Waqas Ashraf Chaudhary, MD, MS (Pain Mgt.)*,Hafiz Sheraz Arshad, BSPT, PP-DPT**, Sarwat Waqas, BSc, Pharm-D***, Farah Khan Jatoi, DPT**, Umar Khyyam DPT**, Maryam Tahir Dar, Pharm-D**, Amna Amhed, Pharm-D**, Qasim Zafar, Pharm-D***
*Center for Pain Management, Iqbal Mahmuda (Trust) Hospital, Lahore (Pakistan)
**Azra Naheed Medical College, Superior University, Lahore (Pakistan)
***University College of Pharmacy, University of Lahore, Lahore (Pakistan)
Correspondence: Dr. Waqas Ashraf Chaudhary, Department of Physiotherapy, Azra Naheed Medical College, Superior University, 18km Raiwind Road, Lahore (Pakistan); E-mail: wacc1-12@alumni.le.ac.uk; pain.specialist@yahoo.com; pain.clinic42@gmail.com
ABSTRACT
Vitamin D deficiency has been implicated in chronic musculoskeletal pain. This article reviews two cases of improvement/resolution of chronic pain after vitamin D repletion in a pain practice setting in Lahore. Vitamin D insufficiency is common and its repletion may improve chronic musculoskeletal pain and improve quality of life. In some cases, it may result in complete resolution of symptoms. We observed 521 patients over a period of 1 year, of which 2 patients are described in detail. Written informed consent was taken from the patients to publish this report.
Key words: Vitamin D; Chronic musculoskeletal pain; Cholecalciferol (D3); Ergocalciferol (D2); 1, 25 hydroxyvitamin D; Osteomalacia; Myopathy
Citation: Chaudhary WA, Arshad SH, Waqas S, Jatoi FK, Khyyam U, Dar MT, Amhed A. Vitamin D supplement as an adjuvant in the management of chronic musculoskeletal pain. Anaesth Pain & Intensive Care 2013;17(3):296-300
INTRODUCTION
Pain is the most common complaint for which patients seek medical care and much of it is chronic, involving muscles, bones and joints. Chronic pain is among many conditions that have recently been associated with vitamin D deficiency.1 A number of studies have suggested a link between low levels of vitamin D and higher incidence of chronic musculoskeletal pain.2 According to peer reviewed clinical research examining adult patients of all ages, chronic musculoskeletal pain of various types,3 muscle weakness or fatigue, fibromyalgia syndrome, rheumatic disorders, osteoarthritis, hyperesthesia, migraine headaches, and other chronic somatic complaints have been linked to low vitamin D level. It also has been implicated in the mood disturbances of chronic fatigue syndrome and seasonal affective disorder.4 For many patients, helping to ameliorate these problems may be as simple, well tolerated, and economical as an extra daily supplement of vitamin D.
Vitamin D is required for the differentiation, proliferation, and maturation of cartilage cells and for the production of proteoglycan synthesis in articular chondrocytes.5 Restoring vitamin D levels to normal in patients with osteomalacia has resulted in complete resolution of pain within 4 weeks.5 Improvement in pain and weakness in patients with osteomalacia has also been noted. Patients who have chronic, nonspecific pain may have an underlying vitamin D insufficiency/deficiency.6
Adequate vitamin D intake is essential for healthy bones to maintain calcium and phosphorus homeostasis. However, vitamin D does far more than build strong bones; in fact, its major biologically active metabolite works throughout the human body at special receptors. Vitamin D and its roles in health and disease have recently received great interest both in the scientific community7 and in the popular press. Many tissues express vitamin D receptors and it is not surprising that a physiological role for vitamin D has been proposed in a number of organs and organ systems, not limited to the skeleton. Vitamin D deficiency likewise has been implicated in a number of diseases ranging from autoimmune disease and diabetes to cardiovascular disease, various cancers, and chronic pain. A meta-analysis even suggested reduced all-cause-mortality with vitamin D supplementation..
Vitamin D has 2 major forms D2 (ergocalciferol) and D3 (cholecalciferol). Vitamin D3 is synthesized in skin via exposure to ultraviolet B (UVB) radiation from sunlight.9 It also is consumed to a small extent in the diet, primarily from fatty fish, while vitamin D2 is found in only a few plant based foods. Additionally, certain foods, such as milk or cereals, may be fortified with vitamin D – usually D3. Both D2 and D3 are absorbed in the small intestine, and neither has any biological activity.10 They then go through a two-stage process of metabolism, first in the liver to form 25-hydroxyvitamin D, abbreviated as 25(OH) D, also called calcidiol.
This has minimal biological activity but is stored in many tissues, particularly adipose tissue, and it is the major circulating form of vitamin D in the blood. The 25(OH)D metabolite is then converted primarily in the kidneys to 1,25-dihydroxyvitamin D abbreviated as 1,25(OH)2D and also called calcitriol, which is the most important and biologically active vitamin D metabolite. The ultimate role of vitamin D, via the 1, 25(OH)2 D metabolite, is to facilitate the absorption of dietary calcium from the intestine. 1, 25(OH)2 D functions as a hormone, and sustains a wide range of metabolic and physiologic functions throughout the body.11 This has been referred to as the “vitamin D endocrine system.”
Risk factors for persistence or recurrence of low back pain (LBP) after surgery include infection and smoking. A less common reason is low vitamin D levels. Repletion with 1000 IU/day improves vitamin D status by approximately 20 to 25 nmol/L, and 2000/day IU will improve levels by 40 to 50 nmol/L, except in darker-skinned people, who may require significantly more. Patients with back pain who also have other risk factors for low vitamin D levels may benefit from a screening 25 (OH) D levels and repletion therapy. Muscle pain has been commonly seen in patients with vitamin D deficiency,12 as well as myopathy, which usually resolves more quickly than bone pain.13
CASE REPORT 1
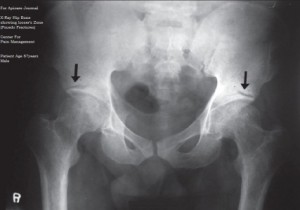
A 67-year-old man had a 2 to 3 years history of episodic headache. The headache was described as dull, mild and holocephalic, occasional exacerbations escalating to moderate to severe intensity. Stress and sleep deprivation were noted as triggering factors. Photophobia, phonophobia, nausea or vomiting were not reported. Physical exercise did not worsen the headache. The frequency was 5–6 times per month in early part of the illness; however, it increased to more than 15 times per month for the last 5 months. The duration of an episode ranged from a few hours to few days. General and neurological examinations were unremarkable. He was prescribed metoclopramide, propranolol 40 mg and amitryptiline (75 mg PO daily) as preventive therapy and naproxen (550 mg PO) as abortive therapy. He came after one month for follow-up in pain clinic and reported no significant improvement in the frequency or intensity of his headache. In fact, he noted development of new symptoms and now experienced pain in his back and both lower limbs. He also complained of difficulty in climbing stairs and getting up from a sitting posture. He had curtailed his physical activity because of easy fatigability. Repeat neurological examination revealed quadriparesis (especially proximal muscles) with normal muscle bulk and deep tendon reflexes. Sensation was intact and gait was unremarkable. There was generalized muscle tenderness (including pericranial muscle). Bone tenderness was noted on the tibia, femur, humerus, ulna, and skull. A working diagnosis of osteomalacia (with myopathy) was made and the patient was subjected to various investigations. Hip joint AP view film showed demineralization and Looser’s zone (pseudofracture). Serum (total) calcium was 7.8 mg/100 ml (normal 8.5–10.5 mg/dl), serum ionized calcium was 3.8 mg/100 ml (4.5–5.6 mg/dl), serum phosphate 2.2 mg/100 ml (3–4.5 mg/dl), serum alkaline phosphatase 264 IU/Ll (30–120 IU/Ll), 25-hydroxy vitamin D [25(OH) D] 5 ng/ml (> 20 ng/ml), serum parathyroid hormone (PTH) 162 pg/ml (10–60 pg/ml), serum creatine kinase phosphokinase (CPK) 212 U/L (25–200 U/Ll). Electromyography (EMG) showed short and low muscle action potentials (MUAP’s), polyphasic MUAP’s and early recruitment. Other routine hematological and biochemical examinations were within normal limits. Muscle and bone biopsy were not done. The cause of vitamin D deficiency (osteomalacia) was not evident, and it was considered nutritional in origin. The patient was treated with vitamin D 200000 IU capsules and then maintenance dose of vitamin D3 (1500 IU/day) with calcium supplementation (1500 mg/day) and propranolol 40 mg, duloxetine 30 mg and naproxen (550 mg) with physical therapy + TENS as adjuvants. Within 4 weeks the head and body pains started to improve. Headache subsided completely in 6 weeks, while body pains were relieved within 11 weeks. Motor weakness improved to near normal in 6 months. However, mild bone tenderness was present up to 7 months after the initiation of vitamin D and calcium therapy. Repeat vitamin D level was normal after 9 months.
Figure 1: X-Ray hip joint showing Looser’s zone (pseudofractures)
CASE REPORT 2
A 49-year-old woman presented with 1.5 year history of LBP. She described the pain as mild, burning, radiating to leg for most of the times; occasional exacerbations, escalating to moderate to severe intensity (occasionally requiring bed rest) for a few hours. The frequency of exacerbation was 1–2 times per month in the early part of the illness. However, it had increased to 3–4 times per week for the last few months and she was unable to do household tasks, such as washing the floor or hovering. She was unable to participate in any social welfare activities that she previously used to do. Her pain was only partially controlled with ibuprofen 400 mg PO. On direct questioning, she admitted to having intermittent muscle aches, especially of the low back, hip and lower limb for the last few months. She denied the presence of any muscle weakness. However, she had curtailed her physical activities because of easy fatigability.
Neurological examination revealed symmetrical proximal muscles of lower limb with normal muscle bulk. All deep tendon reflexes were brisk, and the plantar responses were flexor. Sensation was normal. There was generalized muscle and bone tenderness.
Investigations revealed her total serum calcium 7.1 mg/100 ml, serum ionized calcium 2.7 mg/dl, serum phosphate 2.2 mg/100 ml, serum alkaline phosphatase 263 IU/L, and vitamin D (25[OH] D) 8 ng/ml. EMG confirmed myopathy. Other routine haematological and biochemical examinations (including CBC and ESR) were normal. Muscle and bone biopsies were not performed. Vitamin D deficiency was probably due to under nutrition as there was no other obvious cause for it.
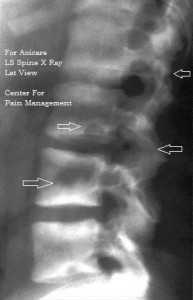
The patient was treated with Vitamin D Stat Cap 200000 IU and then vitamin D (1500 IU/day) with calcium supplementation (1500 mg/day), tramadol for pain and advised physiotherapy and TENS as adjuvant therapy. Within a few weeks of commencing the treatment her headache started to improve. The headache and spontaneous muscle pain subsided completely in about 5 weeks’ duration. The bone tenderness reduced markedly in about 4 months’. Vitamin D level returned to normal after 3.5 months. Her symptoms of back pain and spasms resolved completely within 6 months and she was able to do her routine work, and participate in social work. Her mood improved markedly.
Figure 2: X-ray L/S spine lateral view
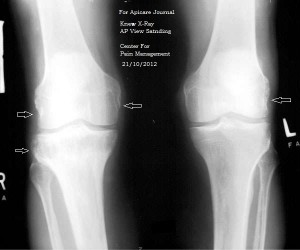
Fig 3. X-ray knee (bilateral) AP view standing (OA)
DISCUSSION
For a country like Pakistan that gets so much sun, it’s pretty unbelievable that so many Pakistanis are vitamin-D deficient. The first thing one need to know about vitamin-D is that it’s not really a vitamin at all! It’s in fact more of a ‘sun hormone’ that is produced naturally in the human body when the skin is exposed to sunlight. Food and sun-light (ultra-violet rays to be exact) are the best sources of vitamin D. In the case of a vitamin D deficiency, the absorption of calcium and phosphorous is reduced, which can adversely affect the strength of teeth and bones. This can lead to conditions such as rickets, osteomalacia and osteoporosis. Since vitamin D also aids in reducing cancer cell growth, controlling infections, blood pressure and artery damage, a deficiency can increase the risk of diabetes, cardiovascular and autoimmune diseases and cancer. A very common symptom is depression, fatigue and being more asthma-prone.
A deficiency could be a result of insufficient exposure to the sun due to excessive sun block, an indoors lifestyle or simply wearing the wrong kind of fabric, or inefficient synthesis in the body.
The process that links vitamin D to musculoskeletal pain is presumed to begin with a lack of circulating calcium (hypocalcemia) due to inadequate vitamin D. This calcium deficiency stimulates increased parathyroid hormone secretion and sets in motion a cascade of biochemical reactions negatively affecting bone metabolism. Even mild hypocalcemia results in an elevation of parathyroid hormone that can diminish bone density (osteopenia) and/or more severely affect bone architecture (osteoporosis).14
The effect relating most closely to musculoskeletal aches and pains is that the increase in parathyroid hormone levels can lead to a softening of bone surfaces – or osteomalacia – which generates pain in periosteal tissues covering the skeleton. Osteomalacia has been proposed as an explanation of why many patients with vitamin D inadequacies may complain of dull, persistent, generalized musculoskeletal aches and pains. Myopathy also is part of the osteomalacic complex, so fatigue or decreased muscle strength, usually in lower limbs, may appear prior to any pain.15
In many cases involving pain and myopathy, defects of bone metabolism and osteomalacia may not be clinically detectable but are nonetheless present, or are “subclinical.” Such disorders are considered to be nonspecific or idiopathic in that an explanatory injury, bone pathology, or anatomical or neurological defect is not evident.16 This may occur in a significant number of chronic musculoskeletal pain cases that have been resistant to treatment, and, according to the research, many of the problems can be attributed to inadequate vitamin D.
Clinical researchers have found that the role of vitamin D extends beyond bone and muscle involvement in chronic musculoskeletal pain syndromes.17 For example, vitamin D receptors have been found in various brain structures, the spinal cord, and sensory ganglia. Accordingly, results of some studies suggest benefits of vitamin D supplementation in helping to ameliorate neuropathies, as well as relieving mood disturbances such as depression or anxiety often associated with pain syndromes or seasonal affective disorder (SAD).18
While a universal consensus is lacking, most researchers agree that a minimum 25(OH)D serum level of about 30 ng/ml or more is necessary for favourable calcium absorption, suppression of parathyroid hormone (PTH), and good health. Optimal 25(OH)D concentrations generally range up to 50 ng/ml or somewhat higher, although those greater than 150 ng/ml can be toxic.19
Deficiency has been defined as circulating 25(OH)D concentrations <20 ng/ ml, which may be associated with elevated PTH and greater bone turnover, potentially fostering subclinical osteomalacia. Concentrations ≤8 ng/ml have been considered as highly predictive of fully-developed, or clinical, osteomalacia. Furthermore, levels of circulating 25(OH)D concentrations below an optimal range but above true deficiency status i.e., 20 to 29 ng/ml could have a negative impact on health and constitute insufficiency.20
According to the latest US Dietary Guidelines, at least 1000 IU/day of vitamin D3 is necessary to maintain adequate serum 25(OH)D levels at or above 30 ng/ml in healthy persons (without chronic pain).21 Prior guidance from the US Institute of Medicine in 1997, specified daily vitamin D intake ranging from 200 IU/day in children and younger adults to 600 IU/day in those older than age 70. Still, many experts believe that even the newly recommended 1000 IU/ day may be inadequate for healthy persons, and some have suggested that up to 4000 IU/day of vitamin D3 could be optimal. For patients with chronic musculoskeletal pain syndromes, 2000 IU/day or more may be proposed as a reasonable and conservative approach.21
From a Pain Clinic perspective, a number of factors may suggest that chronic musculoskeletal pain and related problems could be due to inadequate vitamin D intake. Researchers have stressed that the “gold standard” for a presumptive diagnosis is a review of patient history, lifestyle, and dietary habits that might pose risks for deficiency.22 Indicators of defects in bone metabolism may include chronic muscle, bone, or joint pains, as well as persistent muscle weakness, fatigue, and possibly difficulty walking. Signs / symptoms of hypocalcemia and/or clinical osteomalacia most typically relate to severe vitamin D deficiency and are more likely to appear late in the course of the disease.23 Based on positive clinical findings, a trial of vitamin D supplementation might be recommended to the patient without further assessment. This takes into account that; a) vitamin D is well tolerated, with minimal likelihood of adverse effects, b) over-the-counter supplements are very economical, and c) laboratory assessments of biochemical markers sometimes provide little additional information of value.24
From the clinical research to date, it appears that individual responses to vitamin D therapy can vary. Other factors such as sun exposure, season of year, diet, age, physical health, and medications can also play vital roles. Therefore, optimal vitamin D dosing requirements for individual patients having different types of chronic musculoskeletal pain and related symptoms are not fully defined.25 Various vitamin D dosing protocols to rectify inadequacies of the 25(OH) D metabolism in patients with pain syndromes have been reported in the literature. Some are quite aggressive, using higher doses of vitamin D ranging from 5,000 IU/day to 50,000 IU/day, or greater, for limited periods of time. More conservatively, a recently reported trial of 2000 IU/day in patients with neuropathic pain produced a 67% increase in 25(OH) D levels and a 50% improvement in pain scores during 3 months of treatment. In some cases, the particular chronic pain condition might be such that it cannot be alleviated by vitamin D3 supplementation alone. For example, there may be a previously undetected anatomic defect, disease, or other pathology that would benefit from another type of therapeutic intervention in addition to vitamin D therapy.26
When vitamin D supplementation is effective, some patients may start to notice improvements within weeks, if they are alert to subtle changes. Others may become discouraged unless they are advised at the start that vitamin D supplementation could take a number of months, even up to 9 months,27 to reach its full potential in helping to relieve musculoskeletal aches, pains, and/or related symptoms. In most cases rapid and complete pain resolution would be an unrealistic expectation.The evidence for pain-relieving and other benefits of vitamin D therapy is suggestive of a potential range of improvements, some more obvious than others. Physicians should have a high index of suspicion for low vitamin D levels in patients with LBP,and determining the vitamin D status in these patients may be important. In those cases with vitamin D insufficiency/deficiency, repletion of vitamin D may offer dramatic results. Determining vitamin D levels and vitamin D repletion before and after back surgery may be prudent.27 The patients in this study who responded best used between 4000 and 5000 IU of vitamin D3/day.
CONCLUSION
This case series supports information that has recently become apparent in the literature about vitamin D deficiency and its influence on chronic musculoskeletal pain. Doses in the range of 4000 to 5000 IU of vitamin D3/day may be needed for an adequate response. Guidelines from the Institute of Medicine call for increasing the Recommended Dietary Allowance (RDA) of vitamin D to 600 international units (IU) for everyone aged 1-70, and raising it to 800 IU for adults older than 70 to optimize bone health. The adequate vitamin D daily intake for Pakistani population also needs to be set at least twice of that is recommended intakes for Western populations.
Acknowledgments: The pain research on vitamin D is supported by the Scottman Pharma (Pvt.) Ltd., who is a pioneer in Halal Vitamin D3 capsule manufacturing in Pakistan. We acknowledge their help with gratitude.
REFERENCES
- Holick MF. Vitamin D deficiency. N Engl J Med. 2007 Jul 19;357(3):266-81. [Publisher]
- Atherton K, Berry DJ, Parsons T. Macfarlane GJ.Power C.Vitamin D and chronic widespread pain in a white middle-aged British population: evidence from a cross-sectional population survey. Ann Rheum Dis. 2009 Jun;68(6): 817–822.[PubMed]
- Benson J, Wilson A, Stocks A and Moulding N. Muscle pain as an indicator of vitamin D deficiency in an urban Australian Aboriginal population. Med J Aust. 2006 July;185(2):76–77. [Scopus]
- Lotfi A, Abdel-Nasser AM, Hamdy A, Omran AA, El-Rehany MA. Hypovitaminosis D in female patients with chronic low back pain. Clin Rheumatol. 2007 Nov;26(11):1895-901. [PubMed]
- Mitsikostas DD, Tsaklakidou D, Athanasiadis N and Thomas A. The prevalence of headache in Greece: correlations to latitude and climatological factors. Headache. 2002 Jun;36(3):168–173. [Publisher]
- Saps M, Blank C, Khan S, et al. Seasonal variation in the presentation of abdominal pain. J Ped Gastro Nutri. 2008;46(3):279–284. [Scopus]
- Zeng QY, Chen R, Xiao ZY, et al. Low prevalence of knee and back pain in southeast China;the Shantou COPCORD study. J Rheumatol. 2004;31(12):2439–2443.[Scopus]
- Straube S, Moore RA, Derry S and McQuay H J. Vitamin D and chronic pain. Pain. 2009;141(1-2):10–13. [PubMed]
- Glerup H, Mikkelsen K, Poulsen L, et al. Hypovitaminosis D myopathy without biochemical signs of osteomalacic bone involvement. Calc Tis Intel. 2000;66(6): 419–424, 2000. [Publisher]
- Grootjans-Geerts I and Wielders JPM. A pilot study of hypovitaminosis D in apparently healthy, veiled, Turkish women: 82% had severe vitamin D deficiency. Nederlands Tijdschrift voor Geneeskunde. 2002;146(23):1100–1101. [Scopus]
- Holvik K, Meyer HE, Haug E and Brunvand L. Prevalence and predictors of vitamin D deficiency in five immigrant groups living in Oslo, Norway: the Oslo Immigrant Health Study. Euro J Clin Nut. 2005;59(1): 57–63. [Publisher]
- Mytton J, Frater AP, Oakley G, Murphy E, Barber MJ and Jahfar S. Vitamin D deficiency in multicultural primary care: a case series of 299 patients. Bri J Gen Pract. 2007;57(540):577–579. [Scopus]
- Serhan E, Newton P, Ali HA, Walford S and Singh BM. Prevalence of hypovitaminosis D in Indo-Asian patients attending a rheumatology clinic. Bone. 1999;25(5):609–611. [Scopus]
- Macfarlane GJ, Palmer B, Roy D, Afzal C, Silman AJ and O’Neill T. An excess of widespread pain among South Asians: are low levels of vitamin D implicated? Ann Rheum Dis. 2005;64(8):1217–1219. [Google Scholar]
- Erkal MZ, Wilde J, Bilgin Y, et al. High prevalence of vitamin D deficiency. secondary hyperparathyroidism and generalized bone pain in Turkish immigrants in Germany: identification of risk factors. Osteoporos Int. 2006;17(8):1133–1140. [PubMed]
- Laurance J. The virtues of Vitamin D: it’s time we saw the light. The Independent. 2007 Sep. Available on [http://www.independent.co.uk/life-style/health-and-families/health-news/the-virtues-of-vitamin-d-its-time-we-saw-the-light-402749.html] Accessed on 27 July 2013.
- Autier P and Gandini S. Vitamin D supplementation and total mortality: a meta-analysis of randomized controlled trials. Arch Intern Med. 2007;167(16):1730–1737. [Publisher]
- Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG and Gluud C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Systematic Rev. 2008;(2) CD007176. [Publisher]
- Adams JS and Lee G. Gains in bone mineral density with resolution of vitamin D intoxication. Ann Int Med.1997;127(3)203–206. [Scopus]
- Nellen JF, Smulders YM, Jos Frissen PH, Slaats EH and Silberbusch J. Hypovitaminosis D in immigrant women: slow to be diagnosed. BMJ. 1996;312(7030):570–572. [Scopus]
- De Torrenté de la Jara G, Pécoud A and Favrat B. Musculoskeletal pain in female asylum seekers and hypovitaminosis D3. BMJ. 2004;329(7458):156–157.[Scopus]
- Lowenthal MN and Shany S. Osteomalacia in Bedouin women of the Negev. I J Med Sci. 1994;30(7):520–523. [Scopus]
- Plotnikoff GA and Quigley JM. Prevalence of severe hypovitaminosis D in patients with persistent, nonspecific musculoskeletal pain. Mayo Clinic Proceedings. 2003;78(12):1463–1470. [Scopus]
- Helliwel PS, Ibrahim GH, Karim Z, Sokoll K and Johnson H. Unexplained musculoskeletal pain in people of South Asian ethnic group referred to a rheumatology clinic-relationship to biochemical osteomalacia, persistence over time and response to treatment with calcium and vitamin D. Clin Experimental Rheumatol. 2006;24(4):424–427. [Scopus]
- Bartley J. Prevalence of vitamin D deficiency among patients attending a multidisciplinary tertiary pain clinic. N Zealand Med J. 2008;121(1286):57–62. [Scopus]
26. Badsha H, Daher M and Ooi Kong K. Myalgias or non-specific muscle pain in Arab or Indo-Pakistani patients may indicate vitamin D deficiency. Clin Rheumatol. 2009;28(8):971–973.[PubMed]
27. Schwalfenberg G. Improvement of chronic back pain or failed back surgery with vitamin D repletion: a case series. J Am Board Fam Med. 2009;22(1):69–74. [PubMed]