Seyed Mohammad Reza Hashemian1,2, Payam Tabarsi2,3, Majid Malek Mohammad, Majid Marjan2, Masoud Shamaei2, Seyed Alireza Nadji5, Mandana Chitsazan1, Golnar Radmand1, Seyed Amir Mohajerani4, Hamid Reza Jamaati1
1Chronic Respiratory Disease Research Center, NRITLD, Masih Daneshvari Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
2Clinical Tuberculosis and Epidemiology Research Center, NRITLD, Masih Daneshvari Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
3Mycobacteriology Research Center, NRITLD, Masih Daneshvari Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
4Tracheal Disease Research Center, National Research Institute of Tuberculosis and Lung Diseases, Shahid Beheshti University of Medical Sciences, Tehran, Iran
5 Virology Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Correspondence: Hamidreza Jamaati, Masih Daneshvari Hospital, Darabad, Niavaran Sq, Tehran (Iran); Tel/Fax: 009821-26109512; E-mail: Hrjamaati@NRITLD.ac.ir
ABSTRACT
Background: An outbreak of antibiotic resistant gram-negative bacteria, particularly Acinetobacter, in intensive care unit is of great importance for intensivists due to high mortality and low effective therapy.
Objective: We aimed to evaluate antibiotic susceptibility of isolated gram-negatives from patients with ventilator-associated pneumonia (VAP) in ICU.
Material and Methods: In a prospective, descriptive study, ICU patients who were under mechanical ventilation and had resistant ventilator-associated pneumonia (VAP) were evaluated for prevalence and antibiotic susceptibility of identified pathogens. The data, including clinical outcome and microbiological results were analyzed.
Results: Fifteen ICU patients were included with mean age of 50.5 years (the range between 20-96 years). Median of hospitalization was 53 days (the range of 15 to 135 days), median of hospitalization before admission to ICU was 2.5 day (the range was from zero to 27 days), and median days of bacterial isolation was 18 days (the range of 5 to 30 days). Six patients (42.9%) patients were admitted in ICU after surgery, and 8 patients (57.1%) due to another medical problem. Acinetobacter was dominant isolated pathogen in VAP patients. Imipenem did better than other antibacterials against E.coli, Klebsiela, and Enterobacter, but Tazobactem did worse than imipenem against imipenem-resistant Acinetobacter (p = 0.032). Treatment outcome in 9 cases (50%) was death. No deaths after 28 days of follow-up were observed.
Conclusion: It seems that, gram-negative pathogens are the main cause of morbidity and mortality in ICU admitted ventilator induced infection. Imipenem resistant Acinetobacter is among the most prevalent and difficult to treat pathogens in ICU, and needs special consideration for initial therapy.
Key words: Pneumonia, mechanical ventilation, Acinetobacter, ICU, Imipenem
CITATION: Hashemian SMR, Tabarsi P, Mohammad MM et al. A prospective study of antibiotic susceptibility in patients with ventilator associated pneumonia. Anaesth Pain & Intensive Care 2012;16(1):31-37
INTRODUCTION
Intensive care unit (ICU) patients are susceptible to gram-negative bacterial infections due to several predisposing factors. Many of these bacterial agents are resistant to regular anti-bacterial therapies. Essentially, each ICU have an established protocol in place for initial empirical therapy based on previously accepted guidelines modified by local knowledge of prevalence of resistant serotypes unique to that ICU[1]. Notably, empiric therapy should be both appropriate by using more specific and adequate antibiotics through using correct dose and good penetration to the site of infection. Furthermore, the amount of wide-spectrum antibiotics used in ICUs is enormous particularly in combination therapy. Besides, resistance is on the rise among different bacterial agents in ICU which increases morbidity and mortality of patients[2]. Imipenem resistant Pseudomonas and Acinetobacters are among the most prevalent bacterial agents with high mortality in ICU patients[3],[4].
On the other hand gram negative bacteria contribute to a large fraction of ICU nosocomial infection mostly contributing to pneumonia and ventilator associated pneumonia (VAP)[5].VAP is the most frequent ICU acquired infection, and its etiology is almost the same among different population due to quick transmission of resistant agents between different ICU[6]. Increased mortality rate in VAP patients is associated with bacteremia, especially with Pseudomonas aeruginosa or Acinetobacter species, medical rather than surgical illness, and ineffective antibiotic therapy[7]. Multidrug resistant Acinetobacters and pseudomonas are isolated in ICU VAP infections all over the world[8],[9],[10] due to higher than usual ability of bacteria to develop various mechanisms to acquire resistance to main antibiotics used in ICU[11], especially in VAP patients[12],[13]. Most gram negative infections occur in immune compromised individuals and in critically ill patients often due to limited treatment with antibiotics[14],[15],[16]. However, whether colonization or infection with gram negative pathogens independent of other factors could genuinely increase the risk of mortality and morbidity in critical care units is still controversial[17] ,[18],[19],[20].
This controversy has sparked off a considerable debate among clinical practitioners and researchers about different factors associated with mortality of patients with gram negatives induced VAP in ICU. Antibiotic resistance is definitely one of the major determinants among different factors particularly when it comes to the gram negative induced VAP in ICU. Here in this prospective study we aimed to evaluate antibiotic susceptibility of isolated gram-negatives from patients with ventilator-associated pneumonia (VAP) in ICU.
METHODOLOGY
The study was reviewed and approved by the hospital ethics committee and performed in accordance with the ethical standards laid down in an appropriate version of the 2000 Declaration of Helsinki (http://www.wma.net/e/policy/b3.htm). All patients gave their informed consents prior to their inclusion in the study according to University Hospital Ethical Committee.
In this descriptive, cohort study, all patients admitted to ICU of Masih Daneshvari Hospital, since April 2009 till June 2010, with ventilator-associated pneumonia (VAP) were evaluated. Inclusion criteria were VAP in patients who were under mechanical ventilation in ICU for more than a week. Then a bacteriological study was performed on sample of BAL, tracheal washing, blood and urine samples. Clinical, laboratory and demographic information was collected. Demographic information, including age, sex, and clinical information including hypertension, chronic renal disease, diabetes, malignancy, hepatitis, heart failure, obstructive or restrictive lung disease, presence of intravenous or urine catheters were collected by using medical records. Exclusion criteria were concurrent source of infection in inserted catheters other than VAP.
One year recent history of surgery, history of hospitalization in the past year and prior antibiotic therapy before mechanical ventilation was evaluated. First, all samples were cultured in Macconkey blood agar growth media. Blood samples were cultured in trypticase soy broth (TSB) and chocolate agar (CA). Also, apart from urine samples, all samples were cultured in chocolate agar (CA). Pus was also incubated in thioglycolate for growth augmentation, cultured on plates as above at twenty-four, forty-eight hours and at the end of incubation. Chocolate agar plates were incubated in CO2 environment for twenty-four hours. Microorganisms were identified from growth plate by colony morphology (shape, color, size) and gram stain; Gram-negative bacteria grew on selective MacConkey’s agar plate, and then identified as lactose fermenter or non-fermenter. Gram staining, morphologic assessment, negative oxidase and other biochemical reactions were performed for all suspected colonies accordingly18.
Disc antibiogram was performed for the desired samples. All disks were standard according to Clinical Laboratory and Standards Institute (CLSI). The results are reported as minimum inhibitory concentration (MIC) according to antibiotic potency of disks which was standardized by Escherichia coli ATCC 25922, Staphylococcus aureus ATCC 25923 and Pseudomonas aeruginosa ATCC 27853.
Severity of the disease was determined based on Apache II scoring and to determine the involvement of pneumonia in ventilator-dependent patients CPIS form was used. Patients treated with usual protocol implementation in ICU, who had not responded well and were resistant to antibiotics in terms of qualified laboratory microbes, have been reported in this study too and duration of stay in ICU, mortality of patients in ICU, way of antibiotic therapy, subsequent review cultures and possible resistance to later antibiotics and accompanying other diseases were monitored.
Statistical analysis using SPSS version 16 was performed. To compare bivariate value, square test k(chi-square) and fisher exact test were used and for continuous variables such as age, weight and ICU stay ,first Kolmogorov-Smirnov continuity test and then T student test or square K ² were performed. P-Value less than 0/05 (P <0.05) was considered significant. The differences in susceptibility among gram-negative pathogens were calculated using 95% confidence intervals and P-value (p-value to be < 0.05). P-value was calculated based on Imipenem as a reference for other selected antimicrobials. Sample size was calculated by a statistical software based on variance extracted from previous similar.
RESULTS
In this study, 15 patients were entered the study from intensive care unit (ICU) admitted patients. The mean age was 50.5 (Range 20-96) years and six patients were male (42.9%). Median hospital stay was 53 days (Range 15-135 days). The underlying diseases in 12 patients (85.7%) were observed listed in Table 1. Four patients suffered from complications during hospitalization, and three cases from acute renal failure. One patient developed cardiac tamponade after heart surgery. Despite intensive care, 9 patients (50%) died before discharge from ICU. No deaths after 28 days of follow-up were observed (Table 2). We used different antibiotic therapy in our ICU patients. Median days of gram-negative bacteria isolation were 18 days, (range 5 to 30 days). Median hospitalization before admission to ICU was 2.5 day (Range 0-27 days). The length of ICU stay was not statistically correlated with mortality (P=0.45), but colonization of gram-negatives bacteria was statistically higher in longer ICU stay (P=0.022).
The median hospitalization in intensive care unit (ICU) was 36.5 days and the median stay before admission to ICU was 2.5 days that one patient were referred from another center (Table2). Six patients (42.9%) were admitted to ICU after surgery; four patients (28.6%) for elective surgery and 2 patients (14.3%) for open heart surgery. Eight patients (57.1%) were admitted due to another medical problem like pneumonia, heart disease or malignancy. Four patients had been diagnosed with lung cancer. Ten patients (71.4%) underwent mechanical ventilation promptly as they were admitted to ICU and 2 patients (14.3%) had CPR on admission. Also, 2 patients had a history of hospitalization in ICU in the last 6 months.
We have reported concomitant viral and bacterial infection in Table 2. Two patients had history of hospitalization in the ICU in the last 6 months; six patients had been admitted after urgent or elective surgical operation. APACHE II Score was from 4 to 20 score points in different patients, and the scores point of 18-20 was accompanied with death rate about 65% (Table 2).
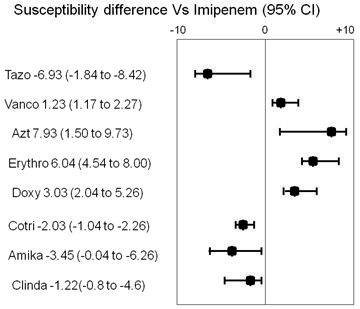
In our study, Acinetobacter dominated the isolates (in 58% of cases), thus, being frequent candidate pathogens causing VAP in ICUs. However our results show that Enterobacteriaceae are less than what physicians use to diagnose and treat in VAP in ICU. Imipenem showed superior activity over other tested antimicrobial classes against E. coli, Klebsiela pneumonia and Enterobacter (Table3). Imipenem did better than Amikacin against Acinetobacter species; However Imipenem has 11.2% resistance rates among the 34 tested gram-negative bacilli. Imipenem did better than other antibacterials against E.coli, Klebsiela, and Enterobacter, but Tazobactem did worse than Imipenems against Imipenem-resistant Acinetobacter (p = 0.032) against gram-negative pathogens all together (Figure 1). Other empiric antimicrobials like Amikacin -3.45 (CI:-0.04 to -6.26) show lower susceptibility pattern. Isolation date, antibiogram, DST, and outcome of the patients with VAP infection in ICU is listed in Table2.
DISCUSSION
Gram negative bacteria are nosocomial pathogenes which colonize on ICU people healthy looking skin and are able to colonize in instruments, catheters, and environments that can be even transmitted through the air[21]. They will be easily transferred through medical devices commonly used for monitoring in intensive care unit (ICU) as well as by health care personnel[22],[23]. Some risk factors that were recognized in association with gram negative infections include older age 21, immune suppression[24], surgery[25] and prior treatment with broad spectrum antibiotics[26]. In our study, there were no significant differences in age, underlying disease, and final diagnosis of our patients. However, four patients of our study with lung cancer seemed to have diminished immune response, considering that these patients had received immunosuppressive drugs.
Acinetobacter was the dominant pathogen isolated from VAP patients in our study. Although this may be in contrary to other studies, but considering that Enterobacteriaceae are mostly isolated from urinary sources of infections, our results are not far from others. Acinetobacter indeed has a wonderful ability to grow in various inserted catheters in patients in ICU particularly endotracheal tube[27]. Josef et al showed that Enterobacteriaceae, Haemophilus influenzae, Staphylococcus aureus, Streptococcus pneumoniae, and Candida were more common in early-onset VAP, while non-fermenters (Pseudomonas and Acinetobacter) were significantly associated with late-onset VAP[28]. However, in our study we showed that Acinetobacter could truly be a major determinant of VAP mortality in patients under ventilator. This is not in contrast to previous reports that actually mentioned the ability of Acinetobacter to colonize inside ventilators in ICU[29].
Our results showed that Imipenem is not a trustworthy antibiotic in initial empiric therapy in VAP patients. In fact, the rate of Imipenem-resistant Acinetobacter is the highest among tested agents in ICU infection leading to high mortality rates. Moreover, Tazobactem showed tendency towards less activity than Imipenem when gram-negative pathogens were isolated together. However, considering all gram-negative pathogens together, the same as what actually happens in the clinical approach for initial empiric therapy, Imipenem is the only antimicrobial that have a reliable activity against all gram-negative except for Imipenem-resistant Acinetobacter.
In our study, eight patients were transferred from a general ward or another hospital that had enough time required for colonization and a history of extensive spectrum antibiotics commonly used in hospitals. In addition, surgery was a risk factor in our study so that six patients developed VAP infection after post-operative admission to ICU. This issue shows importance of infection control in the pre-operation, operating room and post-operation sections. As a matter of fact, gram-negative pathogens have been remained largely susceptible to disinfectants and antiseptics, and occasional reports of disinfectant failure are more likely to represent the failure of personnel to follow cleaning procedures than disinfectant resistance[30]. Besides, length of stay in ICU is also an important factor in development of VAP with gram-negative pathogens which showed a direct correlation to colonization of gram-negative bacteria, however, it was not statistically correlated with mortality of VAP patient. On the other hand, increase in APACHE II score was found to be risk factor for death predicts in VAP (Table3). Our study supports the idea which mortality was higher based on higher APACHE II score. In fact APACHE II score could be an independent predictor of mortality rate in VAP patients. Conceivably, several underlying diseases related to higher APACHE II score could expose patient to the risk of VAP25.
In conclusion, it seems that VAP infection with multi drug resistant gram negative bacteria is a clinical dilemma for intensivists around the world. As bacterial resistance exceeds the available antibiotic options for these patients, hospital infection control would have a determining role in preventing gram negative induced infections in ICU. It seems that empirical therapy should be considered with regard to patient conditions, besides to continuous monitoring of microbial pathogens and bacterial resistance. For future studies, we consider, polymerase chain reaction (PCR) to determine species of bacteria and drug resistance.
Conflict of Interest
The authors of this article declare no conflict of interest, no financial, consulting, and personal relationships with any other people or organizations that could influence (bias) the authors’ work.
Table 1. Demographic data and final diagnosis of the patients with infection in ICU.
| Patient | Gender | Age | Underlying Disease | Diagnosis |
| 1 | F | 57 | IHD | CABG |
| 2 | F | 55 | COPD | Sleep apnea |
| 3 | M | 47 | Cirrhosis | Esophageal perforation |
| 4 | F | 48 | Adenocarcinoma | Lobectomy |
| 5 | M | 54 | IHD | CABG |
| 6 | F | 76 | IHD/old TB | Pneumonia |
| 7 | M | 50 | IPF/CF | IPF |
| 8 | M | 95 | CHF/lung adenocarcinoma | Pneumonia |
| 9 | F | 35 | TB | Stevens Johnson syndrome |
| 10 | F | 20 | Ataxia telangectasia | Pneumonia |
| 11 | F | 36 | Lung cancer | Apnea |
| 12 | M | 21 | Trauma | Pneumonia |
| 13 | M | 63 | Pericardial effusion | Pericardial effusion |
| 14 | F | 49 | Bronchiectasis/ TB | Pulmonary emboli |
| 15 | M | 35 | COPD | Apnea |
| * IHD: ischemic heart disease, CHF: congestive heart failure, TB: Tuberculosis | ||||
Table 2. ICU length of stay, complication, death predict and outcome of the patients with infection in ICU.
| Patient | ICU length
of Stay |
Bacterial Infection | Viral Infection | APACHE II Score | Death
Predict |
Outcome |
| 1 | 58 | PA, Acin, Candida | No | 18 | 0.29 | Expired |
| 2 | 135 | PA, Kleb | No | 13 | 0.17 | Expired |
| 3 | 45 | Enterobacter | HSV1/CMV | 20 | 0.36 | Expired |
| 4 | 76 | Kleb, E.coli | No | 6 | 0.07 | Uneventful |
| 5 | 42 | Enterobacter | No | 12 | 0.15 | Uneventful |
| 6 | 15 | Citrobacter | No | 13 | 0.17 | Expired |
| 7 | 30 | PA, Citrobacter | HSV1 | 17 | 0.26 | Expired |
| 8 | 19 | Acin,Ecoli | No | 20 | 0.36 | Expired |
| 9 | 106 | Kleb, Acin, PA | No | 8 | 0.09 | Expired |
| 10 | 31 | Acin, Enterobacter | HSV1/EBV | 10 | 0.11 | Expired |
| 11 | 4 | Acinetobacter | No | 4 | 0.05 | Uneventful |
| 12 | 23 | Acinetobacter | HSV1/CMV | 12 | 0.15 | Uneventful |
| 13 | 35 | Acinetobacter | No | 10 | 0.11 | Expired |
| 14 | 45 | Acinetobacter | HSV1/EBV | 18 | 0.29 | Uneventful |
| 15 | 25 | Acinetobacter | CMV | 12 | 0.12 | Expired |
| *Acin: Acinetobacter; PA: Pseudomonas Aeroginosa; Kleb: Klebsiela; | ||||||
Table 3. Antimicrobial Susceptibility of all gram-negative pathogens isolates in ICU patients.
| Antibiotic(S/R)* | Imi | Tazo | Vanco | Azt | Erythro | Doxy | Cotri | Amika | Clinda |
| Psudomonas | 12/10 | 18/2 | 17/8 | 22/4 | 9/6 | 17/3 | 18/4 | 23/5 | 0/3 |
| Acinetobacter | 2/22 | 2/25 | 24/1 | 26/2 | 6/0 | 4/0 | 2/23 | 0/11 | 17/11 |
| E.coli | 34/1 | 17/22 | 4/11 | 7/8 | 4/6 | 4/9 | 8/10 | 54/2 | 6/7 |
| Klebsiella | 27/2 | 17/11 | 12/13 | 6/0 | 15/26 | 1/4 | 0/11 | 19/4 | 1/24 |
| Entrobacter | 14/4 | 11/3 | 19/2 | 24/1 | 17/12 | 18/10 | 9/4 | 0/6 | 10/12 |
| Citrobacter | 11/22 | 5/6 | 15/3 | 3/0 | 2/12 | 6/3 | 5/1 | 4/0 | 8/17 |
| *S: Susceptible; R: Resistant
**Vanco: Vancomycine; Azt: Azithromycine; Doxy: Doxycycline; Cotri: Co-trimoxazole; Erithro: Erythromycine; Imi: Imipenem; Tazo: Tazobactam; Clinda: Clindamycine;Amika: Amikacin |
|||||||||
Figure 1. The Percent difference of each antimicrobial agent vs. Imipenem and 95% Confidence interval, comparing Imipenem activity against all gram-negative pathogenes in ICU. Vanco: Vancomycine; Azt: Azithromycine; Doxy: Doxycycline; Cotri: Co-trimoxazole; Erithro: Erythromycine; Imi: Imipenem; Tazo: Tazobactam; Clinda: Clindamycine;Amika: Amikacin.
REFERENCES
- Raineri E, Crema L, Dal Zoppo S, Acquarolo A, Pan A, Carnevale G, Albertario F, Candiani A.Rotation of antimicrobial therapy in the intensive care unit: impact on incidence of ventilator-associated pneumonia caused by antibiotic-resistant Gram-negative bacteria. Eur J Clin Microbiol Infect Dis 2010;29(8):1015-24.
- Chastre J WM, Wolff M, Fagon JY. Comparison of 8 vs 15 days of antibiotic therapy for ventilator associated pneumonia in adults: a randomized trial. JAMA 2003;290:2588-98.
- Jamaati HR, Malekmohammad M, Hashemian SM, Nayebi M, Basharzad N. Ventilator-Associated Pneumonia: Evaluation of Etiology, Microbiology and Resistance Patterns in a Tertiary Respiratory Center. Tanaffos 2010;9(1):21-27.
- Vidal F, Mensa J, Almela M, Olona M, Martinez JA, Marco F, Lopez MJ, Soriano A, Horcajada JP, Gatell JM. Bacteraemia in adults due to glucose non-fermentative Gram-negative bacilli other than P. aeruginosa. QJM 2003;96:227-234.
- Kunis KA, Puntillo KA. Ventilator-associated pneumonia in the ICU: its pathophysiology, risk factors, and prevention. Am J Nurs 2003;133(8):64AA-64GG.
- Blot S, Lisboa T, Angles R, Rello J. Prevention of VAP: is zero rate possible? Clin Chest Med 2011;32(3):591-9.
- Timsit JF, Zahar JR, Chevret S.Attributable mortality of ventilator-associated pneumonia.Curr Opin Crit Care 2011;17(5):464-71.
- Fridkin SK. Increasing prevalence of antimicrobial resistance in intensive care units. Crit Care Med 2001; 29 (Suppl 4):N64-8.
- Karlowsky JA, Draghi DC, Jones ME, Thornsberry C, Friedland IR, Sahm DF. Surveillance for antimicrobial susceptibility among clinical isolates of Pseudomonas aeruginosa and Acinetobacter baumannii from hospitalized patients in the United States, 1998 to 2001.Antimicrob Agents Chemother 2003;47:1681-8.
- Dijkshoorn L, Nemec A, Seifert H. An increasing threat in hospitals: multidrug resistant Acinetobacter baumannii. Nat Rev Microbiol. 2007;5(12):939-51.
- Chastre J. Infections due to Acinetobacter baumannii in the ICU. Semin Resp Crit Care Med 2003;24:69-78.
- Tankovic J, Legrand P, De Gatines G, Chemineau V, Brun-Buisson C, Duval J. Characterization of a hospital outbreak of imipenem-resistant Acinetobacter baumannii by phenotypic and genotypic typing methods. J Clin Microbiol 1994;32:2677-81.
- American Thoracic Society; Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare associated pneumonia. Am J Respir Crit Care Med 2005;171(4):388- 416.
- Urban C, Segal-Maurer S, Rahal J. Considerations in control and treatment of nosocomial infections due to multidrug-resistant Acinetobacter baumannii. Clin Infect Dis 2003;36:1268-1274.
- Fournier PE, Richet H. The epidemiology and control of Acinetobacter baumannii in health care facilities. Clin Infect Dis 2006;42:692-9.
- Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 2004;39:309-317.
- Boots RJ, Lipman J, Bellomo R, Stephens D, Heller RF. Disease risk and mortality prediction in intensive care patients with pneumonia. Australian and New Zealand practice in intensive care (ANZPIC II). Anaesth Intensive Care 2005;33:101-111.
- Niederman MS. Impact of antibiotic resistance on clinical outcomes and the cost of care. Crit Care Med 2001;29:114-120.
- Michalopoulos A, Falagas ME. Treatment of Acinetobacter infections. Expert Opin Pharmacother 2010;11(5):779-88.
- Garnacho J, Sole-Violan J, Sa-Borges M, Diaz E, Rello J. Clinical impact of pneumonia caused by Acinetobacter baumannii in intubated patients: a matched cohort study. Crit Care Med 2003;31:2478-2482.
- Kuti JL, Shore E, Palter M, Nicolau DP. Tackling empirical antibiotic therapy for ventilator-associated pneumonia in your ICU: guidance for implementing the guidelines. Semin Respir Crit Care Med 2009;30(1):102-15.
- Ak O, Batirel A, Ozer S, Çolakoğlu S.Nosocomial infections and risk factors in the intensive care unit of a teaching and research hospital: a prospective cohort study. Med Sci Monit 2011;17(5):PH29-34.
- Wahl WL, Zalewski C, Hemmila MR.Pneumonia in the surgical intensive care unit: is every one preventable? Surgery 2011;150(4):665-72.
- Mahgoub S, Ahmed J, Glatt AE. Underlying characteristics of patients harbouring highly resistant Acinetobacter baumannii. Am J Infect Control 2002;30:386-90.
- American Thoracic Society & Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 2005;171:388-416.
- Garnacho-Montero J, Ortiz-Leyba C, Fernandez-Hinojosa E. Acinetobacter baumannii ventilator-associated pneumonia: epidemiological and clinical findings. Intensive Care Med 2005;31:649-55.
- Coppadoro A, Berra L, Bigatello LM. Modifying endotracheal tubes to prevent ventilator-associated pneumonia. Curr Opin Infect Dis 2011;24(2):157-62.
- Joseph NM, Sistla S, Dutta TK, Badhe AS, Rasitha D, Parija SC. Ventilator-associated pneumonia in a tertiary care hospital in India: role of multi-drug resistant pathogens. J Infect Dev Ctries 2010;4(4):218-25.
- Villegas MV, Hartstein AI. Acinetobacter outbreaks, 1977-2000. Infect Control Hosp Epidemiol 2003;24: 284-90.
- Hansen S, Stamm-Balderjahn S, Zuschneid I. Closure of medical departments during nosocomial outbreaks: data from a systematic analysis of the literature. J Hosp Infect 2007;65:348-353.