Henrik Hack
Consultant Anaesthetist,
Dept. of Paediatric Anaesthesia, Royal Manchester Children’s Hospital, Manchester, UK.
Correspondence: Dr. H. Hack, Consultant Anaesthetist, Dept of Paediatric Anaesthesia, Starship Children’s Hospital, Auckland (New Zealand); E-mail: henrikh@adhb.govt.nz
ABSTRACT
Phenoxybenzamine has traditionally been used to control the hypertension associated with pheochromocytoma in children. However, doxazosin has been proposed as the alternative agent of choice, based upon the claimed ability to effectively control perioperative hypertension with a single daily dose without the occurrence of problematic side effects sometimes associated with the use of phenoxybenzamine, e.g. the need for additional β blockers to control secondary tachycardia or vasopressors to correct resistant hypotension following tumor removal.
The perioperative course of two siblings, undergoing anesthesia for removal of extra-adrenal pheochromocytomas (paragangliomas) are compared. Doxazosin and phenoxybenzamine were used in their respective management for preoperative control of hypertension. The proposed benefits of doxazosin over phenoxybenzamine were not observed and the intraoperative control of hemodynamics was significantly poorer with doxazosin. The perioperative course of each sibling is compared and the published evidence supporting the use of doxazosin in children with pheochromocytoma is critically reviewed.
Keywords: Pheochromocytoma; Hypertension; Doxazosin; Phenoxybenzamine; Esophageal Doppler.
Citation: Hack H. Use of doxazosin and phenoxybenzamine in two children with pheochromocytoma. Anaesth Pain & Intensive Care 2013;17(2):198-201
INTRODUCTION
Adequate preoperative control of blood pressure is recognized as the cornerstone of safe management of pheochromocytoma.1 This has traditionally been provided by the nonselective α-blocker phenoxybenzamine. In adults doxazosin, an α 1 blocker, has been reported to provide good perioperative blood pressure control without the troublesome side effects associated with phenoxybenzamine.2 This has led some to consider doxazosin as the agent of choice in children.3 However published data regarding its use in children with pheochromocytoma is limited.
Two cases of related children with extra-adrenal pheochromocytomas (paragangliomas), operated upon by the same surgeon and who received identical anesthetic management except for differing control of their preoperative hypertension, are reported.
CASE REPORT 1
A thirteen year old girl (weight 42kg) was referred from another hospital with hypertension (200/130mm Hg). Subsequent investigations revealed an elevated twenty four hour urinary catecholamine excretion (micromol/24hr): normetanephrine =50.2 (N 0-4.3), metanephrine=0.6 (N 0-2.0). Plasma normetanephrine was >24 (N<1.09) and metanephrine was 0.20 (N<0.46). MRI scan revealed an isolated mass overlying the right psoas muscle adjacent to L3/4 vertebrae. MIBG scan was otherwise negative. Fasting plasma glucose was normal. A diagnosis of a single, extra-adrenal pheochromocytoma (paraganglioma) was made. Transthoracic echocardiogram revealed a mildly thickened left ventricular wall with good function (fractional shortening 35%).
Initial treatment consisted of amlodipine (5 mg OD) and atenolol (25 mg OD). Hypertension persisted despite increasing doses, so doxazosin (2 mg OD increased to 6 mg OD) was added. Labile hypertension persisted, till good control was finally achieved (BP120/85 mmHg, HR 80-100/min) on a combination of doxazosin (4 mg mane, 6 mg nocte) and atenolol (100 mg OD). Intravenous fluids (normal saline plus free oral fluids) were started the day before surgery.
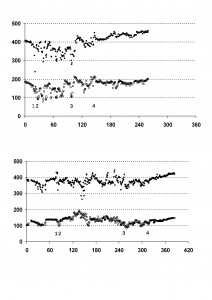
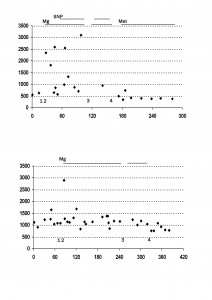
She was premedicated with midazolam (0.5 mg/kg PO) and anesthesia induced with a combination of propofol, remifentanil and atracurium, as previously reported.4 This produced a stable induction. Anesthesia was maintained with a remifentanil infusion, air/sevoflurane mixture and inj. atracurium. Standard monitoring plus direct arterial pressure, central venous pressure and noninvasive cardiac output measurement (Cardio QP esophageal Doppler monitor, Deltex Medical, UK) were used.4 Surgical incision produced a small rise in BP associated with a fall in peak velocity (PV) and corrected flow time (FTc) that was easily controlled with an increase in the remifentanil infusion rate. Initial surgical exploration provoked repeated rises in blood pressure up to 190/100 mmHg , associated with falls in PV, FTc. and elevated systemic vascular resistance (SVR 2348 dynes.s.cm-5) (Figs. 1&2 ). These were treated with repeated boluses of inj. magnesium sulphate (MgSO4) 25 mg/kg followed by an increasing infusion rate up to 70 mg/kg/hr. Despite this the BP remained very labile during further dissection and sodium nitroprusside (SNP) was added. SNP at 4 µg/kg/min lowered the BP to 100/55 (SVR 867). However any reduction of the SNP infusion rate provoked a rise in BP (maximum BP 208/116, SVR 3105) requiring elevation of the SNP back to 4 µg/kg/min. The MgSO4 remained at 70 mg/kg/hr. BP control was complicated by a brief episode of bleeding from the IVC requiring a temporary cessation of the SNP and MgSO4 together with fluid boluses. Following restoration of an adequate circulating volume both agents were recommenced and continued until 5-10 minutes before the point of anticipated tumor isolation. At this time colloid and blood were infused as usual. Approximately ten minutes after tumor isolation the BP fell (minimum 60/35 mmHg). The initial response to further colloid infusion was disappointing and required the use of boluses of metaraminol (0.5-1.0 mg IV) to achieve a BP>80 systolic. Further boluses of metaraminol (total dose given 20 mg) together with fluid were required to maintain hemodynamic stability (BP >80/40, SVR 380-750). (Figs.1&2).
The patient was extubated at the end of the operation. Total intraoperative and immediate post operative fluid requirements were: Hartmann’s solution 10 ml/kg, Gelofusin 8.3 ml/kg, packed blood cells 12 ml/kg, fresh frozen plasma (FFP) 12 ml/kg. Recovery was uneventful and further vasopressors were not required.
CASE REPORT 2
Following screening of the family of Case 1, her younger sister (9 years; 23 kg) was found to be hypertensive (BP180/120, HR 75-115). Twenty-four hour urine collection confirmed elevated catecholamine excretion: norepinephrine= 0.64 (N 0-0.11 mmol/mol creatinine), dopamine 0.4 (N 0-0.75), HMMA =15.5 (N 0-5.5) and HVA=4.2 (N 0-0.75). CT scan and MRI revealed a mass anteromedial to the right kidney, intimately related to the IVC and renal vessels. MIBG scan confirmed a solitary lesion. Echocardiography showed mild left ventricular hypertrophy (fractional shortening 30%).
One week after commencing phenoxybenzamine (20 mg BID), BP was stable at 110/60 mmHg. Atenolol 25 mg OD was added to control mild tachycardia (95-120 bpm). She was readmitted preoperatively to start intravenous fluids (normal saline plus free oral fluids). Her BP remained stable at 105/65 mmHg supine, 100/60 mmHg erect; HR 80 bpm.
She received an identical general anesthetic to Case 1. Before open surgery was commenced a trans-jugular balloon catheter was placed under radiological guidance into the upper, hepatic portion of the IVC in case of subsequent massive venous hemorrhage. This was not required. The child remained stable throughout. Start of surgery produced a mild rise in BP, which was easily controlled by an increase in the remifentanil infusion rate. Initial exploration and tumor handling produced a rise in BP to 140/85 mmHg (SVR 2886 dynes.s.cm-5) (Figs 1&2). This was treated with a bolus of MgSO4 50 mg/kg and commencement of an infusion at 50 mg/kg/hr with good effect (BP 100/50 mmHg, SVR 1111). Hemodynamic parameters (PV, FTc and SVR) were easily maintained (Figs 1 & 2) by an infusion of MgSO4 with occasional additional boluses (25 mg/kg). Maximum BP subsequently recorded was 145/60 mmHg.
Figure 1: Changes in FTc (msec) (♦) and PV (cm/s) (□) with Time (mins) for Case 1(upper) & Case 2 (lower). 1. Start of surgery. 2. Start of tumour handling and dissection. 3. Significant bleeding. 4. Approximate time of tumour isolation and removal.
Figure 2: Changes in SVR (dynes.s.cm-5) (♦) with time (mins) for Case 1(upper) & Case 2 (lower). 1. Start of surgery. 2. Start of tumour handling and dissection. 3. Significant bleeding. 4. Approximate time of tumour isolation and removal. Mg = Magnesium sulphate infusion +boluses ; SNP= Sodium Nitroprusside infusion; Met = Metaraminol boluses.
An episode of bleeding from the IVC responded to volume replacement. Clear identification of the tumor’s venous drainage was difficult and made it difficult to anticipate when MgSO4 should be stopped and fluid loading started. Subsequently there was a brief drop in BP to 65/35 mmHg that responded rapidly to fluid loading alone. Hemodynamics remained stable for the rest of the operation (BP 80-90/35-50 mmHg, SVR 750-1000) without the need for vasopressors. Total fluids administered were Hartmann’s solution 20 ml/kg, gelofusin 70 ml/kg, packed blood cells 20 ml/kg and FFP 20 ml/kg. Postoperatively she remained stable on a routine crystalloid infusion.
Histology showed both tumors to be extra-adrenal pheochromocytomas (paragangliomas).
DISCUSSION
Doxazosin, a competitive α 1-blocker, has been recommended as the agent of choice for the preoperative control of hypertension in adults and subsequently children with pheochromocytoma.2,3,5 This has been based upon several supposed pharmacological advantages. Firstly, it has a long duration of action allowing once daily dosage. Secondly, the absence of any presynaptic α2-blockademeans that there should be no reflex tachycardia requiring additional β-blockade. Thirdly, it produces good perioperative blood pressure control and lastly, its short duration of action (compared to phenoxybenzamine) means that any hypotension following tumor removal does not require large volume transfusions or vasopressor agents.
But what other clinical evidence is there to support its use? Doxazosin has been reportedly used in a wide range of dosage regimes with and without additional β-blockade. However, scrutiny of the published data, particularly in the non–anaesthetic literature, reveals a lack of crucial detail regarding both preoperative regimes used to achieve “adequate” preoperative control and the subsequent intraoperative control observed.6-9 One report comparing phenoxybenzamine with doxazosin in children appeared to show no clinically significant differences8. A large, comparative case series comparing phenoxybenzamine and doxazosin in adults with pheochromocytoma found that the use of phenoxybenzamine was associated with better intraoperative hemodynamic control of hypertension. The incidence of hypotension following tumour removal was similar in both groups although the use of vasopressors was more frequent in the phenoxybenzamine group. There was however a greater use of intravenous fluids in the doxazosin group. They found no significant difference in outcome between the two techniques9.
Doxazosin has been used to control the hypertension associated with a catecholamine secreting neuroblastoma in a child10. Using combined general and thoracic epidural anesthesia the simultaneous use of three hypotensive agents was required to control intraoperative hypertension. Following tumor removal volume transfusion combined with both phenylephrine and dopamine infusions were required to maintain an adequate blood pressure.
The children in this report were related and presented with identical tumors. With the exception of their preoperative blood pressure control their surgical and anesthetic management was identical. In Case 1 the difficulty in controlling the preoperative hypertension with doxazosin was disappointing. None of the previously claimed advantages of its use were observed. The use of phenoxybenzamine in Case 2 produced a much more rapid and sustained control of hypertension. More importantly the use of phenoxybenzamine was associated with much easier control of intraoperative hypertension. The frequent and dramatic changes in PV and FTc (reflecting sudden increases in afterload) observed in Case 1 were less frequent and far easier to control with a single hypotensive agent than in Case 2. This presumably reflects a greater degree of α adrenergic blockade achieved by phenoxybenzamine compared to doxazosin.
The rarity of this condition probably precludes adequately powered & controlled trials and so the limited evidence from reports such as this or the larger, adult series of Weingarten9 become more important when comparing efficacy of individual drugs.
CONCLUSION
In conclusion, the originally proposed advantages for the use of doxazosin over phenoxybenzamine for the perioperative control of hypertension in children with pheochromocytoma have not been supported by subsequently published evidence. In addition the detailed observations reported here lend no support to the recommendation to the use of doxazosin rather than phenoxybenzamine. Of particular concern to the anesthesiologist is the possibility of poorer intraoperative hemodynamic control when doxazosin is used.
Conflict of interest: No conflict of interest declared.
- Written parental consent for publication obtained for both cases.
- An abstract of this report was presented at the European Society of Paediatric Anaesthetists ASM, Mallorca, Spain 2011.
REFERENCES
- Hack H. The perioperative management of children with pheochromocytoma. Pediatr Anesth 2000;10:463–476. [PubMed]
- Pry-Roberts C, Farndon J. Efficacy and safety of doxazosin for perioperative management of patients with pheochromocytoma. World J Surg. 2002;26:1037-1042. [PubMed]
- Tobias J. Preoperative blood pressure management of children with catecholamine-secreting tumours: time for a change. Pediatr Anesth 2005;15:537-540. [PubMed]
- Hack H. Use of the Esophageal Doppler machine to help guide the intraoperative management of two children with pheochromocytoma. Pediatr Anesth 2006;16:867–876. [PubMed]
- Bravo EL, Tagle R. Pheochromocytomas: State of the art and future prospects. Endo Rev 2003;24(4):539-553. [PubMed] [Free Full Text]
- Beltsevich DG, Kuznetsov NS, Karazyan AM, Lysenko MA. Pheochromocytoma surgery: Epidemiological peculiarities in children. World J Surg 2004;28:592-596. [PubMed]
- Ganesh HK, Acharya SV, Goerge J, Bandgar TR, Menon PS, Shah NS. Pheochromocytoma in children and adolescents. Indian J Pediatr 2009;76(11):1-3. [PubMed]
- Kocak S, Aydintung S, Canakci N. α blockade in preoperative preparation of patients with pheochromocytomas. Int Surg 2002;87:191-194. [PubMed]
- Weingarten TN, Cata JP, O’Hara JF, Prybilla DJ, Pike TL, Thompson GB et al. Comparison of two preoperative medical management strategies for laparoscopic resection of pheochromocytoma. Urology 2010;76(2):508.e6-11. [PubMed]
- Seefelder C, Sparks JW, Chirnomas D, Diller l, Schamberger RC. Perioperative management of a child with severe hypertension from a catecholamine secreting neuroblastoma. Pediatr Anesth 2005;15:606-610. [PubMed]