Arvinder Pal Singh, MD1, Sunil Chawla, MD2, Sukhminder Jit Singh Bajwa, MD, MBA, FACEE3, Menaal Khanna, MBBS4
1Associate Professor; 2Professor
Sri Guru Ram Das Institute of Medical Sciences & Research, Mehta Road, PO Vallah, Amritsar – 143501. Punjab, India
3Director Professor; 4Junior Resident,
Department of Anesthesiology and Intensive Care, Gian Sagar Medical College and Hospital, Banur, Patiala, Punjab, India.
Correspondence: Dr. Sukhminder Jit Singh Bajwa, House No. 27-A, Ratan Nagar, Tripuri, Patiala, Punjab, (India) PIN-147001; Ph: No-+919915025828, +911752352182; E-mail: sukhminder_bajwa2001@yahoo.com
ABSTRACT
Background and Objectives: Dexmedetomidine is increasingly being used in regional anesthesia as an adjuvant but there is no consensus on exact minimal and safe dose when used intrathecally. The present study was conducted to evaluate the efficacy of three different doses of dexmedetomidine when given intrathecally as an adjuvant to bupivacaine in patients undergoing lower abdominal surgery.
Methodology: After taking ethical committee approval, and obtaining patients consent, 60 American Society of Anesthesiologist (ASA) physical status I and II patients in the age group of 25-60 years undergoing lower abdominal surgeries were divided randomly into three groups in this randomized double blind study. An intrathecal dose of 2.5 ml of 0.5% hyperbaric bupivacaine was administered to all patients supplemented with dexmedetomidine. In Group A 5 mg, Group B 7.5 mg and in Group C 10 mg of dexmedetomidine was administered with a volume of 3ml of study drug in each group. The onset time, time to peak sensory level, motor blockade, sedation, duration of motor block, analgesia and any adverse effects were noted. At the end of the study, data were compiled and analyzed with appropriate statistical tests.
Results: Demographic profile was comparable in all the three groups. The time to onset of sensory block in Group C (1.55 ± 0.510 min) was significantly lower than Group A (2.15 ± 0.745 min) and Group B (2.20 ± 0.410 min). Sensory regression by two segments was significantly higher in Group C (220.65 ± 25.86 min) as compared to Group A (104.7 ± 25.5min) and Group B (145.10 ± 24.54 min). (p < 0.05) The duration of motor block was shortest in Group A (243.8 ± 22.0 min) as compared to Group B (305.4 ± 35.8 min) and Group C (387.0 ± 39.4 min). The visual analogue scale score for pain was lesser in Group C as compared to the other two groups.
Conclusion: Optimal dose of dexmedetomidine is difficult to define especially with prolonged duration of sensory and motor blockade in a dose dependent manner with minimal increase in side effects with higher doses. However, we conclude that 7.5 mg seems to be just optimal intrathecal dose of dexmedetomidine and can be used safely and effectively in lower abdominal surgeries.
Key words: Bupivacaine; Dexmedetomidine; Intrathecal; Postoperative analgesia.
Citation: Singh AP, Chawla S, Bajwa SJS, Khanna M. Efficacy and safety of dexmedetomidine as an intrathecal agent: a dose finding clinical study. Anaesth, Pain & Intensive Care 2017;21(1):13-18
Received: 31 Jan 2017; Reviewed: 2 Feb 2017; Corrected: 11 Feb 2017; Accepted: 14 Feb 2017
INTRODUCTION
With advancements in modern surgical and anesthetic techniques, trend is towards use of minimal and safe dose of local anesthetics (LA) made possible by addition of adjuvants to the LA solution.1 Higher dose of local anesthetics is associated with more complications and neurological sequelae.2 Addition of adjuvants to intrathecal anesthetics not only reduce the dose of local anesthetics but also minimize the incidence of complications as well as prolong post-operative analgesia.3,4 Alpha-2 agonists are widely used nowadays as they are free from the side effects associated with use of intrathecal opioids.5 Dexmedetomidine, a highly selective alpha-2 adrenoceptor agonist, is an imidazole compound. It produces dose dependent sedation; anxiolysis and analgesia without respiratory depression when used as a supplement or adjuvant.6 Alpha-2 adrenoreceptor agonists are also known to reduce anesthetic requirements because of their sympatholytic actions.7
Researchers have used dexmedetomidine intrathecally in varying doses with varying results. Though different studies have been published worldwide, there is no uniform consensus on optimal dose of intrathecal dexmedetomidine. Commonly 5 mg of intrathecal dexmedetomidine has been evaluated considering its dose equivalence to clonidine as 1:10 through intrathecal route. However, some researchers have used even 15-20 µg of intrathecal dexmedetomidine as an adjuvant which clearly establishes a non-consensus on optimal intrathecal dose of dexmedetomidine as an adjuvant.8
Keeping in consideration, the various merits of dexmedetomidine, the present study was performed to compare the analgesic efficacy of intrathecally administered dexmedetomidine as adjuvant in three different comparative doses with hyperbaric bupivacaine (0.5%) in patients undergoing lower abdominal surgeries so as to determine an optimal dose when used as an intrathecal adjuvant.
METHODOLOGY
The present study was conducted after obtaining the approval of institutional ethics committee. A written and informed consent was obtained from all patients for this prospective randomized, double blind study in which 60 ASA I and II patients in the age group of 25-60 years of either gender, undergoing elective lower abdominal surgery were included. Preoperatively patients were counseled and familiarized with the use of visual analogue scale (VAS) pain score for the assessment of perioperative pain. A sealed envelope was randomly selected and opened by an assistant, with instructions to draw up the relevant drug. The syringe was labeled with the patient’s coded number and handed to the investigator who performed the block. Three independent observers (senior anesthesiologist posted on duty, not included in the study) then observed the onset and offset of sensory and motor blockade and analgesia as per the written protocol supplied to them. Data was compiled when blinding was revealed at the end of the study. Considering a difference of 30 mins in post-op analgesia, a sample of 18 was considered adequate for the study keeping α-error at 0.05 and power of the study at 80%. However we took 20 patients in each group for better validation of results.
All the three groups received 2.5 ml of 0.5% hyperbaric bupivacaine with varying concentrations of dexmedetomidine and addition of saline to make a universal volume of 3 ml in each group. Group A patients were administered 5 mg of dexmedetomidine, Group B was administered 7.5 mg of dexmedetomidine while in Group C, patients were administered 10 mg of dexmedetomidine.
Patients on alpha adrenergic receptor blockers, local sepsis at the site of proposed puncture, known hypersensitivity to drug, treated with anticoagulants, CNS active drugs, suffering from bleeding diathesis or coagulation disorders, spinal deformity or tuberculosis of spine, having increased intracranial pressure were excluded from the study.
Preoperative assessment was done for each patient and patients were given tab. alprazolam 0.25 mg and tab ranitidine 150 mg at night before surgery and 6 am on the day of surgery. Patients were kept fasting as per the standard protocols and guidelines. Before shifting all the patients to operation theater, baseline parameters were observed and recorded which included an ECG (5 lead), noninvasive blood pressure and pulse oximeter and other vital parameters. An intravenous line was secured with 18 gauge cannula and patients were preloaded with ringer lactate 10 ml/kg 15-20 min before anesthesia. Subarachnoid block was performed with patients in sitting or left lateral position, using 25G Quincke needle and adopting a midline approach at L2-L3/L3-L4 intervertebral space. Patients were turned to supine position immediately and supplemental oxygen was administered. The following parameters were observed: time to onset of sensory blockade and motor blockade, time to maximum level of sensory blockade, time to attain complete motor blockade, two dermatomal segment regression time, total duration of motor blockade, sedation levels, post-op analgesia assessment using VAS, recue dosage of analgesia and any side effects occurring peri-operatively.
Sensory blockade was assessed using pinprick method with a blunt tipped needle every minute for the first 10 min, then at 5 min for the next 60 min and then every 15 min until regression to two segments from the maximum level achieved. Quality of motor blockade was assessed using modified Bromage scale. Inj fentanyl 100 µg was used as rescue analgesic in case of any complaint of pain.
Hemodynamic parameters were observed continuously and recorded every minute for first 10 min, then at 5 min for the next 60 min and then every 15 min till the end of surgery and then hourly during postoperative period for the next 6 hours.
Statistical analysis was done using student’s t- test for parametric data. Intergroup analysis was done using paired t-test. Chi-square test was used to analyze the categorical data and wherever frequencies were less than 5, Fisher’s exact test was used. Hemodynamic and other vitals were compared using ANOVA and applying post hoc significance. Kruskal Wallis H-test was used to assess the VAS scale. Values of p < 0.05 were considered statistically significant, and p < 0.001 was considered highly significant.
RESULTS
All the three groups were comparable in terms of age, weight, height and duration of surgery (Table 1). The distribution of the patients according to ASA grades was statistically insignificant (p > 0.05).
Table 1: Demographic variables in all the groups
| Variable | Group A
(n = 20) |
Group B
(n = 20) |
Group C
(n = 20) |
Intergroup significance |
| Age (years) | 46.35 ± 9.103 | 46.05 ± 8.249 | 45.50 ± 8.971 | NS |
| Gender (M/F) | 11/9 | 9/11 | 10/10 | NS |
| Weight (kg) | 68.70 ± 7.491 | 69.60 ± 8.444 | 68.95 ± 6.219 | NS |
| Height (cm) | 157.50 ± 2.819 | 158.55 ± 3.379 | 157.35 ± 4.017 | NS |
| Duration of surgery (min) | 79.25 ± 15.600 | 82.75 ± 10.062 | 81.00 ± 15.694 | NS |
| ASA Grade I* | 15 (75) | 14 (70) | 14 (70) | NS |
| ASA Grade II* | 5 (25) | 6 (30) | 6 (30) | NS |
Data: Mean ± SD; NS- Non-significant (p ˃ 0.05); *n (%)
The mean time of onset of sensory block in Group C was lower than Group A and Group B and the difference was statistically highly significant (p < 0.001). However, when Group A and B were compared it was statistically insignificant (p > 0.05) The mean time taken for regression of sensory block by two segments was statistically significantly higher in Group C as compared to the other two groups (p < 0.001)(Table 2).
The mean time taken for onset of motor block to grade 3 in all the three groups was statistically insignificant whereas the duration of motor block when compared in all the three groups was statistically significant (p < 0.001), Group A having the shortest duration of motor blockade. The duration of analgesia was significantly increased in Group C as compared to Group B and Group A (p < 0.001) (Table 2).
The mean pain score among all the three groups was statistically insignificant till 90 min postoperatively. At 105 min, there was increase in visual analogue scale (VAS) in Group A which was statistically significant as compared to Group B and Group C. At 240 min the difference between Group B and C became statistically significant. At 24 hours the pain score among the three groups showed statistically significant difference with lowest VAS in Group C (p < 0.05) (Table 3).
Table 2: Showing comparison of regional anesthesia characteristics in all the groups
Regional anesthesia parameters
| Group A
mean ± SD |
Group B
mean ± SD |
Group C
mean ± SD |
Intergroup Significance | |||
| A to B | A to C | B to C | ||||
| Onset of sensory blockade (mins) | 2.15 ± 0.745 | 2.20 ± 0.410 | 1.55 ± 0.510 | NS | HS | HS |
| Time to 2 segment sensory regression (mins) | 104.7 ± 25.5 | 145.10 ± 24.542 | 220.65 ± 25.869 | HS | HS | HS |
| Motor Block to reach Bromage 3 (mins) | 3.10 ± 0.852 | 3.30 ± 1.031 | 2.75 ± 1.020 | NS | NS | NS |
| Regression to bromage scale 0 (mins) | 243.8 ± 22.0 | 305.4 ± 35.8 | 387.0 ± 39.4 | HS | HS | HS |
| Duration of Analgesia (mins) | 200.0 ± 26.8 | 310.0 ± 65.8 | 412.7 ± 68.4 | S | HS | S |
Data: Mean ± SD, NS-Non-significant (p > 0.05), HS-Highly significant (p < 0.001)
Table 3: Showing visual analogue scale at various time interval post-operatively
| Time (min) | Group A
mean ± SD |
Group B
mean ± SD |
Group C
mean ± SD |
Intergroup Significance | ||
| Group A to B | Group A to C | Group B to C | ||||
| 75 | 0.30 ± 0.57 | 0 | 0 | NS | NS | NS |
| 90 | 0.80 ± 0.75 | 0 | 0 | NS | NS | NS |
| 105 | 2.9 ± 1.21 | 0 | 0 | S | S | NS |
| 120 | 3.85 ± 0.98 | 0 | 0 | S | S | NS |
| 240 | 4.80 ± 0.69 | 1.90 ± 0.68 | 0 | S | S | S |
| 360 | 5.60 ± 0.59 | 3.50 ± 0.68 | 0 | S | S | S |
| 480 | 6.80 ± 0.69 | 4.75 ± 0.71 | 2.90 ± 0.85 | S | S | S |
| 720 | 7.55 ± 0.68 | 5.85 ± 0.81 | 4.35 ± 0.74 | S | S | S |
| 1440 | 7.90 ± 0.30 | 6.85 ± 0.88 | 6.25 ± 0.81 | NS | S | S |
Data: Mean ± SD, S-significant (p < 0.05), NS-Non-significant (p ˃ 0.05)
Table 4: Showing total no. of doses of rescue analgesia and total dosage
| Group A | Group B | Group C | Intergroup Significance | |||
| n = 20 | n = 20 | n = 20 | A to B | A to C | B to C | |
| No. of doses | 12.6 ± 1.4 | 8.4 ± 1.1 | 6.75 ± 1.0 | HS | HS | S |
| Total Dosage | 900 ± 4.5 | 630 ± 8.4 | 502 ± 7.6 | HS | HS | S |
Data: Mean ± SD, HS- Highly significant (p < 0.01), S- Significant (p < 0.05)
Figure 1: Showing the incidence of adverse effects in all the three groups
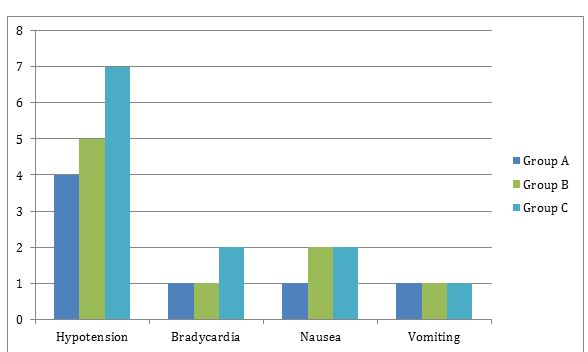
On intergroup analysis the requirement of rescue analgesia was significantly lower statistically in Group C as compared to Group B and Group A (p < 0.001) (Table 4). The intergroup comparison of adverse effects was found to be statistically insignificant (p > 0.05) and there was no incidence of adverse effects like dry mouth, urinary retention, and respiratory depression (Figure 1).
DISCUSSION:
In order to maintain the advantage of low dose local anesthetics while improving intra operative quality of anesthesia, different agents have been used as adjuvants for prolonging the duration of spinal anesthesia.9 The usage of lower doses of local anesthetic minimizes the incidence of side effects associated with higher dosages.10
Increasing use of dexmedetomidine has almost revolutionized the modern anesthesia practice. However, its off-label use as an intrathecal agent still remains questionable in terms of dosage. It has been used in varying doses with varying results with local anesthetic agents such as bupivacaine.
Dexmedetomidine and clonidine have been extensively studied and employed clinically to achieve the desired effects in regional anesthesia.11-14 The mechanism of action include their binding to presynaptic C fibers and postsynaptic dorsal horn neurons which causes decreased release of C fiber neurotransmitters and hyper polarization of postsynaptic dorsal horn neurons.15, 16 However, no study till date has compared the different lower intrathecal doses of dexmedetomidine for optimal clinical effectiveness. In the present study, the time taken from the completion of injection of study drug till the patient felt loss to pin prick sensation at T10 was considered as time of onset of sensory block. On comparing all the groups there was decrease in onset time with dose dependent increase of intrathecal doses of dexmedetomidine whereas no significant difference in the level of sensory blockade was found with increasing dose of dexmedetomidine. This finding was in concordance with Al- Mustafa et al.17 and Saadawy I et al.18 (Table 2).
There was a significant increase in regression time with 10 mg dose in comparison with 5 mg and 7.5 mg. This observation was in accordance with the study results of Al-Mustafa et al.17 and Eid MD et al.19 The possible reason for this finding could be that dexmedetomidine stimulates alpha-2 receptors directly in the spinal cord, thus inhibiting the firing of nociceptive neurons. The local anesthetics act by blocking sodium channels, whereas the alpha-2 adrenoceptor agonists act by binding to presynaptic C fibers and postsynaptic dorsal horn neurons.20 The effect of dexmedetomidine is additive or synergistic to the action of local anesthetics and increases in dose dependent manner (Table 2).
In the present study no statistical difference was observed in the duration of onset of motor block to grade 3. Similar results were seen by Eid et al.19 and Sunil et al.21 (Table 2). This is a surprising result as dose dependency is seen more in sensory effect and minimal in onset of motor blockade with intrathecal dexmedetomidine.
The patients receiving 10 mg of dexmedetomidine showed maximum prolongation in duration of motor block (387 min) followed by 7.5 mg of dexmedetomidine (305 min) and least prolongation in patients receiving 5 mg of dexmedetomidine (243 min). The most probable reason for this might be direct impairment of excitatory amino acid release from spinal interneurons. (Table 2). This observation was comparable with earlier studies done by Al-Mustafa et al.17, Eid MD et al.19 and Sunil et al.21
The total dosage of rescue analgesics was significantly decreased with higher dose of dexmedetomidine; this was in accordance to Gupta et al.4 This could have been because of the administration of alpha-2 adrenergic agonist through intrathecal and epidural route provides augmentation of analgesic effect in postoperative period. At spinal cord level, activation of both a2-C and a2-ARs, in the neurons of superficial dorsal horn especially lamina II, directly reduces pain transmission, by suppressing the release of pro-nociceptive transmitter, substance P and glutamate from primary afferent terminals and by hyperpolarizing spinal interneurons via G-protein mediated activation of potassium channels.22, 23
The incidence of adverse effects was comparable in all the three groups and was statistically non-significant. Cardiovascular depression from alpha-2 adrenergic agonists can occur at both brain and spinal cord sites. Postsynaptic activation of a2adrenoceptors in the central nervous system inhibits sympathetic activity and thus can decrease blood pressure and heart rate.24
Major limitations of our study were that we could not analyze biochemically the blood concentration of drugs due to non-availability of such advanced facilities at our institution. The optimal dose is difficult to define as postoperatively sensory and motor blockade increases in dose dependent manner. However, these effects can be utilized in different clinical situations on individual basis where early or late discharge, early or late mobility or prolonged sensory and motor blockade is warranted. Both 7.5 and 10 mg of dexmedetomidine can be used intrathecally for better surgical anesthesia and post-operative comfort. Though on statistical comparison, incidence of side effects is not significant, clinically 10 mg of dexmedetomidine is associated with a higher incidence of side effects as compared to 7.5 and 5 mg of dexmedetomidine. On the other hand, 7.5 mg of dexmedetomidine is more effective as compared to 5 mg dose which is clearly evident from the regional anesthesia characteristics in the present study.
CONCLUSION:
To conclude, addition of dexmedetomidine prolonged the sensory and motor block significantly when used with hyperbaric bupivacaine intrathecally in a dose dependent manner, without increasing the incidence of significant adverse effects. From the findings of the present study, 7.5 mg of dexmedetomidine seems to be a better intrathecal adjuvant dose to local anesthetics clinically as compared to other two doses with regards to optimal synergistic effects and minimal side effects.
Conflict of interest: Nil
Author contribution: All authors took part in design, planning, conduct of the study and data collection, literature review and manuscript preparation
REFERENCES:
- Gupta M, Shailaja S, Hedge K.S. Comparison of intrathecal Dexmedetomidine with Buprenorphine as adjuvant to Bupivacaine in Spinal Anaesthesia. J Clin Diagn Res. 2014 Feb;8(2):114-7. doi: 10.7860/JCDR/2014/7883.4023 [PubMed] [Free full text]
- Lambert, Donald H. Complications of spinal anesthesia. International. Anesthesiology. Clinics. 1989; 27(1): 51-55. [Free full text]
- Bajwa SJ, Bajwa SK, Kaur J, Singh A, Bakshi G, Singh K, et al. Admixture of clonidine and fentanyl to ropivacaine in epidural anesthesia for lower abdominal surgery. Anesth Essays Res. 2010 Jan-Jun;4(1):9-14. [PubMed] [Free full text]
- Gupta R, Verma R, Bogra J, Kohli M, Raman R, Kushwaha JK. A comparative study of intrathecal dexmedetomidine and fentanyl as adjuvants to bupivacaine. J Anaesthesiol Clin Pharmacol. 2011 Jul;27(3):339-43. doi: 10.4103/0970-9185.83678. [PubMed] [Free full text]
- Bajwa S, Arora V, Kaur J, Singh A, Parmar SS. Comparative evaluation of dexmedetomidine and fentanyl for epidural analgesia in lower limb orthopedic surgeries. Saudi J Anaesth. 2011 Oct;5(4):365-70. doi: 10.4103/1658-354X.87264. [PubMed] [Free full text]
- Bajwa S, Kulshrestha A. Dexmedetomidine: An adjuvant making large inroads into clinical practice. Ann Med Health Sci Res. 2013 Oct;3(4):475-83. [PubMed] [Free full text]
- Kamibayashi T, Maze M. Clinical uses of α2- adrenergic agonists. Anesthesiology 2000 Nov;9(5)3:1345-9 . [PubMed] [Free full text]
- Eid HA, Shafie M, Yousef H. Dose –Related prolongation of Hyperbaric Bupivacaine Spinal Anaesthesia By Dexmedetomidine.Ain Shams J Anesthesiol 2011;4:83-95.
- Buvanendran A, Krroin JS. Useful adjuvants for post-operative pain management. Best Pract Res Clin Anaesthesiol. 2007 Mar;21(1):31-49. [PubMed]
- Bernards CM. Epidural and spinal anaesthesia. In: Barash PG, Cullin BF, Stoelting RK editors. Clinical Anaesthesia. 5th Philadelphia: Lippincott Williams & Wilkins; 2009.p. 928-37.
- Bajwa SJ, Bajwa SK, Kaur J, Singh A, Bakshi G, Singh K, et al. Admixture of clonidine and fentanyl to ropivacaine in epidural anesthesia for lower abdominal surgery. Anesth Essays Res. 2010 Jam-Jun;4(1):9-14. doi: 10.4103/0259-1162.69299. [PubMed] [Free full text]
- Hall JE, Jurich TD, Barney JA, Arian SR, Ebert TJ. Sedative amnestic and analgesic properties of small dose of dexmedetomidine infusions. Anesth Analg. 2009 Mar; 90(3):699-705. [PubMed]
- Bajwa SJ, Bajwa SK, Kaur J, Singh G, Arora V, Gupta S, et al. Dexmedetomidine and clonidine in epidural anaesthesia: A comparative evaluation. Indian J Anaesth. 2011 Mar;55(2):116-21. doi: 10.4103/0019-5049.79883. [PubMed] [Free full text]
- Gabrial JS, Gordin V. Alpha-2 agonists in regional anaesthesia and Curr Opin Anaesthesiol. 2001 Dec;14(6):751-3. [PubMed]
- Bajwa SJ, Gupta S, Kaur J, Singh A, Parmar S. Reduction in the incidence of shivering with perioperative dexmedetomidine: A randomized prospective study. J Anaesthesiol Clin Pharmacol. 2012 Jan;28(1):86-91. doi: 10.4103/0970-9185.92452. [PubMed] [Free full text]
- Eisenach James C, De Kock M, Klimscha W. Alpha sub 2-adrenergic agonist for regional anesthesia. A clinical review of clonidine (1984-1995). Anesthesiology. 1996 Sep; 85(3):655-74. [PubMed] [Free full text]
- Al-Mustafa MM, Abu-Halaweh SA, Aloweidi AS, Murshidi MM, Ammari BA, Awwad ZM, et al. Effect of dexmedetomidine added to spinal bupivacaine for urological procedures. Saudi Med J. 2009 Mar;30(3):365-70. [PubMed]
- Saadawy I, Boker A, Eishahawy MA, Almaazrooa A, Melibary S, Abdellatif AA, et al. Effect of dexmedetomidine on the characteristics of bupivacaine in a caudal block in pediatrics. Acta Anaesthesiol Scand. 2009 Feb;53(2):251-6. doi: 10.1111/j.1399-6576.2008.01818 [PubMed] [Free full text]
- Shah A, Patel I, Gandhi R. Haemodynamic effect of intrathecal dexmedetomidine added to ropivacaine intraoperatively and for postoperative analgesia. Int J Bas Clin Pharmacol 2013; 2(1):26-9. [Free full text]
- Sunil BV, Sahana KS, Jajee PR. “ Dexmedetomidine as an adjuvant with hyperbaric bupivacaine for spinal anaesthesia: a double blind controlled study”. J Evolution med dent Sci 2013, 2(40), 7604-11.
- Grewal A. Dexmedetomidine: New avenues. J Anaesthesiol Clin Pharmacol. 2011 Jul; 27(3):297-302. [PubMed] [Free full text]
- Ishii H, Kohno T, Yamakura T, Ikoma M, Baba H. Action of dexmedetomidine on the substantia gelatinosa neurons of the rat spinal cord. Euro J Neurosci. 2008 Jun; 27(12):3183-90. doi: 10.1111/j.1460-9568.2008.06260 [PubMed] [Free full text]
- Eisenach JC, Shafer SL, Bucklin BA, Jackson C, Kallio A. Pharmacokinetics and pharmacodynamics of intraspinal dexmedetomidine in sheep. Anesthesiology. 1994 Jun;80(6):134-59. [PubMed]

