Manpreet1, Sarvjeet Kaur, MD2, Naresh Baghla, MD3, Hiteshi Aggarwal1
1Post-graduate student; 2Associate Professor; 3Professor
Department of Anesthesiology, Guru Gobind Singh Medical College and Hospital, Faridkot, Punjab, (India)
Correspondence: Dr. Sarvjeet Kaur, C/o Dr. S.S. Ratta, Sikhanwala Road, Kotkapura, Faridkot, Punjab, (India); E-mail: drsarvjeetk@gmail.com
ABSTRACT
Background and Aims: Regional anesthesia offers benefits to patients and anesthetists by keeping the patients awake, preserving the airway reflexes, by providing cardiovascular stability during the procedure and fast postoperative recovery. Patients are often uncomfortable, because of pain at puncture site, recall of procedure and limited duration of blockade. Alpha-2 adrenoreceptor agonists were introduced in anesthesia for their sedative and analgesic effects. The aim of this study was to compare the effects of intravenous dexmedetomidine or clonidine as adjuvants during bupivacaine spinal anesthesia.
Methodology: A prospective, randomized study was conducted involving 120 patients scheduled for elective infraumbilical surgery under spinal anesthesia. Patients were randomly divided into three groups (n = 40) and were given the following drugs intravenously as per group allocation: Group A received 1 μg/kg of dexmedetomidine, followed by an infusion at the rate of 0.5 µg/kg/h; Group B received 1 μg/kg of clonidine, followed by an infusion at rate of 1 µg/kg/h and Group C received normal saline bolus and infusion. Loading dose was given over 10 min, prior to (SAB), followed by a maintenance infusion. Ramsay sedation score of 3-4 was considered as target sedation. Patients were assessed for time required to achieve target sedation, prolongation of analgesia and motor blockade. The hemodynamic parameters and side effects were also observed.
Results: The target sedation was achieved significantly earlier in Group A (14.32 ± 5.25 min) as compared to Group B (30.01 ± 2.33 min) (P = 0.001). In Group A, the mean duration of analgesia was 208.25 ± 28.29 min as compared to 169.75 ± 20.15 min in Group B and 135.25 ± 22.60 min in Group C (P < 0.05). Duration of motor blockade was increased in Group A (217 ± 24.697 min) as compared to Group B and C.
Conclusion: Intravenous dexmedetomidine infusion is better than intravenous clonidine as it provides earlier onset of adequate sedation along with prolongation of analgesia and motor blockade during bupivacaine spinal anesthesia.
Key words: Intravenous; Dexmedetomidine; Clonidine; Bupivacaine; Spinal anesthesia
| Citation: Manpreet, Kaur S, Baghla N, Aggarwal H. Is dexmedetomidine a better sedative agent than clonidine in spinal anesthesia? Anaesth Pain & Intensive Care 2016;20(4):451-456 |
Received: 24 Jul 2016; Reviewed: 8 Aug, 9 Sep 2016; Corrected: 29 Sep 2016; Accepted: 5 Jan 2017
INTRODUCTION
Intra-operative sedation is important for surgery under spinal anesthesia. Adequate sedation relieves anxiety and allows the patient to lie down in the same position for long duration.1 Clonidine and dexmedetomidine are α2 agonists which provide adequate sedation as well as analgesia. Commonly used sedative agents have drawbacks2 and development of newer α2 agonists has improved the quality of sedation. They do not depress respiration, patients are easily arousable and able to communicate regarding their pain.3,4
We designed a clinical study to evaluate and compare the effect of infusion of dexmedetomidine with clonidine during spinal anesthesia for elective infraumbilical surgeries. Our primary objectives were to compare time to achieve target sedation (RSS 3-4) and postoperative analgesia. Secondary comparison end points were motor blockade, hemodynamics parameters and adverse effects.
METHODOLOGY
The present study was conducted between November 2014 to May 2015, after obtaining permission of institutional Ethical Committee and written informed consent of patients. A total of 120 patients were enrolled for this prospective, randomized controlled, double-blinded study.
Male/female patients aged between 30 and 55, ASA I and II physical status, scheduled for elective lower abdominal and lower limb surgery under SAB, were enrolled.
Obese patients (BMI ˃ 30 kg/m2), patients with a known allergy to study drugs, on chronic analgesic medication, with significant cardiovascular, renal, or hepatic dysfunction, neurological or psychiatric disease or on concurrent medication or pregnant patients were excluded.
A computer generated randomization list was created and randomization assignment was kept in sealed opaque envelops. These envelopes were opened at the time of study drug preparation. Blinding was achieved through the preparation of equal amount of clear drug solutions in normal saline for both bolus and maintenance doses. They were labeled as study drugs by an anesthesiologist not involved in the study. The study drug solutions were handed over to the attending anesthesiologist who performed the procedure of SAB, but was unaware of group allocation. Patients were allocated into three groups of 40 patients each as follows;
Group A (n = 40): Intravenous dexmedetomidine 1 µg/kg diluted to 20 ml with normal saline and infused over 10 min as a loading dose prior to SAB, followed by a maintenance dose of 0.5 µg/kg/h.
Group B (n = 40): Intravenous clonidine 1 µg/kg diluted to 20 ml with normal saline and infused over 10 min as a loading dose, prior to SAB, followed by a maintenance dose of 1 µg/kg/h.
.Group C (n = 40): Intravenous, 20 ml normal saline over 10 min as a loading dose, prior to SAB, followed by maintenance infusion.
Preoperatively the patients were explained about the procedure and visual analogue scale (VAS) during the preanesthetic visit. Two hours before surgery, tablet ranitidine 150 mg and alprazolam 0.25 mg were given orally to all patients. On arrival in the operation theatre, standard monitors like electrocardiography (ECG), non‑invasive blood pressure (NIBP) and pulse oximetry (SpO2) were attached and the baseline parameters were recorded. After securing an 18G venous cannula, all patients received 500 ml of normal saline solution.
The 20 ml solution of drug as per study group (loading dose: dexmedetomidine or clonidine 1 µg/kg) was infused for the first 10 minutes. Following the loading dose, SAB was performed under all aseptic precautions in sitting position at L3‑L4 level through a standard midline approach using a 25 G Quincke spinal needle and 15 mg of 0.5% heavy bupivacaine was injected after aspiration of clear cerebrospinal fluid. After SAB, patient was repositioned supine and infusion of maintenance dose was initiated as appropriate for the study group with help of an infusion pump. After sensory block reached level of T8, surgeons were allowed to start the surgery.
Bilateral sterile pin-prick method was used to check the sensory level while a modified Bromage scale5 (grade 0: No paralysis; 1: Unable to raise extended leg; 2: Unable to flex knee; 3: Unable to flex ankle) was used to assess the motor blockade. Sensory and motor block was assessed at every minute for the first 10 min and thereafter every 30 min upto 6 h postoperatively.
Intravenous fluids in the form of Ringer lactate were administered as per body weight and operative losses during surgery. Infusion of study drug was stopped approximately five minutes before the end of surgery. After surgery, the patients were shifted to post anesthesia care unit (PACU). In PACU, pain score was noted by an anesthesiologist who was blinded to group allocation at every 30 min for 6 hours by using the visual analogue scale of 0-10; 0 being no pain, 1-3 mild, 4-6 moderate, and 7 or more as severe pain.6 Patients with VAS score of more than 3 received intravenous diclofenac 75 mg as rescue analgesic.
The following observations were recorded by the attending anesthesiologist;
- Heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP) and oxygen saturation were recorded every 10 minutes till the end of surgery.
- Onset of target sedation which was, from administration of sedative (loading dose) to achievement of RSS of 3-4. Sedation was also evaluated intraoperatively every 10 min from starting of loading dose till the end of surgery using Ramsay Sedation Score7:
- Onset times of both sensory and motor blockade, the highest dermatomal level of sensory blockade and duration of both sensory and motor blockade were recorded. Onset of sensory block was defined as the time to achieve T8 level of sensory block. Sensory block duration was the time to two dermatome regression from the maximum sensory block level. Time for motor block onset was assumed when modified Bromage score became three. Motor block duration was the time to return to grade-0 on the modified Bromage scale.
- Perioperative side effects such as, nausea and vomiting, hypotension, bradycardia, shivering, respiratory depression.
- Duration of analgesia was recorded as the time from intrathecal injection to the time of first request for rescue analgesia after surgery.
Statistical analysis: The data were analyzed by SPSS (Statistical Package for Social Sciences) 11.5 software. A power analysis was performed to determine the necessary number of patients for each group based on duration of analgesia. With α = 0.05 and study power at 80%, it was estimated that 30 patients would be needed in each group in order to detect a 35% difference in the duration of analgesia between the study groups. The inclusion of 40 patients in each group was done for better validation of results.
Parametric testing was done by using ANOVA (one way analysis of variance) test and categorical variables were compared by Chi-square test. Value of P < 0.05 was considered significant and P < 0.0001 as highly significant.
RESULTS
The demographic data and duration of surgery were comparable between the three groups. (Table 1) In all the three groups, changes in the parameters (MAP & HR) were within clinically acceptable range of i.e. 20% from the baseline, indicating the hemodynamic stability in dexmedetomidine and clonidine groups at given doses (Figure 1 & 2). There were no differences in respiratory end points between all three study groups.
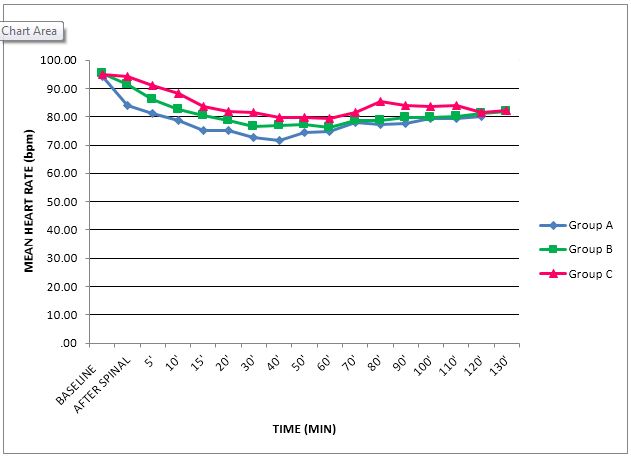
Figure 1: Trends in intraoperative heart rate
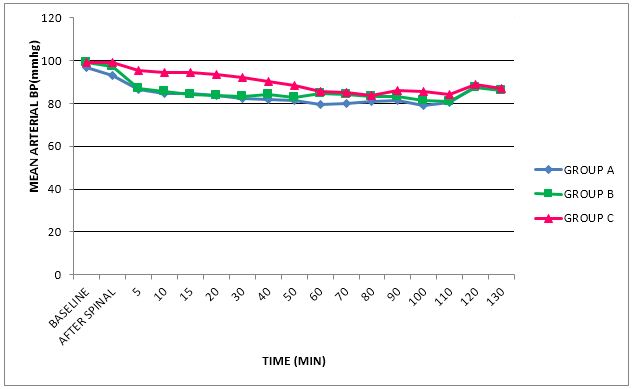
Figure 2: Trends in intraoperative mean arterial pressure
There was a significant difference in the time required to achieve targeted level of sedation. The time to achieve targeted sedation (RSS 3-4) was significantly shorter in Group A when compared to Group B (14.32 ± 5.25 min vs 30.01 ± 2.33 min) (p = 0.001), while no patient achieved RSS 3-4 in Group C (Figure 3).
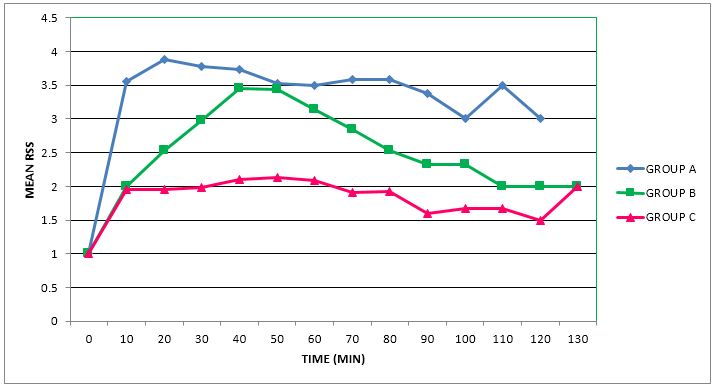
Figure 3: Ramsay Sedation Score in three study groups
Patients with Ramsay Sedation Score more than three were 61% in dexmedetomidine group and 28% in clonidine group. Mean sedation score was found to be significantly higher in Group A (p < 0.05) (Table 3).
Table 1: Demographic distribution
| Variables | Group A (n = 40) | Group B (n = 40) | Group C (n = 40) | p-value |
| Age (year) | 39.83 ± 14.225 | 37.85 ± 10.935 | 37.38 ± 12.646 | 0.664 |
| Sex ( M:F) | 33:7 | 29:11 | 31:9 | NA |
| Weight (Kg) | 68.90 ± 9.803 | 69.26 ± 11.121 | 67.00 ± 12.690 | 0.647 |
| ASA (I:II) | 36:4 | 36:4 | 34:6 | NA |
| Duration of surgery (min) | 73.00 ± 15.392 | 67.75 ± 16.562 | 71.00 ± 17.365 | 0.331 |
Values are mean ± SD, and numbers; ASA = American Society of Anesthesiologists, n-Number of patients
Table 2: Comparison of mean duration of analgesia, complete recovery and rescue analgesic requirement
| Variables | Group A
(n = 40) |
Group B
(n = 40) |
Group C
(n = 40) |
p-value |
| Highest sensory level (segment) | T4-T5 | T6-T7 | T7-T8 | 0.01 |
| Complete recovery (min) | 217 ± 24.69 | 180.25 ± 16.44 | 150.50 ± 19.27 | 0.0001 |
| Duration of analgesia (min) | 208.25 ± 28.29 | 169.75 ± 20.15 | 135.25 ± 22.60 | 0.0001 |
| Rescue analgesics (number) | 9 | 11 | 14 | NA |
Values are mean ± SD, and numbers
Table 3: Adverse events
| Variables | Group A
(n = 40) |
Group B
(n = 40) |
Group C
(n = 40) |
| Hypotension | 7 (17.5%) | 5 (12.5%) | 4 (10%) |
| Bradycardia | 4 (10%) | 2 (5%) | 0 |
| Sedation score(>3) | (61%) | (30%) | 0 |
| Shivering | 1(2.5%) | 0 | 6 (15%) |
Values are numbers and percentage
During bupivacaine spinal anesthesia, intravenous dexmedetomidine also resulted in achieving an earlier and maximum sensory block level compared to clonidine and placebo (Table 2).
The mean duration of analgesia in Groups A, B and C was 208.25 ± 28.29 min, 169.75 ± 20.15 and 135.25 ± 22.60 min, respectively which was significantly longer in Group A (p = 0.0001) (Table 2).
The mean duration for complete motor recovery was maximum in Group A (217 ± 24.697 min) followed by Group B (180.25 ± 16.448 min) and was least in Group C (150.50 ± 19.279 min) and this was statistically significant. (p = 0.0001) (Table 2) Nine patients needed rescue analgesia in Group A, 11 in Group B and 14 in Group C (Table 2).
DISCUSSION
Our results showed that onset of sedation was achieved earlier in patients receiving intra-operative dexmedetomidine infusion during infraumbilical surgery under spinal anesthesia and the difference in the findings was seen to be significant (p < 0.001).
Based on the observation of a previous study,8 a bolus dose of 1 µg/kg/h was selected by us. Various studies have established that the dose of clonidine is 1.5-2 times higher than the dose of dexmedetomidine.9,10 Thus, we selected an infusion dose of 0.5 µg/kg/h for dexmedetomidine and 1 µg/kg/h for clonidine.
The continuous infusion used in our study produced easy controllability over depth of sedation and less side effects.11 The percentage of patients achieved target sedation score (RSS 3-4) was more (61%) in dexmedetomidine group compared to (28%) in clonidine group (p < 0.05). None of the patients in control group achieved target sedation score. Our results are similar to previous study12 which demonstrated intravenous dexmedetomidine to be an effective agent in term of onset of target sedation during various surgical procedures. Distribution half-life of dexmedetomidine is short (6 min) and is responsible for rapid titration of sedation in Group A. Secondly, this could be due to difference of α2/α1 receptors selectivity among the study drugs. Dexmedetomidine is highly specific α2 adrenoceptor agonist (α2/α1 = 1620/1) compared to clonidine (α2/α1 = 220/1).13 Tamman et al.,14 found that in pediatric patients undergoing MRI scan the onset of satisfactory sedation was shorter in intravenous dexmedetomidine group in comparison to intramuscular dexmedetomidine (7.93 ± 0.884 vs. 16.87 ± 4.49 min). In contrast Ragab A et al.15 found delayed onset of sedation in patients receiving intravenous dexmedetomidine as compared to intravenous midazolam for conscious sedation in rhinoplasty under local anesthesia. This discrepancy may be due to different score utilized for sedation assessment and only loading dose given to the patients in their study.
Mean sedation scores were significantly higher in the dexmedetomidine group (p < 0.05).We noted that the mean sedation scores during surgery were 3-5 in the dexmedetomidine group, 2-4 in the clonidine group and 2 in the control group (p < 0.0.05). In present study, dexmedetomidine provide good sedation without causing significant respiratory depression. This result indicates that higher level of sedation can be achieved with intraoperative dexmedetomidine infusion with minimal or no respiratory depression. This was also reported by other authors.16,17 In the present study, there was no significant difference in the SpO2 levels between the groups during surgery and in the postoperative period.
In our study, dexmedetomidine provided the earliest favorable motor condition for surgery
(Bromage motor score 3). It also prolonged the duration of motor blockade. These results are in accordance with two previous study by Dinesh et al.16 and Harsoor et al.18 Reddy et al.17 observed that intravenous dexmedetomidine did not prolonged the motor block duration. This might be due to difference in our study design consisting of continuous dexmedetomidine infusion throughout the surgery whereas only bolus dose was used in their study.
In comparing the time of the first dose of postoperative analgesic request between the three groups, there was statistically significant increase in the time to request of first dose of postoperative analgesia by patients in dexmedetomidine group as compared to clonidine and placebo group. Similarly, postoperative diclofenac requirements during first 6 h were significantly less in patients who received dexmedetomidine infusion. Reddy et al.14 also showed that time of first request for analgesic were significantly prolonged in the dexmedetomidine group than clonidine and placebo groups. The antinociceptive action of dexmedetomidine is mediated by activation of alpha 2-B agonist and inhibition of substance P release at the level of dorsal horn.19
Limitations: Our study has some limitations. First, we chose only ASA physical status I and II patients, so efficacy of the two drugs in high risk patients could not be assessed. Second, there is paucity of studies which have compared the equivalence dose of these two drugs.
Conflict of interest: None declared by the authors
Authors’ contribution: All authors took part in the study and manuscript preparation
CONCLUSION
Intravenous dexmedetomidine infusion, during bupivacaine spinal anesthesia, was found better than intravenous clonidine in order to provide earlier onset of adequate sedation and prolongation of analgesia.
REFERENCES
- Schnider TW, Minto CF. Predictors of onset and offset of drug effect.Eur J Anaesthesiol Suppl. 2001;23:26-31. [PubMed]
- Alhashemi JA. Dexmedetomidine vs midazolam for monitored anaesthesia care during cataract surgery. Br J Anaesth 2006;96:722‑ [PubMed] [Free full text]
- Pichot C, Ghignone M, Quintin L. Dexmedetomidine and clonidine: From second- to first-line sedative agents in the critical care setting? J Intensive Care Med 2012;27:219-37. [PubMed] doi: 10.1177/0885066610396815.
- Grewal A. Dexmedetomidine: New avenues. J Anaesthesiol Clin Pharmacol 2011;27:297‑ [PubMed] [Free full text] doi: 10.4103/0970-9185.83670.
- Korula S, George GM, Ipe S, Abraham SP. Epidural anesthesia and post-operative analgesia for bilateral inguinal mesh hernioplasty: Comparison of equipotent doses of ropivacaine and bupivacaine. Saudi J Anaesth 2011;5:277–81. [PubMed] [Free full text] doi: 10.4103/1658-354X.84101.
- Shahi V, Verma AK, Agarwal A, Singh CS. A comparative study of magnesium sulfate vs dexmedetomidine as an adjunct to epidural bupivacaine. J Anaesthesiol Clin Pharmacol 2014;30:538-42. [PubMed] [Free full text] doi: 10.4103/0970-9185.142852.
- Ramsay MA. Measuring level of sedation in the Intensive Care Unit. JAMA 2000;284:441. [PubMed]
- Hazra R, Manjunatha SM, Manuar MDB, Basu R, Chakraborty S. Comparison of the effects of intravenously administered dexmedetomidine with clonidine on hemodynamic responses during laparoscopic cholecystectomy. Anaesth Pain & Intensive Care 2014;18:25-30. [Free full text]
- Bajwa SJ, Bajwa SK, Kaur J, Singh G, Arora V, Gupta S, et al. Dexmedetomidine and clonidine in epidural anesthesia:A comparative evaluation. Indian J Anaesth 2011;55:116‑ [PubMed] [Free full text] doi: 10.4103/0019-5049.79883.
- Boku A, Hanamoto H, Oyamaguchi A, Inoue M, Morimoto Y, Niwa H.The clinical use of dexmedetomidine. Rev Bras Anestesiol 2016 Jan-Feb;66(1):37-43. [PubMed] [Free full text] doi: 10.1016/j.bjan.2015.11.001.
- Patki A, Shelgaonkar. A comparison of equisedative infusion of propofol and midazolam for conscious sedation during spinal anesthesia- A prospective randomized study. J Anaesthesiol Clin Pharmacol 2011;27:47–53. [PubMed] [Free full text]
- Srivastava U, Sarkar ME, Kumar A, Gupta A, Agarwal A, Singh TK, et al. Comparison of clonidine and dexmedetomidine for short-term sedation of intensive care unit patients.Indian J Crit Care Med 2014;18:431–6. [PubMed] [Free full text] doi: 10.4103/0972-5229.136071.
- Mukherjee A, Das A, Basunia SR, Chattopadhyay S, Kundu R, Bhattacharyya Emergence agitation prevention in paediatric ambulatory surgery: A comparison between intranasal dexmedetomidine and clonidine. J Res Pharm Pract 2015;4:24–30. [PubMed] [Free full text] doi: 10.4103/2279-042X.150051.
- Tammam Tarek F, Wahba Sherif S. Quality of MRI pediatric sedation: comparison between intramuscular and intravenous dexmedetomidine. Egyptian J Anaes 2013;29:47–52. [Free full text]
- Ragab A, Elshamaa H, Ibrahim M. Dexmedetomidine, morphine, propofol vs midazolam, morphine, propofol for conscious sedation in rhinoplasty under local anesthesia. A prospective, randomized study. Egyptian J Anaes 2013;29:181-7. [Free full text]
- Dinesh CN, Sai Tej NA, Yatish B, Pujari VS, Kumar RMM, Mohan CV. Effects of intravenous dexmedetomidine on hyperbaric bupivacaine spinal anesthesia: A randomized study. Saudi J Anaesth 2014 Apr;8(2):202-8.[PubMed] [Free full text] doi: 10.4103/1658-354X.130719.
- Reddy VS, Shaik NA, Donthu B, Sannala VK, Jangam V. Intravenous dexmedetomidine versus clonidine for prolongation of bupivacaine spinal anesthesia and analgesia: A randomized double-blind study. J Anaesthesiol Clin Pharmacol 2013;29:342-7. [PubMed] [Free full text] doi: 10.4103/0970-9185.117101.
- Harsoor S, Rani DD, Yalamuru B, Sudheesh K, Nethra S. Effect of supplementation of low dose intravenous dexmedetomidine on characteristics of spinal anaesthesia with hyperbaric bupivacaine. Indian J Anaesth 2013;57:265-9. [PubMed] [Free full text] doi: 10.4103/0019-5049.115616.
- Jooste EH, Muhly WT, Ibinson JW, Suresh T, Damian D, Phadke A, et al.Acute hemodynamic changes after rapid intravenous bolus dosing of dexmedetomidine in pediatric heart transplant patients undergoing routine cardiac catheterization. Anesth Analg 2010;111:1490-6. [PubMed] [Free full text] doi: 10.1213/ANE.0b013e3181f7e2ab.

