscopically guided cervical interlaminar epidural injections using the midline approach can provide optimal dispersion of contrast in a ventral and longitudinal spread.67
Cranial spread of contrast in the high thoracic epidural space is limited. However, neck flexion increases cranial spread. When thoracic epidural analgesia (TEA) is high, the tip of the epidural catheter should be located at the upper part of the level to be blocked and that neck flexion may cause an unwanted cervical block.68
In a study by Tomczak and his colleagues, they proved that in EG there is a railroad track phenomenon. They proved that it was a sign of a rhythmically variable filling of segments of the lateral epidural space with contrast medium. 76% of the patients demonstrated no ventral epidural space at the thoracic level. 56% of the patients showed a medial area of translucence combined with a band-shaped contrasting of the epidural space in the standard AP view. This was proven in all cases to be a plica mediana dorsalis by CT.69
Transforaminal approach; This approach has been advocated for selective nerve root blocks for radicular pains, and in pain associated with disc prolapse or vertebral body disease. The spread of contrast by this route is consistently higher in the ventral side than dorsal flow. The side affected most by the disease can be selected for injection and spread achieved will be higher on this side with only rarely a cross over to the opposite side. There have been no significant differences in contrast flow noted between patients with herniated nucleus pulposus or lumbar spinal stenosis. Vascular injection patterns were noted with 2 injections, which required repositioning of the needles.70
The contrast flowed cephalad only in 20%, caudad only in 28%, and bidirectional in 52%. Contrast spread less than three spinal levels 64% of the time. The desired nerve root was visualized in 62%. Contrast was seen in the ventral epidural space on lateral views 88% of the time. Ventral spread was always fewer levels than the dorsal spread. 71
A significant correlation has been found between the longitudinal spread of iohexol and aging, but no correlation was found between the leakage of iohexol from the intervertebral foramina and aging. This finding may deny the hypothesis that in elderly patients, the more extensive longitudinal spread of local anesthetics in the epidural space may be caused by its smaller leakage from the intervertebral foramina. 71
EG in prolonged EA:
Prolonged use of EA is very common now a days to treat the cancer pain or other painful ailments. In this case an EC may become displaced, leading to unilateral or no analgesia despite adequate loading of the drugs. Permanent EC’s for continuous analgesia may also cause pressure on the dural sac and become encapsulated, mimicking a growth of the dura.72
EG plus CT/MRI:
Combining CT with EG has been a major breakthrough in diagnosing epidural failures. It was first described perhaps by Hårdstedt and Vucetic in 1986 as a method of diagnosis of disc herniation and nerve root compression.73
Some Japanese investigators74 have shown that local anaesthetics injected into the epidural space may deform the dural sac to a variable degree, thereby contributing to variability in the extent of the block. They used ten millilitres of local anaesthetic injected into the epidural space followed by 20 mL of contrast medium. Computed tomographic EG was undertaken approximately 30-min after the epidural injection at the mid-vertebral and mid-discal levels from the first lumbar through to the first sacral vertebrae. The dural sac usually showed an oval or hexagonal shape on the transverse views at the first and second lumbar vertebral levels, and the shape of an inverted triangle below the level of the third lumbar vertebra. A median line of translucency was also observed on the posteroanterior epidurographic view in 25 of the 26 patients. This line was though to be a manifestation of the dural deformation to the inverted triangle. Dural sac deformation usually shows a specific pattern, although there are individual variations. Dural deformability is an important consideration in any analysis of the spread of epidural block or of the changes of epidural pressure after epidural injection of local anaesthetics. 74
Computed tomography (CT) epidurographies have been used to analyze epidural analgesia failure after major abdominal surgery to compare the incidence of dislodgement of epidural catheters and leakage of solution from the epidural space. When failure was not due to unintentionally withdrawn, kinked catheters or adverse events, a CT scan with contrast injection was helpful in demonstrating unidirectional spread, intravascular or peripheral leakage of the contrast allowing reintroduction of the catheters. The major cause of epidural analgesia failure has been shown to be dislodgment of the catheter. CT scans can be useful for detecting leakage of injectate, which may be the early phase of dislodgment. 75
CT EG is well suited for obtaining new insights into the interpretation of findings obtained by conventional EG. After injection of Gadolinium-DTPA MR-EG or helical-CT EG using twin-beam technology 40% of the patients demonstrated the railroad track phenomenon.76 CT EG is a valuable tool to provide better insight into the morphology of the epidural space when filled with fluid (contrast medium, local anaesthetics), complementing findings using epiduroscopy and anatomic specimens. It stands to reason that this time- and cost-expensive method can never replace conventional epidurographies, but can help to interpret them properly.76,77
MRI EG has been presented as a new imaging tool in which MR EG is done after injection of 20 ml Gd-DPTA solution (1:250/1 ml Gd-DPTA/250 ml normal saline). Magnetic resonance EG is possible. With fat-suppression techniques, the contrast between Gd-DPTA solution in the epidural space and surrounding soft tissue proves adequate. In the 3+ oxidation state the metal has 7 unpaired f electrons. This causes water around the contrast agent to relax quickly, enhancing the quality of the MRI scan. Nephrogenic systemic fibrosis can appear through the administration of gadolinium for MR contrast enhancement. Although rare and only in renally compromised patients, it produces serious side-effects that may involve fibrosis of skin, joints, eyes, and internal organs. Using the multiplanar capability of MRI with MR EG coronal and sagittal projections similar to conventional EG, axial slices comparable to CT EG can be obtained. Magnetic resonance EG is superior to conventional and CT EG. Presently, due to high costs as compared with conventional and CT EG, MRI is not suitable for the routine monitoring of peridural catheters, but it may have a place in the future with decreasing costs for MRI and for the evaluation of patients with spine pathology, especially in describing epidural processes.76,78
INTRAOPERATIVE CERVICAL EG
Intraoperative cervical EG (ICE) has been used during anterior cervical procedures to assess the success of bone resection and indicate the need for immediate correction in the event of inadequate decompression. In one study, the ICE revealed insufficient resections requiring additional bone removal in 14 (36%) of the 39 patients. Furthermore, ICE provides additional real-time control of the position of cages and screws to avoid dural tear or spinal cord compression. Because of this immediate feedback, the success rate of anterior cervical procedures can be improved. The advantages of this modality include real-time assessment, low cost, simplicity, and speed.
COMPLICATIONS
Complications that may occur are usually due to epidural intervention, and may be linked to poor technique or faulty preparation including non-adherence to absolute asepsis. Using the standard techniques described, complications are minimized, and serious complications can be avoided, in experienced hands. Optimal safety and efficacy require an excellent working knowledge of the radiographic anatomy, and the imaging equipment used to perform these procedures. 52
The spinal epilepsy syndrome developed in 17 of 494 patients who were subjected to EG. This syndrome developed evidently with a water-soluble iodine-containing contrast medium gaining entry into the subarachnoid space or due to chance puncture of the dura mater during the manipulation or when the contrast medium flows through an opening in it which failed to close after a previous lumbar puncture. It was recommended that to prevent convulsions, EG should be conducted under careful control of the position of the needle in the epidural space and no earlier than 10 days after lumbar puncture. 79 Subdural introduction of metrizamide may produce headache, and nausea / vomiting. Fever up to 38 degrees C was observed on the first day after the procedure in 28% of cases.80
CONCLUSION
Imaging and epidurogram are essential for confident identification of the epidural space to enable accurate location of steroid and/or local anesthetic agent administration. Studies assessing efficacy of epidural analgesia, epidural steroid injection and individual patient treatments should ensure location of administration with epidurogram to enhance the validity of results. EG is associated with an exceedingly low frequency of untoward sequelae. It can be performed safely on an outpatient basis and does not require sedation or special monitoring.
REFERENCES
1. Dorland’s Medical Dictionary for Health Consumers. © 2007 by Saunders, an imprint of Elsevier, Inc.
2. Saunders Comprehensive Veterinary Dictionary, 3 ed. © 2007 Elsevier, Inc.
3. Pages F: Anestesia metamerica. Rev de la sanidad militar. 11: 351-65; 385-96, 1921.
4. Dogliotti AM: Anesthesia Narcosis.Local.Regional.Spinal . Chicago, SB Debour, 1939.
5. Ruiz V: Pages’ Peridural Anesthesia A Report of 3,826 Cases. Minn Med 22: 363-68, 1939.
6. Sicard JA, Forestier J: M#{233}thodraediographique d’exploration de Ia avit #{23#3{2}33} pidtraplear le Lipiodol. Rev Neurol 28:1264-1266, 1921
7. Sanford H, Doub HP: Epidurography: a method of roentgenologic visualization of protruded intervertebral disks. Radiology 36:712-716, Jun 1941
8. Knutsson F: Experiences with epidural contrast investigation of the lumbosacral canal in disc prolapse. Acta Radiol 22: 694-703, 1941
9. Lewit K, Sereghy T: Lumbar peridurography with special regard to the anatomy of the lumbar peridural space. Neuroradiology. 8:233-240, 1975
10. Lewit K: The contribution of peridurography to the anatomy of the lumbosacral spinal canal. Folla Morphol (Praha) 24:289-295,1976
11. Luyendijk W: Canalography. Roentgenological examination of the peridural space in the lumbosacral part of the vertebral canal. J BeIg Radiol 46:236-254, 1963
12. Luyendijk W, van Voorthuisen AE: Contrast examination of the spinal and epidural space. Acts Radiol (Diag) 5:1051-1066, 1966
13. Nagamine K: Clinical and biochemical study of accidents in peridurography. Nagoya J Med Sci 32:429-444, Mar 1970
14. McCormick CC: Radiology in low back pain and sciatica. An analysis of the relative efficacy of spinal venography. Dlscoıaphy and epidurography in patients with a negative or equivocal myelogram. Clin Radiol 29:393-406, 1978
15. American College of Radiology. Manual on Contrast Media. 4th ed. Reston, Va: ACR; 1998.
16. Van Asten P, Glerum JH, Spaanderman ER, Van Niekerk J, Van Dijk A. Compatibility of bupivacaine and iohexol in two mixtures for paediatric regional anaesthesia. Pharm Weekbl Sci. 1991 Dec 13;13(6):254-6.
17. Matvienko VI, Siuremov MN. Anterior epidurography with verographin. Zh Vopr Neirokhir Im N N Burdenko. 1983;(1):40-3.
18. Hamilton G. Metrizamide epidurography. J R Soc Med.1983;76(2):126-30.
19. Hatten HP Jr. Lumbar epidurography with metrizamide. Radiology.1980;137(1 Pt 1):129-36.
20. Gupta RC, Gupta SC, Dubey RK. An experimental study of different contrast media in the epidural space. Spine. 1984;9(8):778-81.
21. Emery I, Hamilton G. Epidurography using metrizamide an out-patient examination. Clin Radiol.1980;31(6):643-9.
22. Easterbrook JS, Hibri NS. Epidurography using epinephrine and tomography. Radiology. 1981 Sep;140(3):709-11.
23. Gaca AM, Frush DP, Hohenhaus SM, et al. Enhancing pediatric safety: using simulation to assess radiology resident preparedness for anaphylaxis from intravenous contrast media. Radiology. Oct 2007;245(1):236-44.
24. Cutroneo P, Polimeni G, Curcuruto R, Calapai G, Caputi AP. Adverse reactions to contrast media: an analysis from spontaneous reporting data. Pharmacol Res. Jul 2007;56(1):35-41.
25. Cohan RH, Ellis JH. Iodinated contrast material in uroradiology. Choice of agent and management of complications. Urol Clin North Am. Aug 1997;24(3):471-91.
26. Bush WH, Swanson DP. Acute reactions to intravascular contrast media: types, risk factors, recognition, and specific treatment. AJR Am J Roentgenol. Dec 1991;157(6):1153-61.
27. Becker C. [Prophylaxis and treatment of side effects due to iodinated contrast media relevant to radiological practice] [German]. Radiologe. Sep 2007;47(9):768-73.
28. Gentili ME, Samii K. Iterative epidural anesthesia after accidental dural puncture. Analysis with epidurography. Ann Fr Anesth Reanim. 1991;10(6):580-2.]
29. Michelangeli F, Feyler C, Laufer A, Pras N, Migliori G, Maestracci P. Methods and indications for low peridural analgesia in the aged. Report of 300 cases. Ann Anesthesiol Fr. 1977;18(5-6):524-34.
30. Michelangeli F, Feyler C, Donsa E, Carlon M, Maestracci P. Perforations of the dura mater during epidural anesthesia. The value of epidurography. Ann Anesthesiol Fr. 1978;19(6):497-537.
31. Marquort H, Grenzer G, Schroeder U. Routine postoperative epidural analgesia. X-ray control of epidural catheter position and prevention of the spread of epidural contrast media. Anaesthesist. 1993;42(8):501-8.
32. Michelangeli F, Feyler C, Laufer A, Pras N, Migliori G, Maestracci P. Methods and indications for low peridural analgesia in the aged. Report of 300 cases. Ann Anesthesiol Fr. 1977;18(5-6):524-34.
33. Michelangeli F, Feyler C, Migliori G, Maestracci P. Epidurography in the continuous peridural anesthesia technic in geriatric surgery. Ann Anesthesiol Fr.1976;17(5):533-49.
34. Stojanovic MP, Vu TN, Caneris O, SlezakJ, Cohen SP, Sang CN. The role of fluoroscopy in cervical epidural steroid injections: an analysis of contrast dispersal patterns. Spine. 2002 Mar 1;27(5):509-14.
35. Wulf H, Kibbel K, Mercker S, Maier C, Gleim M, Crayen E. Radiologic position control of epidural catheters (epidurography). An instrument of quality assurance for regional analgesia. Anaesthesist. 1993 Aug;42(8):536-44.
36. Bartynski WS, Grahovac SZ, Rothfus WE. Incorrect needle position during lumbar epidural steroid administration: inaccuracy of loss of air pressure resistance and requirement of fluoroscopy and epidurography during needle insertion. AJNR Am J Neuroradiol. 2005 Mar;26(3):502-5.
37. Förster JG, Niemi TT, Salmenperä MT, Ikonen S, Rosenberg PH. An evaluation of the epidural catheter position by epidural nerve stimulation in conjunction with continuous epidural analgesia in adult surgical patients. Anesth Analg. 2009;108(1):351-8.
38. Lewis MP, Thomas P, Wilson LF, Mulholland RC. The ‘whoosh’ test. A clinical test to confirm correct needle placement in caudal epidural injections. Anaesthesia. 1992 Jan;47(1):57-8.
39. Price CM, Rogers PD, Prosser AS, Arden NK. Comparison of the caudal and lumbar approaches to the epidural space. Ann Rheum Dis. 2000 Nov;59(11):879-82.
40. Price CM, Rogers PD, Prosser AS, Arden NK. Comparison of the caudal and lumbar approaches to the epidural space. Ann Rheum Dis. 2001 Jul;60(7):718.
41. Johnson BA, Schellhas KP, Pollei SR.Epidurography and therapeutic epidural injections: technical considerations and experience with 5334 cases. AJNR Am J Neuroradiol. 1999;20(4):697-705.
42. Díez Rodríguez-Labajo A, Reinoso-Barbero F, Sanabria P, Rodríguez E, Suso B. Usefulness of radiologic monitoring of epidural catheters using epidurography. Rev Esp Anestesiol Reanim. 1998 Dec;45(10):416-20.
43. Rotzinger M, Neuburger M, Kaiser H. Inadvertant epidural placement of a psoas compartment catheter. Case report of a rare complication. Anaesthesist. 2004 Nov;53(11):1069-72.
44. Johnson et al. American Journal of Neuroradiology 1999:20(4); 697
45. Wulf H, Kibbel K, Mercker S, Maier C, Gleim M, Crayen E. Radiologic position control of epidural catheters (epidurography). An instrument of quality assurance for regional analgesia. Anaesthesist. 1993 Aug;42(8):536-44.
46. Savolaine ER, Pandya JB, Greenblatt SH, Conover SR. Anatomy of the human lumbar epidural space: new insights using CT-epidurography. Anesthesiology. 1988 Feb;68(2):217-20.
47. Kikuchi S. Anatomical and experimental studies of nerve root infiltration. Nippon Seikeigeka Gakkai Zasshi.1982;56(7):605-14.
48. Morisot P. Is posterior lumbar epidural space partitioned? Ann Fr Anesth Reanim. 1992;11(1):72-81.
49. Seeling W, Rockemann M. Unilateral epidural anesthesia. Reg Anaesth.1990 Mar;13(2):23-8.
50. Boezaart AP. Computerized axial tomo-epidurographic and radiographic documentation of unilateral epidural analgesia. Can J Anaesth.1989;36(6):697-700.
51. Savolaine ER, Greenblatt SH, Conover SR, Lipton S. Computed tomography application to lumbar epidurography. J Comput Tomogr. 1987 Apr;11(2):193-9.
52. Johnson BA. Image-guided epidural injections. Neuroimaging Clin N Am. 2000;10(3):479-91.
53. Johnson BA, Schellhas KP, Pollei SR.Epidurography and therapeutic epidural injections: technical considerations and experience with 5334 cases. AJNR Am J Neuroradiol. 1999;20(4):697-705.
54. Nakatsuka H, Takahashi T, Matsumi M, Takeuchi M, Morita K, Yokoyama M, Hanazaki M, Fujii H, Mizobuchi S. Can Epidurography Help to Predict the Extent of Epidural Blockade? Anesthesiology.2005;102(2):479
55. Terajima K, Oi Y, Ogura A, Sakai N, Takei M, Tamaki T, Ogawa R. Blood patch therapy for spontaneous intracranial hypotension: safe performance after epidurography in an unconscious patient. Anesth Analg. 2002 Apr;94(4):959-61.
56. Stewart HD, Quinnell RC, Dann N. Epidurography in the management of sciatica. Br J Rheumatol. 1987 Dec;26(6):424-9.
57. Sipukhin IaM, Galin IuM, Khmaruk LP. Clinico-roentgenologic diagnosis of spinal arachnoiditis and epiduritis. Zh Nevropatol Psikhiatr Im S S Korsakova. 1985;85(12):1792-6.
58. German DG, Vychuzhanin VS. Verografin epidurography in the diagnosis of spinal and spondylogenic diseases. Zh Nevropatol Psikhiatr Im S S Korsakova. 1979;79(5):546-50.
59. Djurhuus H, Rasmussen M, Jensen EH. Epidural blood patch illustrated by CT-epidurography. Acta Anaesthesiol Scand. 1995;39(5):613-7.
60. Du Pen SL, Williams AR, Feldman RK. Epidurograms in the management of patients with long-term epidural catheters. Reg Anesth.1996;21(1):61-7.
61. Devulder J, Bogaert L, Castille F, Moerman A, Rolly G. Relevance of epidurography and epidural adhesiolysis in chronic failed back surgery patients. Clin J Pain.1995;11(2):147-50.
62. Roberts RE, Selcer BA. Myelography and epidurography. Vet Clin North Am Small Anim Pract. 1993 Mar;23(2):307-29.
63. Seeling W, Tomczak R, Merk J, Mrakovcić N. CT-epidurography. A comparison of conventional and CT-epidurography with contrast medium injection through a thoracic peridural catheter. Anaesthesist. 1995 Jan;44(1):24-36.
64. Botwin KP, Natalicchio J, Hanna A. Fluoroscopic guided lumbar interlaminar epidural injections: a prospective evaluation of epidurography contrast patterns and anatomical review of the epidural space. Pain Physician. 2004;7(1):77-80.
65. Magides AD, Sprigg A, Richmond MN. Lumbar epidurography with multi-orifice and single orifice epidural catheters. Anaesthesia. 1996;51(8):757-63.
66. Seeling W, Tomczak R, Merk J, Mrakovcić N. CT-epidurography. A comparison of conventional and CT-epidurography with contrast medium injection through a thoracic peridural catheter. Anaesthesist. 1995 Jan;44(1):24-36.
67. Kim KS, Shin SS, Kim TS, Jeong CY, Yoon MH, Choi JI. Fluoroscopically guided cervical interlaminar epidural injections using the midline approach: an analysis of epidurography contrast patterns. Anesth Analg. 2009;108(5):1658-61.
68. Lee CJ, Jeon Y, Lim YJ, Bahk JH, Kim YC, Lee SC, Kim CS. The influence of neck flexion and extension on the distribution of contrast medium in the high thoracic epidural space. Anesth Analg. 2007;104(6):1583-6.
69. Tomczak R, Seeling W, Rieber A, Sokiranski R, Rilinger N, Brambs HJ. Epidurography: comparison with CT, spiral CT and MR epidurography. Rofo. 1996 Aug;165(2):123-9.
70. Botwin K, Natalicchio J, Brown LA. Epidurography contrast patterns with fluoroscopic guided lumbar transforaminal epidural injections:a prospective evaluation. Pain Physician. 2004;7(2):211-5.
71. Saitoh K, Hirabayashi Y, Horiguchi Y, Fukuda H, Tsukamoto N, Mitsuhata H, Shimizu R. Lumbar epidurography with iohexol: the effect of aging on the leakage of contrast media from the intervertebral foramina. Masui. 1995 Jan;44(1):66-9.
72. Brems-Dalgaard E, Just SL, Pedersen H. Encapsulated epidural catheter. CT findings. Neuroradiology. 1991;33(1):81-2.
73. Hårdstedt C, Vucetic N. Lumbosacral epidurography and computed tomography. A preliminary report. Acta Radiol Diagn (Stockh). 1986 Mar-Apr;27(2):173-7.
74. Fukushige T, Kano T, Sano T, Irie M. Computed tomographic epidurography: an aid to understanding deformation of the lumbar dural sac by epidural injections. Eur J Anaesthesiol. 1999 Sep;16(9):628-33.
75. Motamed C, Farhat F, Rémérand F, Stéphanazzi J, Laplanche A, Jayr C. An analysis of postoperative epidural analgesia failure by computed tomography epidurography. Anesth Analg. 2006;103(4):1026-32.
76. Tomczak R, Seeling W, Rieber A, Sokiranski R, Rilinger N, Brambs HJ. Epidurography: comparison with CT, spiral CT and MR epidurography. Rofo. 1996 Aug;165(2):123-9.
77. Seeling W, Tomczak R, Merk J, Mrakovcić N. CT-epidurography. A comparison of conventional and CT-epidurography with contrast medium injection through a thoracic peridural catheter. Anaesthesist. 1995 Jan;44(1):24-36.
78. Tomczak RJ, Seeling W, Mergo P, Rieber A, Aschoff A, Brambs HJ. Magnetic resonance epidurography with gadolinium-DTPA. Eur Radiol. 1998;8(8):1452-4.
79. Margolin GA, Zelentsov EV. Possibilities for preventing complications of epidurography. Zh Vopr Neirokhir Im N N Burdenko. 1986 Jan-Feb;(1):50-2.
80. Smereczyński A, Gdakowicz B. Radiodiagnosis of the spinal canal using a nonionic contrast medium. Neurol Neurochir Pol.1981;15(5-6):581-4.