1Department of Anesthesiology and Critical Care, PT. B. D Sharma PGIMS, Rohtak Haryana 124001 (India)
2Department of Critical and Intensive Care, All India Institute of Medical Sciences (AIIMS)- 110029 (India)
Correspondence: Dr Arnab Banerjee, 184D, Arjun Nagar, New Delhi-110029 (India); Phone: 7042716189; E-mail: dr.arnab77@gmail.com
ABSTRACT
Introduction: Optic nerve sheath diameter (ONSD) using ocular ultrasound is increasingly used now a days as noninvasive means of detecting raised intracranial pressure. We conducted a cross‐sectional, observational study to assess its validity, optimal cutoff limit and inter‐observer variability in our ICU patients.
Methodology: The adult patients admitted to our ICU during six month interval (Jan 2014‐June 2014) were enrolled for the study. The exclusion criteria were patients breathing spontaneously, presence of hyperthyroidism, optic nerve tumor, neuritis, glaucoma and orbital fracture. The patients were divided into two groups. Group I – those having increased ICP clinically or as per radiological findings. Group II – patients having no features of raised ICP. A linear probe with a frequency 13‐7 MHz, SonositeTM USA, was used to measure ONSD by two blinded, experienced investigators. Mean of three readings was taken. The entire data was compiled and analyzed applying appropriate statistical tests.
Results: A total of 114 patients were enrolled and 100 completed the study; 65 in group I and 35 in group II. Mean ONSD in two groups were 0.62 ± 0.07 cm versus 0.476 ± 0.040 (p < 0.001); and 0.62 ± 0.07 versus 0.47 ± 0.042 (p < 0.001) in the right and left eye respectively. The inter-observer variability was nonsignificant, the sensitivity was 88.46%, specificity 93.54%, positive predictive value 95.83%, negative predictive value 82.85% where 5.0mm was taken as cutoff limit.
Conclusion: Bedside ONSD measured with ultrasound is a good screening tool for detecting elevated intracranial pressure in invasively ventilated ICU patients.
Key words: Intracranial pressure; Monitoring; Noninvasive method; Ultrasound; Optic nervesheath diameter
Citation: Bala R, Banerjee A. A study on ultrasonographic measurement of optic nerve sheath diameter for detecting raised intracranial pressure in intensive care unit patients. Anaesth Pain & Intensive Care 2017;21(4): 432‐437
Received: 4 Feb 2017; Reviewed: 6 Mar, 25 Apr 2017; Corrected: 29 Jul 2017; Accepted: 6 Aug 2017
INTRODUCTION
The patients admitted in intensive care unit (ICU) may have raised intracranial pressure (ICP); various etiologies being head injury, stroke, meningoencephalitis, liver failure, metabolic encephalopathy, eclampsia, hanging or drowning etc.1-4 By inducing neurological insults, intracranial hypertension leads to increased morbidity, mortality and poor neurological outcome. Hence its early diagnosis and prompt treatment is of paramount importance.5 The invasive ICP monitoring is considered gold standard but it carries several pitfalls such as risks of hemorrhage, infection andblockage.6 Thus, there is a definite need for noninvasive methods of ICP monitoring. Though several techniques are available, ultrasonographic (USG) measurement of optic nerve sheath diameter (ONSD) has sparked significant interest in recent years. The advantages are bed‐side availability, noninvasive, reliable, inexpensive and ability to repeat any times.7 Literature review revealed studies showing correlation between raised ICP and ONSD but most of the studies are of small sample size and comprised primarily of head injury patients.8-9 In the light of above findings, the present study was planned with a hypothesis that patients diagnosed with increased ICP clinically or radiologically will have increased ONSD. The primary objective was to assess the validity of ONSD measured ultrasonographically for raised ICP. The secondary objectives were to detect inter-observer variability and to find out optimal cutoff value of ONSD to detect raised ICP. The optic nerve sheath is in direct communication with the intracranial subarachnoid space. It is this relationship that forms the physiological basis for using the optic nerve sheath as a surrogate for intracranial pressure measurement. The anatomical relationships underpinning the use of ultrasound to measure ONSD can be readily appreciated on MRI
METHODOLOGY
After obtaining approval from institutional ethics committee, the present cross‐sectional, observational study was conducted in the ICU of our department. The adult patients admitted in the ICU during Jan 2014 to June 2014 were enrolled for the study depending upon the availability of investigators. The spontaneously breathing patients, those having glaucoma, optic nerve tumor, neuritis, orbital fracture, or hyperthyroidism were excluded from the study. Informed written consent for participation in the study was taken from legal guardians of the patients. Detail clinical history and indications for ICU stay was obtained. A thorough general and systemic examination was carried out. Ideal weight of the patients was taken by measuring the height of the patient. The routine as well as special investigations such as CT scan, MRI were checked. The patients were divided into two groups; Group 1 and Group 2 depending upon the presence or absence of raised ICP respectively. The clinical signs and symptoms such as headache, nausea, vomiting, altered sensorium or findings in Computerized Tomography (CT) scan or Magnetic Resonance Imaging (MRI) as reported by radiologist with more than five year experience in the field were suggestive of intracranial hypertension. The vital parameters (heart rate, blood pressure, oxygen saturation, end tidal carbon dioxide and temperature) were recorded. The treatment chart as well as ventilator settings were also checked. Ocular ultrasound was performed using SonositeTM M‐turbo (Sonosite Inc, Bothell, WA, USA) ultrasound machine with a 13‐6 MHz linear‐array probe with orbital imaging settings. The measurements were performed by two investigators who had performed more than 30 ocular ultrasounds for ONSD measurements. Both of them were blinded to patient’s diagnosis and clinical history. The patients were positioned supine with 20‐30◦ head‐up. TegadermTM was applied on closed eyelid and copious jelly was put on probe which was then placed on the superior and lateral aspect of the orbit. It was moved gently from temporal to nasal side until the optic nerve was visualized as a linear hypoechoic structures with clearly defined margins posterior to the globe. The diameter of optic nerve was measured 3 mm behind the globe (Figure1). The images were obtained in transverse plane and the movements of probe were gentle avoiding any pressure on the globe. Average of three readings was taken for each eye. Similarly the procedure was repeated by second investigator. The patients were monitored closely throughout the procedure for any complication or untoward event.
Sample size was calculated keeping in view at most 5% risk, with minimum 80% power and 5% significance level (significant at 95% confidence level) based on the primary objective. If the true relative risk of failure for experimental subjects was 0.55, it was estimated that study of 84 experimental subjects would be able to reject the null hypothesis that this relative risk equals 1 with probability (power) 0.8. The Type I error probability associated with this test of null hypothesis is 0.05.
Statistical analysis: The continuous variables are presented as mean ± SD and compared using student’s t test. The categorical data are expressed as frequency and percentage and compared using Chi square test. The sensitivity, specificity, positive predictive value and negative predictive value along with 95% confidence interval was calculated. Inter and Intra-observer variability was analyzed using Cohen’s kappa coefficient. The receiver operating characteristic curve (ROC) was generated to determine the optimal cutoff value of ONSD. All data were analyzed using SPSS software package (SPSS) version 16, Chicago, Il and p < 0.05 was considered as statistically significant.
RESULTS
A total of 114 patients were enrolled and 100 completed the study; 65 in Group 1 and 35 in Group 2. Demographic profile and patient distribution are shown in Table 1 and 2.
Table 1: Demographic profile of the patients
| Parameters | Group 1
(n=65 ) |
Group 2
(n=35 ) |
| Age (in years) | 48 ± 10( 38 – 58) | 50 ± 10 ( 40 – 50) |
| Gender (male/female) | 48/55 (48%/ 55%) | 52/45 (52%/ 45%) |
| Weight (in Kg) | 60 ± 4 (56-64 ) | 72 ± 5 (67-77 ) |
| Height (in cm) | 164 ± 10 (154-174 ) | 160 ± 10 (150-170 ) |
Table 2: Distribution of patients as per the diagnosis in both groups
| Diagnosis | N (%) |
| Polytrauma | 30 (30) |
| Surgical patients | 30 (30) |
| Poisoning | 8 (8) |
| Eclampsia | 20 (20) |
| Respiratory Distress/ COPD | 9 (9) |
| Metabolic encephalopathy | 2 (2) |
| Others | 1 (1) |
| Total | 100 |
There was no significant difference in vital signs recorded (Table 3). There was statistically significant increase of ONSD in right eye (0.62 ± 0.067 cm) and left eye (0.62 ± 0.07 cm) in patients with neurological symptoms (Group 1) in comparison to ONSD in right eye and left eye (0.47 ± 0.04 cm, p < 0.001)in patients having no features of raised ICP (Group 2) (Table 4 ).
Table 3: Showing vital parameters of the patients in two groups
| Parameters | Group 1
(n=65) |
Group 2
(n=35) |
p value |
| SpO2 (%) | 99 ± 2 | 98 ± 2 | 1.000 |
| HR (beats/min) | 102.46 ± 14.54 | 102.54 ± 12.88 | 1.000 |
| SBP (mmHg) | 134.88 ± 18.58 | 125.45 ± 13.01 | 0.063 |
| DBP (mmHg) | 75.98 ± 8.05 | 72.30 ± 11.71 | 0.700 |
| MAP (mmHg) | 88.73 ± 10.31 | 83.12 ± 11.77 | 0.102 |
| Temp (°C) | 36 ± 1° C | 36 ± 0.8°C | 1.00 |
| EtCO2 (mmHg) | 36 ± 2 | 35 ± 2 | 1.00 |
Table 4: Optic nerve sheath diameter in two groups. Measurements made by two observers in both the eyes
| Eyes | Observer | Group 1
(n=65 ) |
Group 2
(n=35 ) |
P value |
| Right Eye | 1st observer | 0.629 ± 0.067 | 0.476 ± 0.040 | < 0.001 |
| 2nd observer | 0.629 ± 0.070 | 0.471 ± 0.042 | ||
| Left Eye | 1st observer | 0.624 ± 0.071 | 0.473 ± 0.044 | < 0.001 |
| 2nd observer | 0.626 ± 0.070 | 0.476 ± 0.043 |
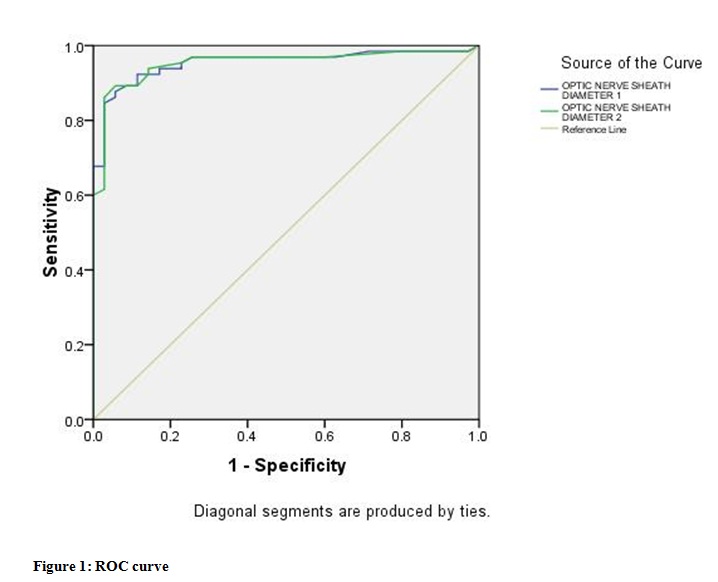
After analyzing the results with Cohen’s Kappa coefficient, with regard to inter-observer variability, there was a good strength of agreement for sonography at a depth of 3 mm (K=0.83). The sensitivity was 88.46%, specificity 93.54%, positive predictive value 95.83%, negative predictive value 82.85% where 5.0 mm was taken as a cutoff limit. The higher ONSD values suitably predicted definite neurological symptoms (area under curve ROC 0.954 and 95% confidence interval were 0.912 and 0.995) (Table 5, Figure 1).
Table 5: Showing measurements of area under the curve (Group I)
| Test Result Variable(s) | Area | Std. Errora | Asymptotic Sig.b | Asymptotic 95% Confidence Interval | |
| Lower Bound | Upper Bound | ||||
| ONSD Right eye | 0.954 | 0.021 | 0 | 0.912 | 0.995 |
| ONSD Left eye | 0.953 | 0.022 | 0 | 0.91 | 0.996 |
A cutoff of 0.51 cm predicted definite neurological symptoms with a sensitivity 93% of and specificity of 89% (Table 6). Scatter diagrams of ONSD of Group 1 and Group 2 are shown in Figures 3 and 4 respectively.
Table 6: Accuracy of different values of ONSD for detecting raised ICP (Group I)
| Positive if ≥
|
Sensitivity | 1 – Specificity | |
| ONSD Right eye | 0.5075 | 0.923 | 0.171 |
| 0.5125 | 0.923 | 0.143 | |
| 0.5175 | 0.923 | 0.114 | |
| 0.525 | 0.908 | 0.114 | |
| 0.5325 | 0.892 | 0.114 | |
| ONSD Left eye | 0.49 | 0.969 | 0.257 |
| 0.5 | 0.954 | 0.229 | |
| 0.51 | 0.938 | 0.143 | |
| 0.52 | 0.923 | 0.143 | |
| 0.53 | 0.892 | 0.114 |
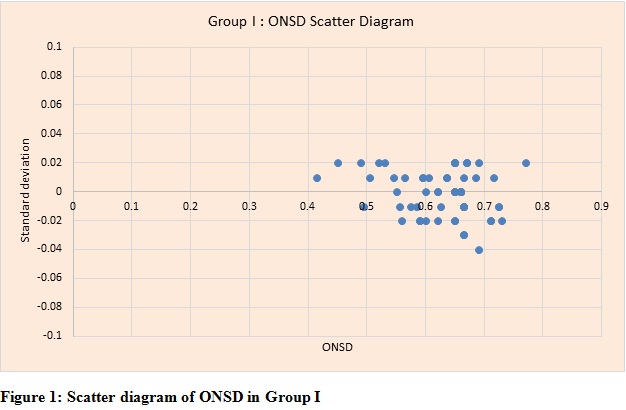
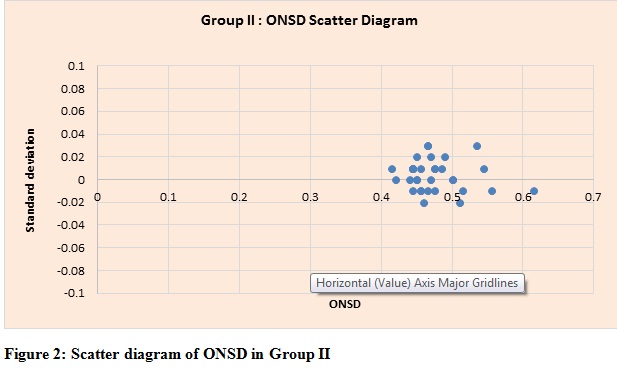
DISCUSSION
The use of ultrasound to measure optic nerve sheath diameter has been described in the literature for a decade. The raised ICP is transmitted through subarachnoid space to optic nerve sheath leading to its distension. It occurs rapidly, hence making optic nerve ultrasound quite useful for the early detection of acute elevation in ICP.10
In our study we found good correlation between raised ICP and ONSD assessed by ocular ultrasound. The criteria of raised ICP was presence of clinical features and/or radiological findings. The intraventricular catheter, the gold standard for ICP monitoring was not used for
comparison since we had heterogeneous group of patients such as eclampsia, hepatic encephalopathy and it was not wise to venture invasive monitoring in them.9Furthermore,earlier studies have clearly documented that ONSD has good correlation with ICP measured invasively.11 Measuring ICP non‐invasively is very important in ICU patients. Neurological examination is difficult or unreliable because the patients are either sedated or paralyzed. Invasive monitoring is expensive, require neurosurgical expertise and may lead to bleeding or infection. The lacuna is being filled by USG‐measured ONSD which is noninvasive, reliable, and inexpensive, can be performed bedside and repeated any times. The optimal cut‐off value of ONSD is still controversial and different authors have taken different values.9 We took 5.0 mm as the cut‐off value which is most commonly described in the literature.5,14-17 The sensitivity and specificity of our study corroborate to previous findings.12 The ONSD may be different in spontaneously breathing patients and those on ventilators. To overcome this bias we enrolled only invasively ventilated patients. Ocular ultrasound like any other technique has learner’s curve as the investigator may require time to obtain reliable and reproducible image and differentiate the artifacts. Both the investigators in our study had the experience of performing scans in more than 30 normal and deceased patients. Ultrasonography is criticized for exhibiting inter-observer variability; in order to overcome it we took average of three readings and found inter-observer variability negligible. The safety of ocular ultrasound is also questioned; however we performed it under ophthalmic settings. There was no untoward event or complication in any patient owing to the procedure. The interesting finding of our study was increased ONSD in eclampsia patients. The most likely mechanism was vasogenic edema as described earlier. It is this group of patients who are true indication of noninvasive monitoring since impaired coagulation profile preclude invasive monitoring. Most of the earlier studies have tested it in neurosurgical patients but our study population comprised of heterogeneous group. They should be screened for intracranial hypertension and this requirement is best met by noninvasive technique.13 Ocular ultrasound being easily available in most of the settings is the perfect tool. It should never be considered as an alternative or substitute of invasive method because both have different indications and pitfalls. Our study had few limitations. First, ONSD was not compared with ICP measured with intraventricular catheter. Second, the timings of radiological scans and clinical findings did not coincide with ocular ultrasound. The patients might have undergone treatment during the intervening period which can alter ICP. Third, the ventilator settings were not uniform in all patients and it may alter ONSD. However, this was beyond the scope of present study and could be a future research topic. Fourth, serial monitoring of ONSD and its relation with patient’s outcome was not studied.
CONCLUSION
We conclude that ONSD measured by ocular ultrasound offers a good and practical method to detect raised ICP in intubated ventilated patients. It helps in detecting the cerebral insults, monitor neurological status, guide treatment strategy and predict prognosis. Further research comprising of large number of patients is required to verify its validity and utility so that this easily available technique can be used more effectively.
Conflict of interest: None declared by the authors. The study was completed with departmental resources.
Author contribution:
RB – Manuscript editing
B – Concept, conduct of study
REFERENCES
- Nabeela HW, Bahr NC, Rhein J, Fossland N, Kiragga AN, Meya DB, et al. Accuracy of noninvasive intraocular pressure or optic nerve sheath diameter measurements for predicting elevated intracranial pressure in cryptococcal meningitis. Open Forum Infect Dis. 2014 Oct 11;1(3):ofu093. doi: 10.1093/ofid/ofu093. [PubMed] [Free full text]
- Kim YK, Seo H, Yu J, Hwang GS. Noninvasive estimation of raised intracranial pressure using ocular ultrasonography in liver transplant recipients with acute liver failure -A report of two cases-. Korean J Anesthesiol. 2013 May;64(5):451–455. [PubMed] [Free full text]
- Manno E, Motevallian M, Mfochive A, Navarra M. Ultrasonography Of The Optic Nerve Sheath Suggested Elevated Intracranial Pressure In Epilepsy: Case Report. Internet J Anesthesiol. 2013;26(1).
- Fagenholz PJ, Gutman JA, Murray AF, Noble VE, Camargo CA Jr, Harris NS. Optic nerve sheath diameter correlates with the presence and severity of acute mountain sickness: evidence for increased intracranial pressure. J Appl Physiol (1985). 2009 Apr;106(4):1207–1211. doi: 10.1152/japplphysiol.01188.2007 [PubMed] [Free full text]
- Tayal VS, Neulander M, Norton HJ, Foster T, Saunders T, Blaivas M. Emergency department sonographic measurement of optic nerve sheath diameter to detect findings of increased intracranial pressure in adult head injury patients. Ann Emerg Med. 2007 Apr;49(4):508–514. [PubMed]
- Hwan Kim Y, Ho Lee J, Kun Hong C,Won Cho K, Hoon Yeo J, Ju Kang M, et al. Feasibility of optic nerve sheath diameter measured on initial brain computed tomography as an early neurologic outcome predictor after cardiac arrest. Acad Emerg Med. 2014 Oct;21(10):1121–1128. doi: 10.1111/acem.12477 [PubMed] [Free full text]
- Legrand A, Jeanjean P, Delanghe F, Peltier J, Lecat B, Dupont H. Estimation of optic nerve sheath diameter on an initial brain computed tomography scan can contribute prognostic information in traumatic brain injury patients. Crit Care. 2013 Mar 27;17(2):R61. doi: 10.1186/cc12589. [PubMed] [Free full text]
- Ballantyne SA,O’Neill G, Hamilton R, Hollman Observer variation in the sonographic measurement of optic nerve sheath diameter in normal adult. Eur J Ultrasound. 2002 Oct;15(3):145-9 . [PubMed]
- Shevlin C. Optic Nerve Sheath Ultrasound for the Bedside Diagnosis of Intracranial Hypertension: Pitfalls and Potential. Critical Care Horizons. 2015;1:22-30. [PubMed] [Free full text]
- The Brain Trauma Foundation. The American Association of neurological Surgeons. The joint Section on Neurotrauma and Critical Care.Indications for intracranial pressure monitoring. J.Neurotrauma. 2000 Jun-Jul;17(6-7):479-91. [PubMed]
- Rajajee V, Vanaman M, Fletcher JJ, Jacobs TL. Optic nerve ultrasound for the detection of raised intracranial pressure. Neurocrit Care. 2011 Dec;15(3):506‑ doi: 10.1007/s12028-011-9606-8 [PubMed]
- Shirodkar CG, Rao SM, Mutkule DP, Harde YR, Venkategowda PM, Mahesh MU. Optic nerve sheath diameter as a marker for evaluation and prognostication of intracranial pressure in Indian patients: An observational study. Indian J Crit Care Med. 2014 Nov;18(11):728-34. [PubMed] [Free full text]
- Hamilton DR,Sargsyan AE, Melton SL, Garcia KM, Oddo B, Kwon DS, et al. Sonography for determining the optic nerve sheath diameter with increasing intracranial pressure in a porcine model. J Ultrasound Med. 2011 May;30(5):651-659. [PubMed] [Free full text]
- Hansen HC, Helmke K. Validation of the optic nerve sheath response to changing cerebrospinal fluid pressure: ultrasound findings during intrathecal infusion tests. J Neurosurg. 1997 Jul;87(1):34–40. [PubMed]
- Kimberly HH, Shah S, Marill K, Noble V. Correlation of optic nerve sheath diameter with direct measurement of intracranial pressure. Acad Emerg Med. 2008 Feb;15(2):201–204. [PubMed] [Free full text]
- Qayyum H, Ramlakhan S. Can ocular ultrasound predict intracranial hypertension? A pilot diagnostic accuracy evaluation in a UK emergency department. Eur J Emerg Med. 2013 Apr;20(2):91–97. doi: 10.1097/MEJ.0b013e32835105c8.[PubMed]
- Blaivas M, Theodoro D, Sierzenski PR. Elevated intracranial pressure detected by bedside emergency ultrasonography of the optic nerve sheath. Acad Emerg Med. 2003 Apr;10(4):376–381. [PubMed] [Free full text]
- LagrèzeWA, Lazzaro A,Weigel M, Hansen HC, Hennig J, Bley TA. Morphometry of the retrobulbar human optic nerve: comparison between conventional sonography and ultrafast magnetic resonance sequences. Invest Ophthalmol Vis Sci. 2007 May;48(5):1913–1917. [PubMed] [Free full text]
- Watanabe A, Kinouchi H, Horikoshi T, Uchida M, Ishigame K. Effect of intracranial pressure on the diameter of the optic nerve sheath. J Neurosurg. 2008 Aug;109(2):255–258. doi: 10.3171/JNS/2008/109/8/0255[PubMed]
——– OPTIC NERVE SHEATH DIAMETER 1 :ONSD Right eye
——– OPTIC NERVE SHEATH DIAMETER 2: ONSD Left eye of Group I
——– Reference line